Proton pump inhibitors (PPIs) have been associated with small intestinal bacterial overgrowth (SIBO), which increases with prolonged PPI use, and SIBO has been associated with irritable bowel syndrome (IBS).
ObjectiveThe aim of the present study was to study the prevalence of bowel symptoms in patients treated with PPIs in Mexico.
MethodsGastroenterologists in 36 cities surveyed patients treated with PPIs, utilizing an ad hoc questionnaire to determine the presence of bowel symptoms and IBS.
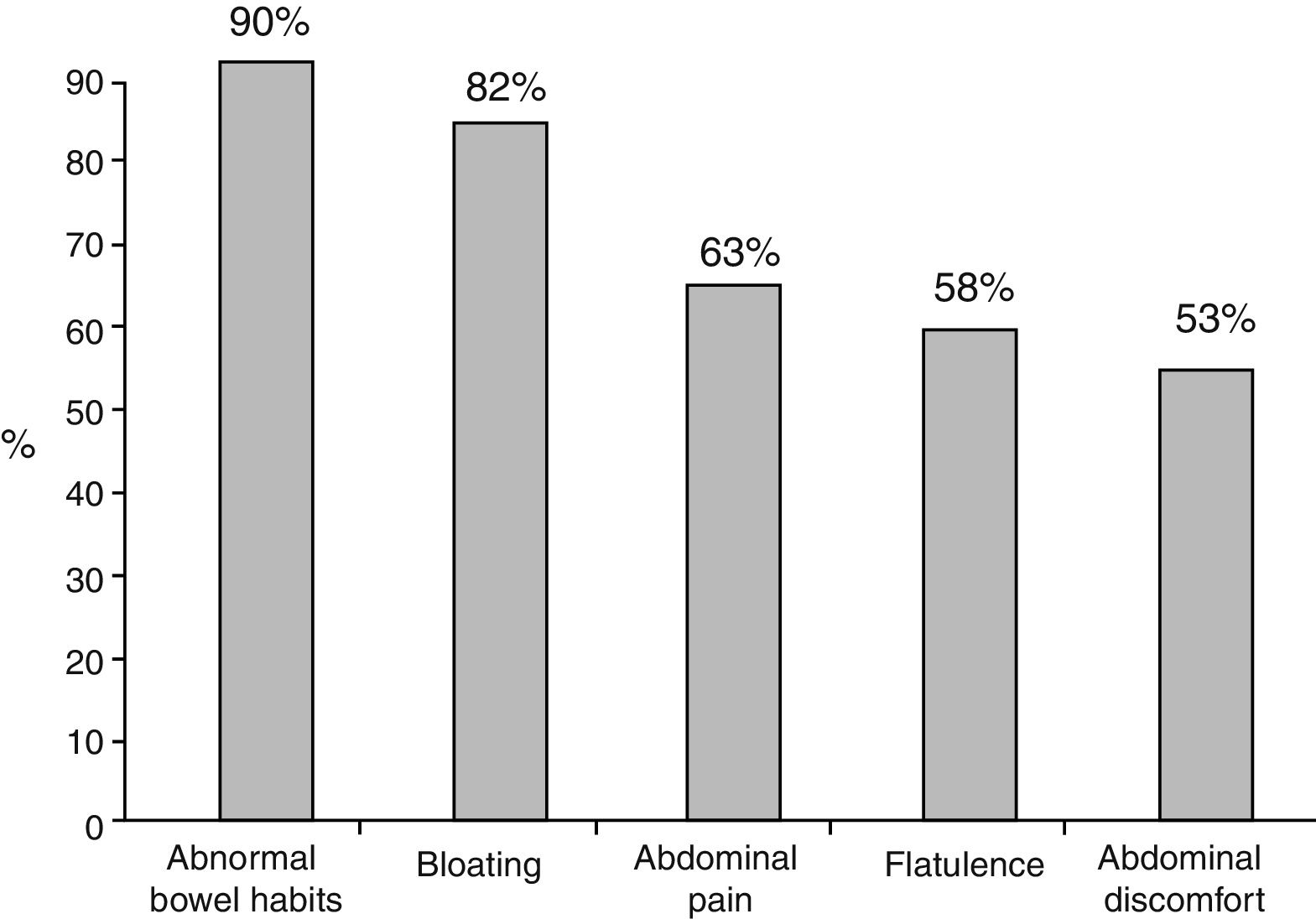
ResultsTwo hundred and fifteen physicians interviewed 1,851 patients. PPI indications were gastritis (48.8%), gastroesophageal reflux (38.5%), peptic ulcer (6.2%), and others (6.5%). A total of 77.5% of the patients received treatment for ≤6 months and 11.9% for ≥1 year. Symptoms were reported in 92.3% of the patients: abnormal bowel habits (90%), bloating (82%), abdominal pain (63%), flatulence (58%), and abdominal discomfort (53%). A total of 67.5% of the patients fit the Rome III criteria for IBS. Symptoms presented in 55.9% of the patients before PPI intake and in 44.1% of the patients after PPI use (P<.005). Constipation (63.8%) predominated in the former, and diarrhea (56.5%) in the latter (P<.0001). The treatments prescribed for managing those symptoms were antispasmodics, antibiotics, prokinetics, and antiflatulents, but patients stated greater satisfaction with antibiotics (mainly rifaximin) (P<.0001).
ConclusionThe association of PPIs with bowel symptoms and IBS is frequent in Mexico. Diarrhea and bloating predominate, and antibiotics produce the greatest treatment satisfaction, suggesting that SIBO or dysbiosis is the cause of the PPI-re
Los IBP se han asociado con sobrepoblación bacteriana del intestino delgado, lo cual se incrementa con el tiempo de uso, y esta se ha asociado con síndrome de intestino irritable (SII).
ObjetivoInvestigar la prevalencia de síntomas intestinales en pacientes tratados con IBP en México.
MétodosGastroenterólogos de 36 ciudades encuestaron pacientes tratados con IBP utilizando un instrumento ad hoc para determinar la presencia de síntomas intestinales y SII.
ResultadosDoscientos quince médicos incluyeron 1,851 pacientes. La indicación de IBP fue gastritis (48.8%), reflujo gastroesofágico (38.5%), úlcera péptica (6.2%) y otras (6.5%); el 77.5% recibió tratamiento por ≤6 meses y 11.9% por ≥1 año. El 92.3% reportó síntomas: evacuaciones anormales (90%), distensión subjetiva (82%), dolor abdominal (63%), flatulencia (58%), malestar abdominal (53%) y el 67.5% llenó criterios de Roma III para SII. En el 55.9% los síntomas se presentaron antes y en 44.1% después de IBP (p<0.005). En los primeros predominó el estreñimiento (63.8%), en los segundos diarrea (56.5%) (p<0.0001). Los tratamientos recetados para manejar estos síntomas fueron antiespasmódicos, antibióticos, procinéticos y antiflatulentos, sin embargo los pacientes consideraron la mayor satisfacción con antibióticos (rifaximina mayormente) (p<0.0001).
ConclusiónLa asociación de IBP con síntomas intestinales y SII es frecuente en México. Predominan la diarrea y la distensión subjetiva, y los antibióticos producen la mayor satisfacción al tratamiento. Lo anterior sugiere la sobrepoblación bacteriana del intestino delgado o disbiosis como causa de estos síntomas por IBP, lo cual debe ser confirmado.
The pharmacologic reduction of gastric acid secretion was revolutionary in the treatment of acid peptic disease, first with the H2 receptor antagonists 35 years ago, and then with the proton pump inhibitors (PPIs) 10 years later.1 Because of their efficacy and safety, PPIs have become the mainstay of treatment of acid-related diseases, such as gastroesophageal reflux disease (GERD).2 Severe adverse events related to PPIs are extremely rare and in general their use is considered sufficiently safe, even in the long term.3,4 However, an increase in the frequency of respiratory infections, including pneumonia,5–8 and gastrointestinal infections,9–11 including greater risk for Clostridium difficile infection,12–14 has been reported in patients taking PPIs.
Gastric acid secretion inhibition is followed by a greater frequency of small intestinal bacterial overgrowth (SIBO).15–20 A meta-analysis conducted on the relationship between PPIs and SIBO confirmed there was a significant association between treatment with a PPI and SIBO, when the latter was diagnosed through duodenal or jejunal aspirate culture. However, the association was not significant when SIBO was diagnosed through breath tests, most likely due to the different methodologies employed.21 The clinical importance of that data was recently reported in a study on a group of 200 patients that received PPIs. Fifty percent of them were diagnosed with SIBO through a glucose hydrogen breath test, compared with only 6% from a group of healthy individuals. In those patients, the prevalence of SIBO and the severity of symptoms related to SIBO were significantly greater with prolonged PPI administration lasting more than one year than with short-term treatment.22
The above suggests that patients under treatment with a PPI may have a high prevalence of digestive symptoms, especially of intestinal origin, as well as irritable bowel syndrome (IBS), which has been associated with SIBO and dysbiosis.22,23 Our hypothesis was that patients under treatment with any PPI would present with a higher frequency of bowel symptoms and IBS with PPI use, the longer it was taken. Thus, our aim was to study the prevalence of bowel symptoms and IBS in patients using PPIs in Mexico, as well as to investigate both the treatments through which physicians managed said symptoms and patient satisfaction with the different treatments.
MethodsWithin the time frame of October 2013 and March 2014, 2,044 gastroenterologists throughout the Mexican Republic were invited to participate in a study that consisted of answering a short questionnaire together with the patient during the medical consultation. The inclusion criterion was 10 consecutive patients seen in consultation that were under treatment with a PPI, regardless of its indication or patient diagnosis.
The questionnaire was specifically designed for the study and included questions about the presence of pain or abdominal discomfort, bloating, meteorism, flatulence, alteration in stool frequency or form, and the presence of IBS according to the diagnostic Rome III criteria; questions to determine whether symptoms began before or after PPI use and the relation of symptoms to the length of time of PPI use; and questions about the treatments utilized by the gastroenterologists for said bowel symptoms, as well as patient satisfaction with the different treatments.
The Ciencia Innovación y Mercadotecnia agency handled the statistical analysis, directed by the researchers. The categorical variables were described in percentages and the continuous variables in mean ± SD. The chi-square tests and Student's t test were utilized, as appropriate. Statistical significance was set at a two-tailed p < 0.05.
The protocol was approved by the Research Ethics Committee (CEDPOEC, the Spanish acronym). All the patients gave their consent to answer the questionnaire.
ResultsPhysicians and patientsOf the gastroenterologists invited, only 215 physicians from 36 Mexican cities accepted to participate. They interviewed 1,851 patients, of whom 1,092 (59%) were women and 759 (41%) were men, with mean ages of 47.5 ± 19 and 47.1 ± 19, respectively (p = 0.585).
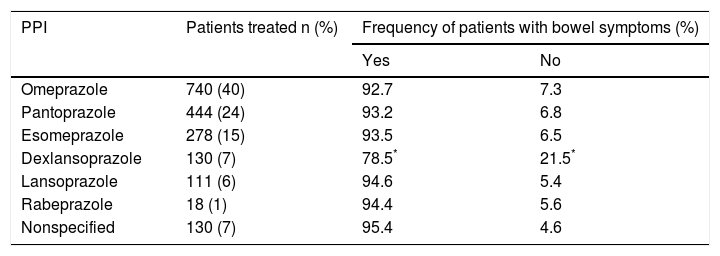
Proton pump inhibitorsPPI indications were gastritis (48.8%), GERD (38.5%), peptic ulcer (6.2%), and others (6.5%). The PPIs used in order of frequency were omeprazole (40%), pantoprazole (24%), esomeprazole (15%), dexlansoprazole (7%), lansoprazole (6%), rabeprazole (1%), and nonspecified (7%) (Table 1). Treatment duration with a PPI was one to 3 months in 56.9% of the cases, 4 to 6 months in 20.6%, 7 to 9 months in 3.7%, 10 to 12 months in 6.9%, and more than one year in 11.9%. The therapeutic indication was similar in the different treatment intervals, with the exception of a greater number of patients with GERD in the cases with treatment longer than one year. However, that difference was not statistically significant. PPI dose was not analyzed.
Frequency of bowel symptoms associated with each PPI utilized.
| PPI | Patients treated n (%) | Frequency of patients with bowel symptoms (%) | |
|---|---|---|---|
| Yes | No | ||
| Omeprazole | 740 (40) | 92.7 | 7.3 |
| Pantoprazole | 444 (24) | 93.2 | 6.8 |
| Esomeprazole | 278 (15) | 93.5 | 6.5 |
| Dexlansoprazole | 130 (7) | 78.5* | 21.5* |
| Lansoprazole | 111 (6) | 94.6 | 5.4 |
| Rabeprazole | 18 (1) | 94.4 | 5.6 |
| Nonspecified | 130 (7) | 95.4 | 4.6 |
Dexlansoprazole was the PPI less frequently associated with intestinal and abdominal symptoms.
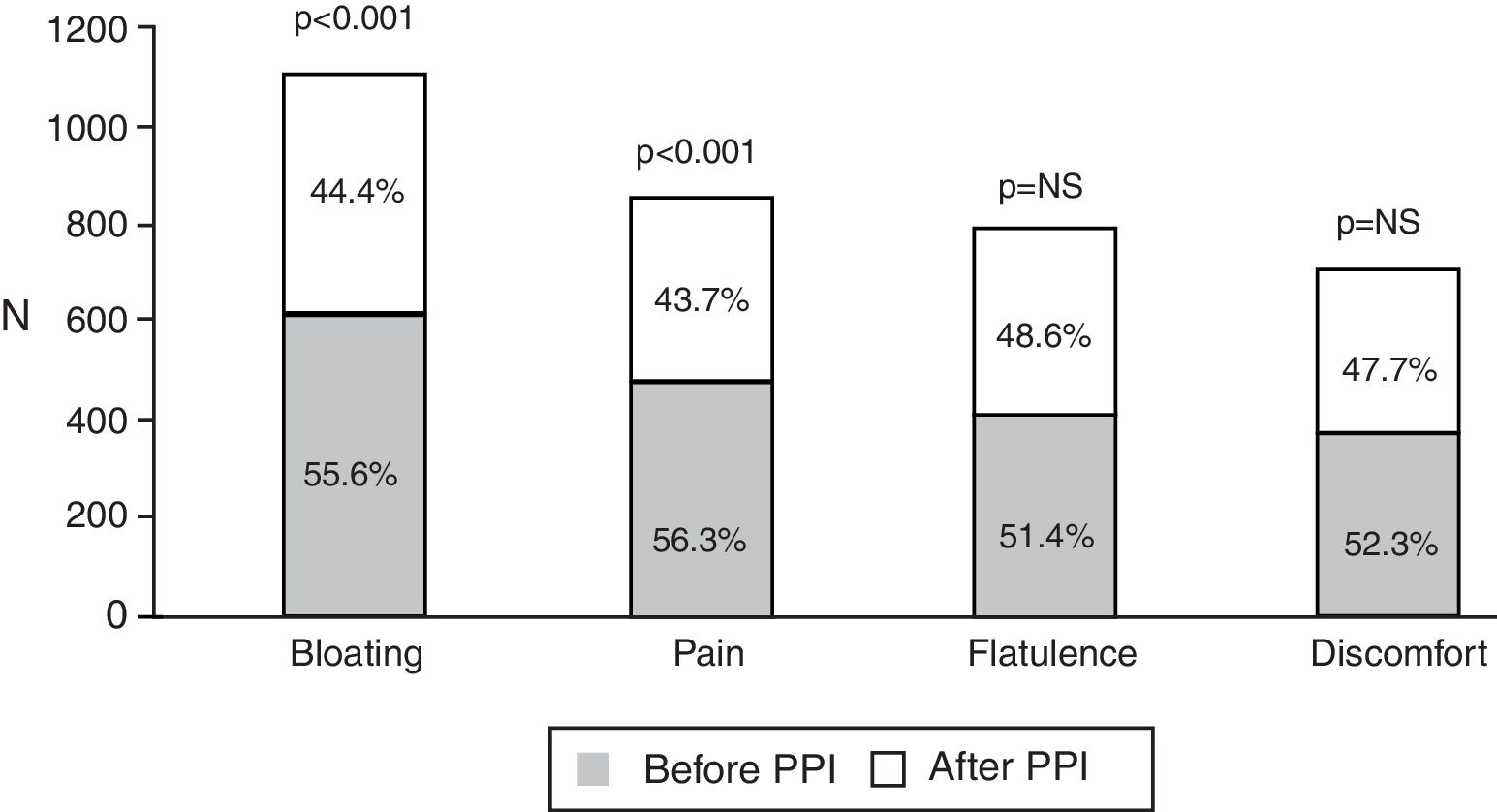
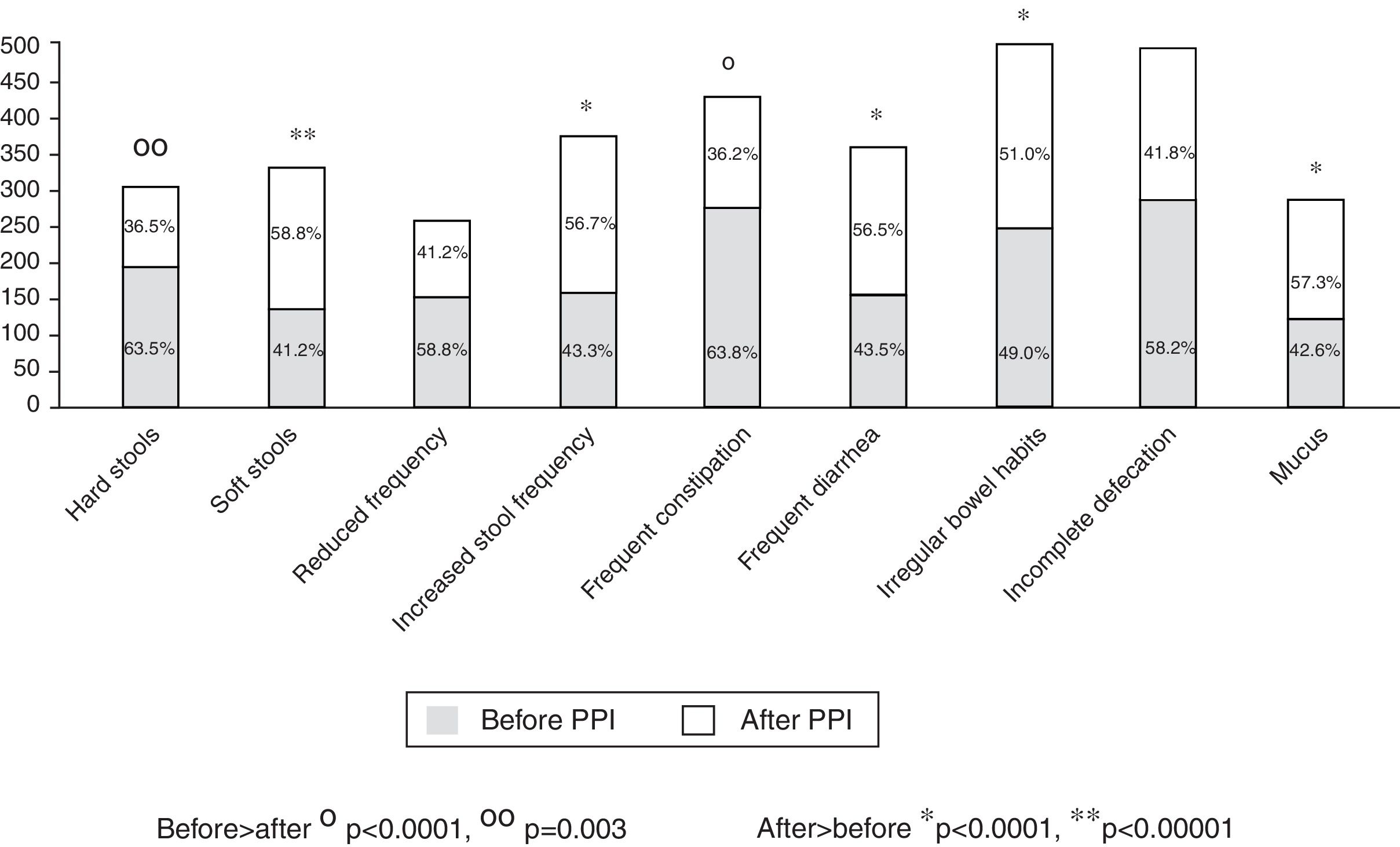
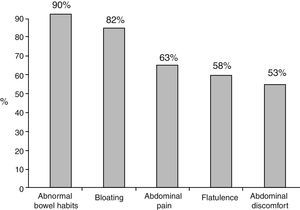
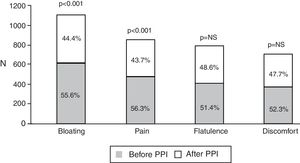
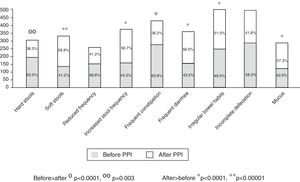
Of the total number of patients treated with a PPI, 1,708 (92.3%) reported an intestinal symptom. The most common were abnormalities in stool frequency, form, or both, followed by bloating, abdominal pain, flatulence, and abdominal discomfort (fig. 1). Abdominal symptoms such as bloating, abdominal pain, abdominal discomfort, and flatulence began before treatment commencement with PPIs in 55.9% of the cases and after treatment commencement in 44.1% (fig. 2). In contrast, irregular bowel habits began more frequently after PPI use (p < 0.0001). Symptoms related to diarrhea, such as soft stools (p < 0.00001), increased frequency (p < 0.0001), and the report of frequent diarrhea (p < 0.0001) appeared more commonly after treatment commencement with PPIs (p < 0.0001), whereas constipation symptoms, such as hard stools (p < 0.0001) and frequent constipation (p = 0.003) were present more frequently before treatment commencement with PPI. It should be noted that there was no difference in relation to reduced frequency of stools (fig. 3). In the cases in which symptoms began after PPI use commencement, symptoms appeared within the first 6 months of treatment in the majority of those patients (92%).
Frequency of bowel habit alterations according to their appearance before or after beginning treatment with a PPI. Symptoms related to constipation (hard stools and frequent constipation) were significantly more frequent before treatment commencement with a PPI, and symptoms associated with diarrhea (soft stools, increased stool frequency, frequent diarrhea, and mucus in the stools) were more frequent after treatment commencement.
Of the 1,708 patients with abdominal/intestinal symptoms, 1,249 (67.5%) met the Rome III criteria for IBS. In order of frequency, the IBS subtypes were distributed into IBS with diarrhea (IBS-D): 43%, unsubtyped IBS (IBS-U): 33%, IBS with constipation (IBS-C): 21%, and mixed-type IBS (IBS-M): 3%. In general, IBS was more frequent in women than in men (69.0 vs 64.6%, p = 0.038), but IBS-D specifically predominated in the male sex (47.8 vs 37.8%, p = 0.033). IBS symptoms were present before treatment with PPIs in 52.4% and after treatment commencement in 47.6% (p < 0.005). There were no differences in the frequency of IBS subtypes, regardless of beginning before or after treatment commencement with PPIs.
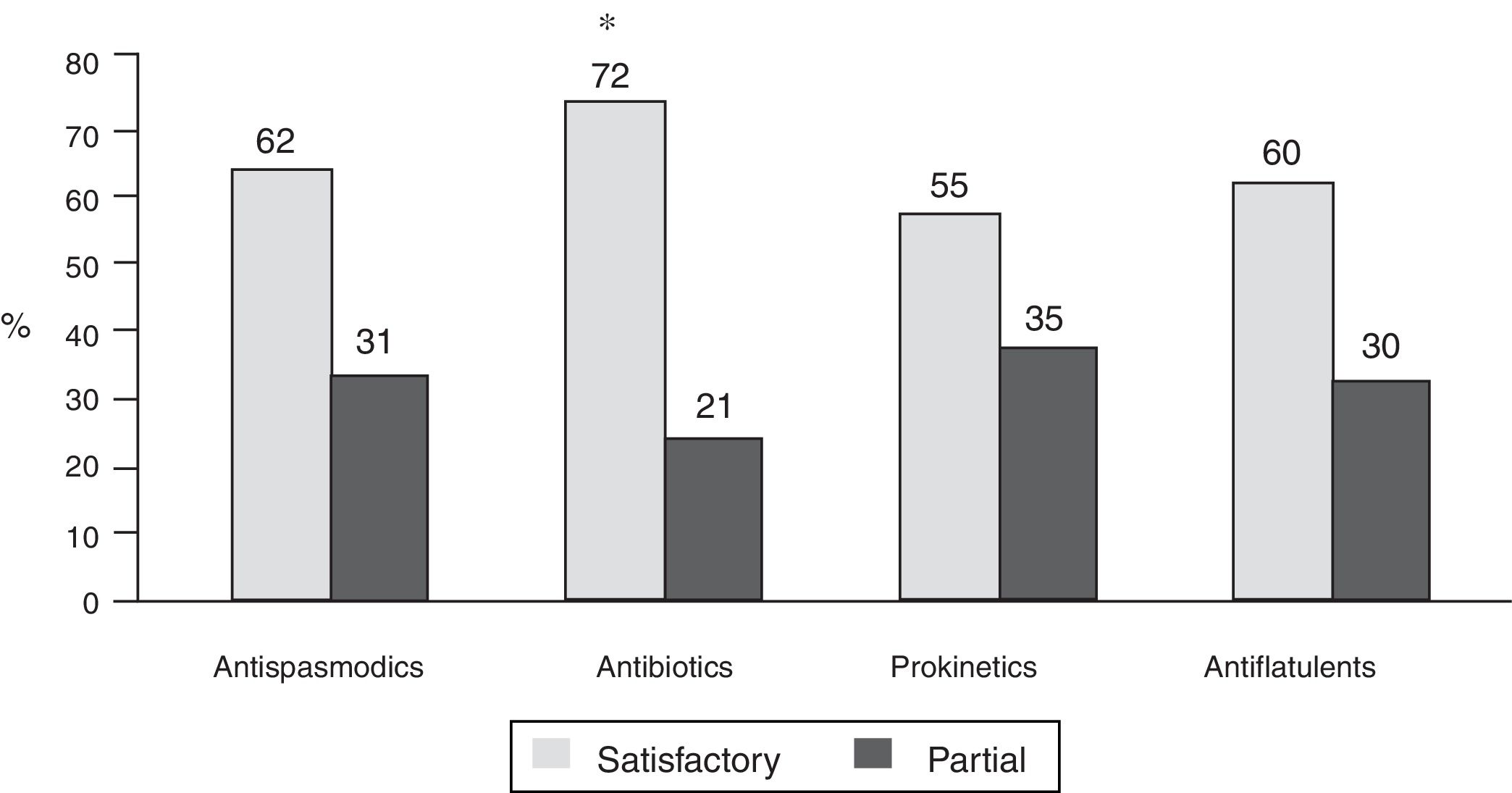
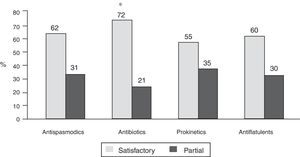
Bowel symptoms according to the proton pump inhibitor used and therapeutic conductThere was not much difference in the presence or absence of bowel symptoms in relation to the PPI used, with the exception of a lower frequency of symptoms with dexlansoprazole (78.5%), compared with the other PPIs evaluated (p < 0.05) (Table 1). In relation to the therapeutic conduct employed by the physicians to manage the bowel symptoms, 61% continued PPI use, 22% suspended PPI use, and conduct was unreported in 17%. In addition, the treatments utilized by the physicians for symptom management were antispasmodics (in 941 patients), antibiotics (in 524), prokinetics (in 439), antiflatulents (in 381), and others (in 31). It should be noted that physicians indicated more than one treatment simultaneously (combinations) in 35.6% of the patients. Fifty-five to 72% of the patients reported satisfactory response to treatments and 21 to 35% reported partial response. The patients that received antibiotics (rifaximin in 82.8% of the cases) more frequently reported satisfactory response (72%) and less frequently reported partial response (21%), compared with the other treatments (p < 0.0001) (fig. 4).
Discussion and conclusionsThe present study consisted of a survey of 1,851 patients under treatment with PPIs seen by gastroenterologists throughout the Mexican Republic. A total of 92.3% of the patients reported an abdominal/intestinal symptom and 67.5% of them met the Rome III criteria for IBS. In almost half the patients, symptoms began after treatment commencement with PPIs. Interestingly, the symptoms most commonly associated with PPI use were diarrhea and bloating, as well as IBS-D. Furthermore, in 2/3 of the cases, the physicians continued treatment with PPIs, but prescribed pharmacologic treatment for symptom management, with antispasmodics being the most frequently prescribed. However, the patients considered antibiotics, mainly rifaximin, the most satisfactory in regard to symptom improvement.
Our results suggest there are several aspects that must be taken into account. First is the indiscriminant use of PPIs in clinical practice, and for long periods of time (in 11% of the patients for more than one year), outside of the recommended guidelines.24,25 Second, the high frequency of symptoms of diarrhea and bloating, as well as IBS-D, in the patients with PPI use and the greater satisfaction with antibiotic treatment, mainly with rifaximin, suggest that those symptoms could be the product of SIBO or dysbiosis that have been associated with PPI use. Third, the fact that the physicians did not suspend PPIs in the majority of cases when symptoms appeared, suggests that they are not aware of the association of PPIs with those disorders and that there is a need for educational campaigns on the rational use of PPIs.
High frequency in the use of proton pump inhibitorsIn recent years there has been concern in the field of medicine in relation to the high frequency of PPI use in clinical practice and the elevated cost of those treatments. PPIs are accepted for treatment of GERD, peptic ulcer disease, H. pylori eradication, eosinophilic esophagitis, and Zollinger-Ellison syndrome. GERD is the main indication for PPI use, but in the majority of cases, the presence of GERD has not been well documented.26 Given that PPIs are a class of medications that are well tolerated and are considered to have one of the better safety profiles, their use has been indiscriminant, with no control over treatment duration and no thought of the potential adverse effects resulting from their long-term use. For example, interference with micronutrient (vitamin B12 and iron) and magnesium absorption, the relation to gastrointestinal infections due to hypochlorhydria, and the presence of a series of gastrointestinal symptoms that are difficult to demonstrate.27 This last condition is suggested in the present study, in which more than 44% of the patients presented with diarrhea and bloating with the use of those medications. In addition, PPIs are now considered the primary cause of medication-related acute interstitial nephritis and are a risk factor for chronic nephropathies and progression to their terminal phases.28 They have been associated with hyponatremia, due to excessive antidiuretic hormone (ADH) secretion29 and have been related to fractures from osteoporosis, to the infections of pneumonia and Clostridium difficile, and to rhabdomyolysis, anemia, and thrombocytopenia.30 They have also been associated with a modest increase in the risk for a first ischemic cerebrovascular disease event, regardless of platelet antiaggregants,31 and even have been associated with dementia. All of this has led to a careful reevaluation of the general use of PPIs and the FDA indications for their use.32
High frequency of abdominal/intestinal symptoms with proton pump inhibitorsOn the one hand, the most frequently reported digestive symptoms in the general Mexican population were abdominal pain, diarrhea, constipation, heartburn, and bloating. Likewise, the most frequent diagnoses after gastroenteritis were GERD and IBS.33 However, the findings of our study showed a surprisingly high frequency of abdominal/intestinal symptoms in patients that used PPIs. Those results do not appear to be explained simply by the coexistence of 2 common gastrointestinal disorders, such as the acid peptic disorders or GERD and IBS, even though it is well known that many patients with IBS present with symptoms of other functional gastrointestinal or extragastrointestinal disorders.34 In fact, a systematic review of the literature described much overlap between GERD and IBS, greatly exceeding the individual prevalence of either of the two disorders: more than 30% of the patients with IBS had GERD, whereas more than 40% of the patients previously diagnosed with GERD had IBS.35 In a Mexican prospective study, the authors found that individuals with IBS had a significantly higher frequency of heartburn, chest pain, epigastric pain, and nausea than the controls.36 The overlap of IBS symptoms with both GERD and functional heartburn suggests a pathophysiologic relation that is not yet clear.37
The possibility that the above can be explained by medication use, especially PPIs, as in our study, is supported by the findings of Choung et al. In a population survey about the relationship between medication administration and digestive symptoms, those authors observed a significant relation between PPI use and different functional gastrointestinal disorders: GERD, dyspepsia, bloating, and IBS. The association with IBS was independent of the presence of comorbidities such as dyspepsia and GERD.38 The true meaning of the PPI-IBS association has been questioned, given that it could simply be the presence of symptoms that are candidates for treatment with a PPI in patients with IBS, or that by reducing acidity, PPIs could facilitate the appearance of SIBO that would be associated with IBS.39 The cause-effect relation was demonstrated in a prospective study that evaluated the appearance of bowel symptoms and SIBO in 42 patients with non-erosive GERD that received esomeprazole for 6 months. During that time interval, 52% of the patients developed bloating, 33% flatulence, 24% abdominal pain, and 17% diarrhea. In addition, 19% of the patients met the Rome III criteria for IBS and 26% had a glucose breath test that was positive for SIBO.40 Long-term PPI administration has also been shown to alter bowel transit and favor the appearance of SIBO through that mechanism.41 The study by Lombardo et al.22 lends support to the causal relationship between PPI use and the presence of SIBO and bowel symptoms. They found that the prevalence of SIBO was greater and bowel symptoms were more intense in those patients that had received PPI for more than one year. The hypothetic mechanism by which PPIs can trigger bowel symptoms is that gastric acid secretion inhibition enables the passage of viable bacteria into the intestine, thus favoring bacterial overgrowth in the upper segments of the small bowel. With respect to that, it is striking that in an intensive care unit, the risk for presenting with C. difficile infection was 3.7 times higher in patients receiving PPIs than in those receiving H2 antagonists, which are weaker antacids.14 Treatment with PPIs has also been associated with the appearance of celiac disease, perhaps through enabling the passage of antigens into the submucosa.42
The majority of patients analyzed in the present study received PPIs for 6 months or less, and only 11.9% received that treatment for more than one year, with the resulting limitation that a relation between symptoms and prolonged treatment could not be established. Nevertheless, it was interesting that before beginning treatment with PPIs, a little more than half the patients presented with bowel symptoms that were mainly related to constipation, whereas the patients that presented with bowel symptoms after beginning treatment with PPIs had irregular bowel habits or diarrheic stools. That could suggest a pathophysiologic relation between bowel symptoms and SIBO. In our case series, 67.5% of the patients with bowel symptoms met the Rome III criteria for IBS. It could be speculated that PPIs produce IBS symptoms through SIBO and/or dysbiosis. Even though the results of the studies in a systematic review with a meta-analysis were heterogeneous, it was found that patients with IBS had breath tests that were positive for SIBO: 3.45-4.7 times more frequently than the asymptomatic controls.43 The microbiota is pathophysiologically related to IBS, not only through SIBO and dysbiosis, but also after enteric infections (postinfectious IBS), most likely through microinflammation production.44,45 A study on the stools of patients with prolonged PPI use (over 5 years) found that those patients had a decrease in the Bacteroidetes phylotypes and an increase in the Firmicutes phylotypes, and at the species level, had an increase in Holdemania filiformis and a decrease in Pseudoflavonifractor capillosus, compared with controls with no history of PPI use.46
In addition, all the treatments for bowel symptoms used in the patients of our study resulted in improvement, but a significant number of patients reported a more satisfactory response with antibiotics, especially with rifaximin. The greater patient response with the use of that luminal antibiotic is another factor supporting dysbiosis or SIBO as the cause of bowel symptoms with PPI use. Rifaximin has shown efficacy in improving symptoms in patients with IBS with no constipation, that is to say, in patients with IBS-D or IBS-M, as well as those with bloating, precisely the symptoms most frequently observed after beginning treatment with PPIs.43–45
Lack of awareness of the associations of proton pump inhibitors with gastrointestinal symptoms on the part of physiciansThe sudden increase in the literature reviewed above is clear evidence that the development of rational use of PPIs in clinical practice is required. Even though only 11.9% of the patients in our study used PPIs for more than a year, use above 6 months is now considered prolonged. The majority of guidelines and consensuses recommend a first treatment with a standard PPI dose for 8 weeks, and if there is no response, a second 8-week cycle at double dose. Despite the fact that more than 44% of our study patients presented with symptoms after beginning treatment with PPIs, treatment was not suspended in more than 60% of the cases, suggesting that educational campaigns on the adequate use of PPIs should be carried out in Mexico. In a Canadian study in which criteria were established for PPI prescription at an assisted-living center, the use of those agents was evaluated 9 months before and 12 months after criteria presentation. The publication of the criteria was associated with a reduction in PPI prescription during the first 6 months, and even though statistical significance was not reached, there was a reduction in monthly costs per resident. However, the prescription of PPIs began to increase 6 months after the publication of the criteria, demonstrating the difficulty in maintaining prescription conduct.47
Our study has certain limitations. It was a retrospective study conducted only on patients under treatment with PPIs, whereas the ideal would have been a prospective study on patients that began PPI use and their comparison with a cohort not under that treatment. There could also have been memory bias in relation to symptoms. Nevertheless, the change from the predominance of constipation before PPI use to the predominance of diarrhea and bloating after beginning treatment was of interest. And lastly, we did not have an objective measurement method for establishing the presence of SIBO, such as breath tests, or for the presence of dysbiosis, such as stool and colonic mucosa sequencing to determine the microbiota before and during the treatment with PPIs.48 However, the strengths of our study are that it is the first on the topic to be conducted in Mexico, it included a large number of patients from different regions of the country, and it demonstrated the elevated lack of awareness in relation to treatment duration with PPIs and the gastrointestinal effects associated with their use.
In conclusion, our study showed a high association between the presence of bowel symptoms and treatment with PPIs. In addition, at least 2/3 of the patients met the Rome III criteria for IBS. The patients that presented with bowel symptoms before treatment commencement with PPIs had symptoms predominantly associated with constipation, whereas those that presented with symptoms after beginning treatment predominantly presented with symptoms of diarrhea and bloating. However, PPIs were not suspended in the majority of cases, and the patients considered treatment with antibiotics the most satisfactory for improving the symptoms associated with PPIs.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureThe present research was financed by Alfa Wassermann, S.A. de C.V.
Declarations of potential conflict of interestMax Schmulson has been a Consultant for Alfa Wassermann, Commonwealth Diagnostics International Inc, and Takeda. He has also been a Speaker for Alfa Wassermann, Commowealth Diagnostics Inc, Mayoly-Spindler, and Takeda and has received research grants from Alfa Wassermann and Nycomed/Takeda. Alberto Frati is employed at Alfa Wassermann SA de CV.
Please cite this article as: Schmulson MJ, Frati-Munari AC. Síntomas intestinales en pacientes que reciben inhibidores de bomba de protones (IBP). Resultados de una encuesta multicéntrica en México. Revista de Gastroenterología de Mexico. 2019;84:44–51.