Strongyloidiasis is an endemic disease in tropical and subtropical regions. It is reported more frequently in immunosuppressed patients and presents with abdominal pain, diarrhea, constipation, nausea and/or anorexia.1 It usually compromises the duodenum and is rare in the colon.
Its prevalence varies, reaching 25% in some areas, and is estimated at 2.7% in Peru. It affects up to 40% of the population in the Peruvian Amazon.2
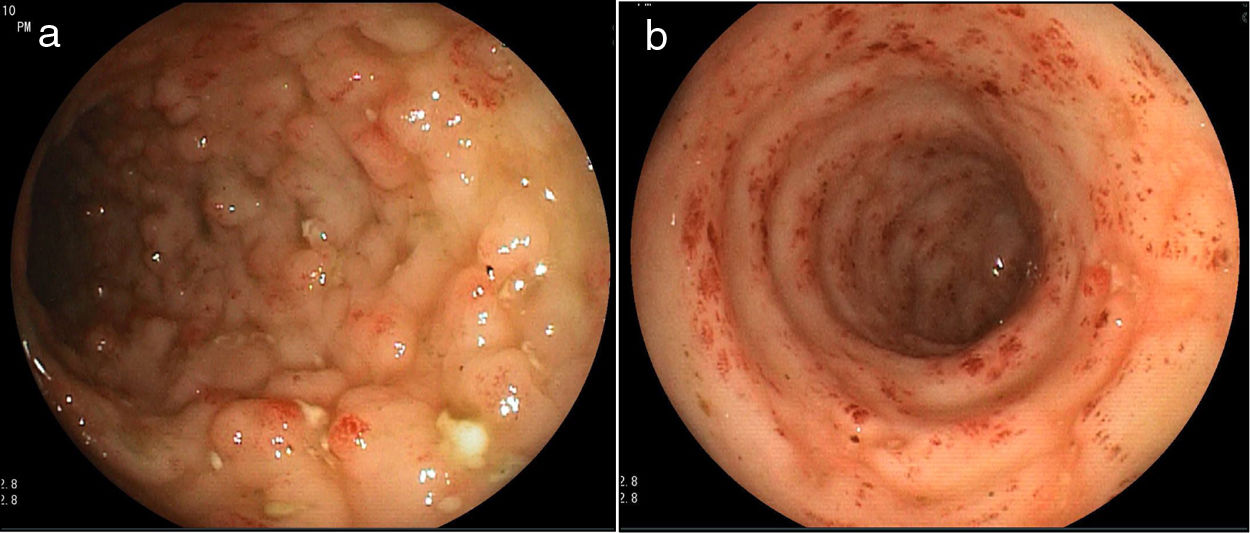
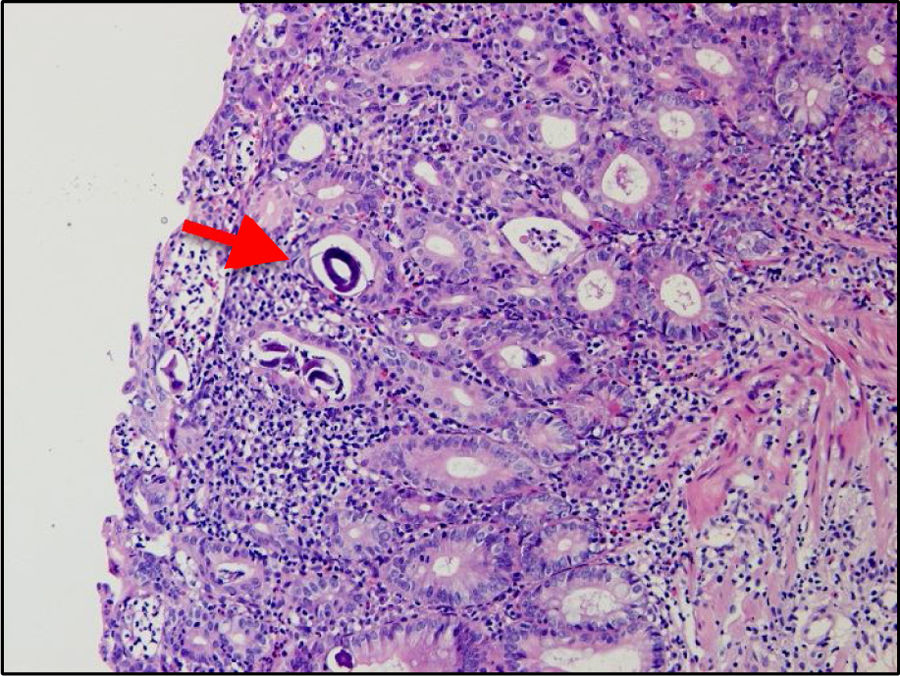
A 32-year-old woman presented with 3-week progression of abdominal pain, liquid stools with mucus but no blood, nausea, vomiting, and weight loss. Physical examination revealed white plaques in her oral cavity and a soft, depressible abdomen whose lower half was painful. Digital rectal examination showed no signs of bleeding. Laboratory test results were: hemoglobin 10.1g/dl, leukocytes 7,900/μl, eosinophils 11.2% (880/μl), total proteins 5.2g/dl, albumin 3g/dl, fecal leukocytes 50–80/per field, red blood cells: 0/per field, negative parasitologic testing, and a positive ELISA HIV test. From the cecum to the proximal rectum, colonoscopy identified vascular pattern loss, shortening of the haustra, mucosal edema, erythema, some 3−4mm ulcers covered with fibrin at the base, and segments with a nodular aspect (Fig. 1a and b). The histology study findings were 30–40 eosinophils per high power field, acute and chronic inflammatory infiltrate, mild distortion of the glandular architecture, and rhabditiform Strongyloides stercoralis larvae (Fig. 2). The evaluation was completed with upper gastrointestinal endoscopy, distinguishing erosions in the duodenum, pale areas, and absence of villi, with histologic evidence of Strongyloides stercoralis larvae.
The patient was treated with 200μg/kg/day of ivermectin for 2 days, with favorable clinical progression, and a third dose 2 weeks later.
Strongyloides stercoralis has a complex life cycle, with the capacity to live and replicate in the host for decades because the rhabditiform larvae mature into filariform larvae in the gastrointestinal tract, penetrating the perianal skin or the colonic mucosa to complete the autoinfection cycle.3,4
The risk factors for acquiring the infection are male sex, low socioeconomic status, alcoholism, white race, immunosuppression, and occupations in which there is contact with soil, such as agriculture and mining.5
The clinical picture is composed of 4 aspects: a) acute strongyloidiasis, with local signs of skin irritation due to the entrance of the larvae, and bronchitis due to their migration to the lungs; b) chronic strongyloidiasis, which is asymptomatic in the majority of patients, but others can present with diarrhea, constipation, abdominal pain, nausea, or asthma; c) superinfection, characterized by a cycle of accelerated autoinfection that exacerbates the gastrointestinal and pulmonary symptoms and generally develops in immunocompromised patients; and d) dissemination, a form of superinfection that includes areas outside of the normal life cycle (gastrointestinal tract, peritoneum, and lungs), in which larvae can be found in the central nervous system, liver, kidney, and other organs.1,5
The presentation of superinfection, with or without dissemination, is a potentially lethal form whose development is generally related to immunosuppression, corticotherapy, post-transplantation, hematologic neoplasia. It is often associated with HTLV-1 coinfection, conditioning failure in the Th-2 lymphocyte response in charge of controlling the infection of the parasite.1,5,6
HIV has also been described as a risk factor for Strongyloides stercoralis infection. However, the Th-2 response is not reduced, explaining the lower risk for the disseminated disease. Our patient tested positive for HIV infection and negative for HTLV-1, developing symptomatic chronic strongyloidiasis associated with peripheral eosinophilia.1,6,7
Colitis due to Strongyloides infection can resemble ulcerative colitis, but its distinctive characteristics are: attenuated lesion in the distal colon and rectum, lesions in patches (areas of normal mucosa), eosinophilic-rich infiltrates, relatively intact crypt architecture, and frequent involvement of the submucosa.3,4,8,9 Colonoscopy can reveal mucosal edema, erosions, pseudopolyps, bleeding, and ulcerations, alternating with normal parts of the mucosa.
The differential diagnoses include inflammatory bowel disease (IBD), amoebiasis, colitis due to Shigella, Campylobacter, or Yersinia, drug-induced colitis, eosinophilic colitis, and ischemic colitis.9
Treatment for chronic strongyloidiasis is 200μg/kg/day oral ivermectin for 2 days, repeating the dose in 2 weeks. In severe cases, administration should be parenteral (subcutaneous) at a dose of 200μg/kg/day for a varied period of time (3–22 doses). Treatment and follow-up are recommended to be continued until a fecal culture in an agar plate for S. stercoralis is negative after 2 weeks of treatment, thus ensuring eradication and eliminating the autoinfection cycle.10
Therefore, clinical, endoscopic, and histologic suspicion of colitis due to Strongyloides infection is important in a patient with inflammatory diarrhea and endoscopic findings suggestive of ulcerative colitis, given that an erroneous diagnosis and late treatment can be fatal.
Ethical considerationsThe present scientific letter follows the guidelines of the 1975 Declaration of Helsinki. No authorization by an ethics committee or patient informed consent for receiving a treatment was required, given that we only described a case, respecting the patient’s privacy.
The authors declare that no personal information of the patient appears in the letter and patient anonymity was respected, making consent for publication unnecessary.
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that the patient data that appear in this article are completely anonymous.
Right to privacy and informed consent. The authors declare that they have respected the privacy of the patient data and have the consent for publication signed by the authors.
Financial disclosureNo specific grants were received from public sector agencies, the business sector, or non-profit organizations in relation to this study.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Gomez-Hinojosa P, García-Encinas C, Carlin-Ronquillo A, Chancafe-Morgan RP, Espinoza-Ríos J. Infección por Strongyloides imitando enfermedad inflamatoria intestinal. Revista de Gastroenterología de México. 2020;85:366–368.