Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) is an idiosyncratic reaction to medication1 that is characterized by skin rash, hematologic alterations, and organ involvement.2 It has been related to the ingestion of phenytoin and other anticonvulsant agents.3 Skin reactions have been described in up to 19% of patients between 6 and 8 weeks after drug initiation.3 The mortality rate is above 10% and death is commonly secondary to acute liver failure (ALF).4
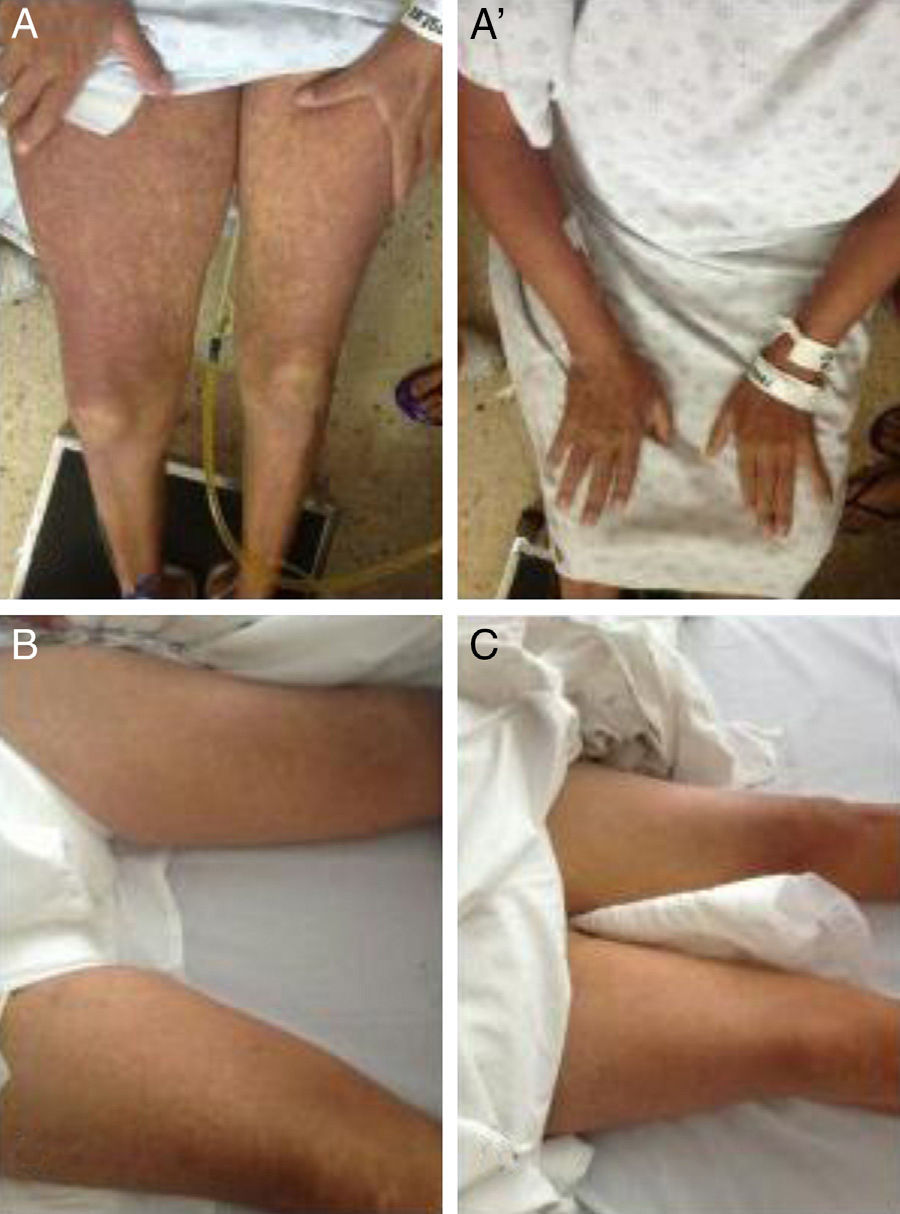
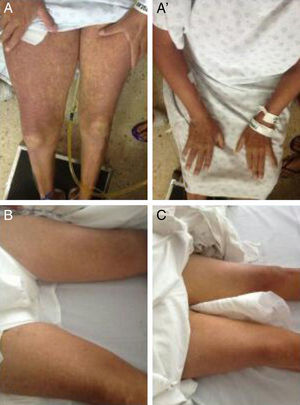
One month before her hospital admittance, a 46-year-old woman presented with subarachnoid hemorrhage due to a ruptured aneurysm of the posterior left cerebral artery; the affected vessel was clipped and she began adjuvant management with 100mg of phenytoin every 8h. She had no past history of herbal medicine or alcohol consumption, or prior use of other medication or hepatotoxic agents. Forty-eight hours after drug initiation, the patient noticed maculopapular lesions on both hands that resolved spontaneously, with no other symptoms. Three weeks later the maculopapular lesions became generalized and turned into an exfoliative dermatitis. She developed unmeasured fever along with pruritus, jaundice, and choluria. Upon hospital admittance the patient presented with dehydration, jaundice, generalized maculopapular lesions with fine flaking (fig. 1), cervical adenomegaly, hepatomegaly of 3cm under the costal margin, and no hepatic encephalopathy (HE). Her laboratory tests reported: leukocytes 5,000L–1, eosinophils 1,800L–1, urea 104mg/dL, creatinine 5.6mg/dL, total bilirubin 8.6mg/dL, albumin 2.9g/L, alanine aminotransferase 171 U/L, aspartate aminotransferase 333 U/L, alkaline phosphatase 751 U/L, gamma-glutamyl transpeptidase 1,814 U/L, prothrombin time 38%, and international normalized ratio (INR) 1.8. Cultures had no pathogen development, the viral panel was negative for hepatitis A, B, and C, and cytomegalovirus (CMV) and Epstein-Barr virus (EBV) IgM serology were negative. Abdominal ultrasound showed no chronic hepatopathy data, no biliary tract dilation, no vascular thrombosis, and no alterations in either kidney. Management was begun with prednisone 1g/kg of weight and pentoxifylline 300mg every 6h; on her second day in the hospital, the patient presented with stage 2 hepatic encephalopathy characterized by asterixis and bradypsychia. The data indicated: stage 2 HE, INR >1.5 and a duration <26 weeks in a patient without cirrhosis, corresponding to ALF; oral N-acetyl cysteine (NAC) 140mg/kg as a loading dose was added to her management, continuing with 17 doses of 70mg/kg every 4h, carrying out the complete dose in 3 days. Vitamin K 20mg/day was also administered. The maculopapular lesions decreased around 36h after treatment initiation and the HE disappeared. Kidney function and liver function tests improved (Table 1). Within the first month of progression, the maculopapular lesions resolved after generalized flaking, as did the jaundice.
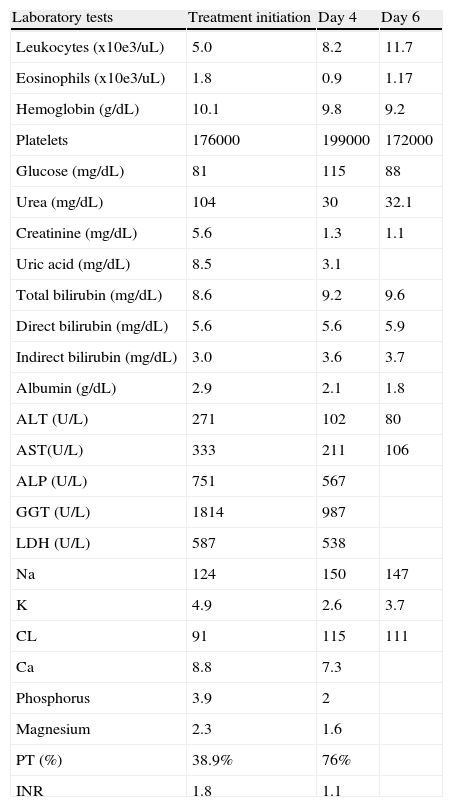
Laboratory test progression at diagnosis, day 4, and day 6.
| Laboratory tests | Treatment initiation | Day 4 | Day 6 |
| Leukocytes (x10e3/uL) | 5.0 | 8.2 | 11.7 |
| Eosinophils (x10e3/uL) | 1.8 | 0.9 | 1.17 |
| Hemoglobin (g/dL) | 10.1 | 9.8 | 9.2 |
| Platelets | 176000 | 199000 | 172000 |
| Glucose (mg/dL) | 81 | 115 | 88 |
| Urea (mg/dL) | 104 | 30 | 32.1 |
| Creatinine (mg/dL) | 5.6 | 1.3 | 1.1 |
| Uric acid (mg/dL) | 8.5 | 3.1 | |
| Total bilirubin (mg/dL) | 8.6 | 9.2 | 9.6 |
| Direct bilirubin (mg/dL) | 5.6 | 5.6 | 5.9 |
| Indirect bilirubin (mg/dL) | 3.0 | 3.6 | 3.7 |
| Albumin (g/dL) | 2.9 | 2.1 | 1.8 |
| ALT (U/L) | 271 | 102 | 80 |
| AST(U/L) | 333 | 211 | 106 |
| ALP (U/L) | 751 | 567 | |
| GGT (U/L) | 1814 | 987 | |
| LDH (U/L) | 587 | 538 | |
| Na | 124 | 150 | 147 |
| K | 4.9 | 2.6 | 3.7 |
| CL | 91 | 115 | 111 |
| Ca | 8.8 | 7.3 | |
| Phosphorus | 3.9 | 2 | |
| Magnesium | 2.3 | 1.6 | |
| PT (%) | 38.9% | 76% | |
| INR | 1.8 | 1.1 |
The exact incidence of DRESS syndrome is not known; it is estimated at 1 for every 1,500 new users of phenytoin and carbamazepine.3 Clinical symptoms are characterized by fever preceding the appearance of maculopapular lesions that advance to exfoliative dermatitis.3 The diagnostic criteria of the disease were described by Bocquet in 1996:1,2,5,61) suspicion of adverse reaction to medications, 2) >1,500 eosinophils and/or atypical lymphocytes, 3) 2 visceral disorders: cutaneous, diffuse adenopathy >2cm, hepatitis (> 2 times the normal transaminase value), interstitial nephritis, pneumonitis, pericarditis, myocarditis, thyroiditis, or meningoencephalitis.
The differential diagnosis with other types of reactions to medication should be made: Steven-Johnson syndrome and toxic epidermal necrolysis,1 in addition to ruling out infectious diseases (chicken pox, infectious mononucleosis) and pathologies such as Kawasaki syndrome, hypereosinophilic syndrome, and drug-induced pseudolymphoma.1
Its pathophysiology is unknown, but it has been associated with immunologic factors and genetic predisposition. Detoxification defects leading to the formation of reactive metabolites, activated T lymphocytes that release cytokines (IFN-γ and IL-5), exerting cytotoxicity, have been proposed;7 others have considered it to be a systemic reaction due to reactivation of the human herpes virus 6 and 7, EBV, or CMV.1,5
The drugs most associated with the development of DRESS are the anti-epileptic agents (phenobarbital, carbamazepine, phenytoin, sodium valproate, lamotrigine), allopurinol, sulfasalazine, dapsone, and minocycline. Antibiotics or nonsteroidal anti-inflammatory analgesics have rarely been reported.2 The disorder is independent of the quantity of the drug, having a greater relation to the susceptibility of the individual.
The first step of treatment is the definitive suspension of the medication at fault. Local steroids and antihistamines can be used in cases that are not severe. There are no criteria for the systemic use of steroids, but they are generally used in patients with organ involvement.2,4 In cases of ALF, 3 doses of intravenous methylprednisolone at 1mg/kg of weight, followed by oral steroid, has shown good results.7
Liver transplantation (LT) is the treatment of choice for patients with ALF that do not respond to high doses of steroids.8 NAC has been proposed as part of the treatment for patients with ALF secondary to the ingestion of paracetamol, as an antidote to intoxication by that drug, and some studies have shown its usefulness in patients with ALF due to the ingestion of other medications.9,10 NAC combined with steroids has been reported mainly in patients with anticonvulsant-induced DRESS, with a favorable response,10 but the number of studies supporting its use is insufficient.9
Our patient's illness was induced by phenytoin and upon admittance she showed data of ALF and acute renal failure (ARF) (probably due to interstitial nephritis). She was successfully treated with the combination of oral steroid and NAC and the suspension of phenytoin. We believe that early initiation of the double therapy in ALF patients can lead to improvement, averting the need for LT.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Pérez-Reyes E, Casanova-Lara A, Pérez-Torres E, Córdova J. Falla hepática aguda en una paciente con síndrome de DRESS que revirtió con N-acetilcisteína y prednisona. Reporte de caso y revisión de la literatura. Revista de Gastroenterología de México. 2014;79:208–210.