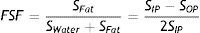The presence of liver fibrosis is the common denominator in numerous chronic liver diseases that can progress to fibrosis and hepatocellular carcinoma. Most important, with respect to frequency, are viral hepatitis and non-alcoholic fatty liver disease, the prevalence of which is increasing in epidemic proportions. Liver biopsy, albeit imperfect, continues to be the criterion standard, but in many clinical situations tends to be replaced with noninvasive imaging methods.
ObjectivesThe aim of the present article was to describe our imaging department experience with magnetic resonance elastography and to analyze and discuss recently published results in gastroenterology, hepatology, and radiology from other authors in the literature, complemented with a PubMed search covering the last 10 years.
Results and conclusionsMagnetic resonance elastography is an efficacious, noninvasive method with results that are concordant with liver biopsy. It is superior to ultrasound elastography because it evaluates a much greater volume of hepatic tissue and shows the often heterogeneous lesion distribution. The greatest advantage of the magnetic resonance protocol described is the fact that it quantifies fibrosis, fat content, and iron content in the same 25min examination specifically directed for that purpose, resulting in a favorable cost-benefit ratio for the patient and/or institution.
La presencia de fibrosis hepática es el denominador común de numerosas enfermedades crónicas del hígado que pueden evolucionar a fibrosis y a hepatocarcinoma. Las más importantes por su frecuencia son las hepatitis virales y el hígado graso no alcohólico, cuya prevalencia aumenta en proporciones epidémicas. La biopsia hepática, aun cuando imperfecta, continúa siendo el estándar de oro que tiende a ser reemplazado en muchas situaciones clínicas por métodos de imagen no invasivos.
ObjetivosDescribir la experiencia obtenida en nuestro departamento de imagen con la técnica de elastografía por resonancia magnética, analizar y comentar los resultados publicados por otros autores en la literatura reciente en gastroenterología, hepatología y radiología, complementada con una búsqueda en PubMed de los últimos 10 años.
Resultados y conclusionesLa elastografía por resonancia magnética es un método no invasivo y eficaz, cuyos resultados tienen concordancia con la biopsia hepática, presenta superioridad sobre los métodos de elastografía por ultrasonido debido a que evalúa un volumen mucho mayor de tejido hepático y muestra la distribución de las lesiones que, a menudo, es heterogénea. La mayor ventaja del protocolo de resonancia magnética descrito estriba en cuantificar en la misma sesión, además de la fibrosis, el contenido de grasa y de hierro, lo cual se realiza en una exploración dirigida específicamente a este fin, en un tiempo de 25 min y a un costo-beneficio favorable para el paciente y la institución.
Liver fibrosis is the cicatrization response that is a consequence of acute or chronic liver damage due to a variety of causes. It is a dynamic and potentially reversible repair process that is associated with hepatocellular regeneration. The activation of stellar cells causes fibroblast proliferation and excessive extracellular matrix deposit. Fibrous bands are produced, distorting the structure of the liver, forming scars and regeneration nodules. Consequently, liver function is altered and there is increased hepatic resistance to the blood flow, causing chronic liver disease (CLD), manifested by cirrhosis and complications such as: liver failure, portal hypertension, and hepatocellular carcinoma (HCC).1,2
The main causes of cirrhosis in the industrialized countries include chronic hepatitis C and B virus infections, immoderate alcohol ingestion, and nonalcoholic fatty liver (NAFL) that can progress to nonalcoholic steatohepatitis (NASH) and cirrhosis.
The prevalence of viral infections and alcoholism as causes of CLD remained stable between 1998 and 2008. In contrast, the prevalence of NAFL as a cause of CLD has increased from 46.8 to 75.1% within the same time frame.3 NAFL has become the most frequent cause of CLD in the United States and Europe, associated with the prevalence of obesity and metabolic syndrome. The continuing increase in NAFL prevalence is estimated to substantially contribute to the increase in CLD and will represent an epidemiologic burden in numerous countries, including Mexico.4–6
The diagnosis of fibrosis has recently gained importance due to the fact that its various causes can be prevented or treated, making it potentially reversible if the causal factor is eliminated.2,5 Clinical management of these patients requires knowing the stage of fibrosis and the frequently coexisting fat and iron contents, and their increase or reduction during the course of treatment.
Liver biopsy, traditionally considered the “criterion standard” for diagnosis has several limitations: it is invasive, costly, subject to complications, and allows only a very small portion of the organ (approximately 1/50,000 of its volume) to be examined. Histologic analysis has great intra and interobserver variability, with low efficacy in the initial stages of fibrosis (F1 and F2), and it does not provide information about the distribution of fibrosis in the parenchyma, which can be heterogeneous.7–9
Some imaging methods such as ultrasound (US), computed tomography (CT), magnetic resonance (MR), and ultrasound elastography (USE) overcome some or several of these limitations and have been increasingly used in recent years, providing a new noninvasive diagnostic option.
A more recently acquired technique is magnetic resonance elastography (MRE), developed by researchers at the Mayo Clinic in Rochester, New York, to obtain quantitative and qualitative information about liver fibrosis.10
This technique has continued to be perfected and has recently been made more attractive because during the same session, it can be combined with other MR sequences that are useful for quantifying the parenchymal infiltrates due to fat and/or iron, which enables a “multiparametric” examination protocol to be established.
The aim of this article was to describe the technique and clinical applications of the multiparametric liver MR protocol used in our department for the diagnosis and quantification of fibrosis, steatosis, and hepatic siderosis, during a 25-min specific examination that is clinically and economically viable for both the patient and the hospital center.
The present review is supported by the experience obtained within the last 18 months at our imaging department and by an ample review of articles published between 2006 and 2016 in journals of hepatology, gastroenterology, and radiology, in English and French, found in the PubMed database with the Keywords
fibrosis, liver fibrosis, elastography, US elastography, and MR elastography.
FibrosisElasticity and stiffnessThe mechanical behavior of the tissues of the human body is similar to that of viscoelastic materials. Liver elasticity is inversely proportional to parenchymal stiffness and the quantity of fibrous tissue. As the fibrosis increases, parenchymal stiffness increases and its elasticity decreases. This physical phenomenon can be measured through elastography techniques.
General principles of elastographyThe purpose of liver elastography is to obtain noninvasive and in vivo information about the mechanical properties of the parenchyma after submitting it to a type of stress that deforms it. Elastography can be performed through ultrasound (USE) or magnetic resonance (MRE).
Three basic stages are required to carry out MRE of the liver: a) excitation, which consists of transmitting stress to the organ by means of a source of movement that sends wavelengths that deform the parenchyma; b) data acquisition, which consists of registering the deformation produced in the tissue as a consequence of the applied stress; and c) the analysis and quantification of the data that the equipment presents in the form of images.11,12
Magnetic resonance elastographyThe physical principles of MRE have been explained in many specialized publications and so only a general description will be provided herein.10,11,13–15 They are based on the transmission of low-frequency longitudinal wavelengths of approximately 65Hz by means of an instrument placed at the rib cage close to the liver. The longitudinal wavelengths that penetrate the organ are transformed into transversal wavelengths, called cuts or shears, which are propagated by the liver parenchyma and calculated through the shear modulus. The measurements obtained are quantified in kilopascals (kPa).
EquipmentIn our department, MRE (also called Fibro-RM) is performed with 1.5 Tesla (Optima 450 w, General Electric, Milwaukee, Wisconsin, USA) RM equipment. The resonator has additional hardware that produces mechanical wavelengths that are sent into the liver and a specific software program for analyzing the information obtained.
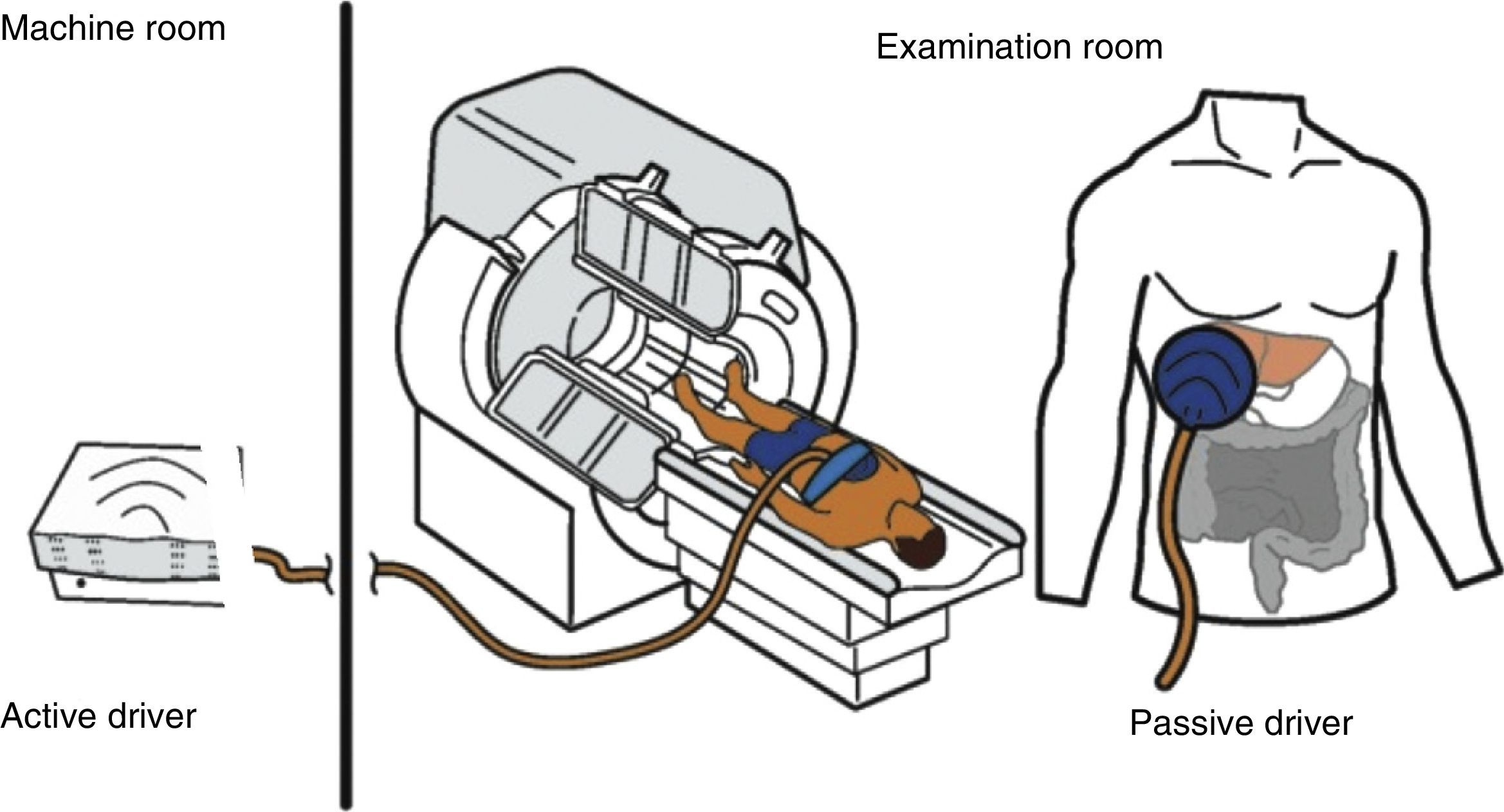
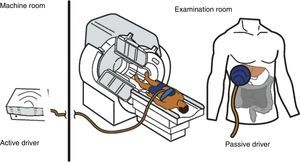
The hardware is made up of 4 mechanical elements (Figure 1): 1) a pulse or vibration generator that acts as an “active driver”, which is located in the neighboring machine room, 2) a flexible tube that transmits the pulses or vibrations produced by the driver, conducting them to 3) a plastic plate, 10cm in diameter, that is placed on the skin of the rib cage at the patient's liver, called the “passive driver”, and 4) an elastic band that attaches the plate to the body of the patient.
Diagram of the components of the magnetic resonance elastography equipment. The active driver that generates the pulses is in the machine room. The pulses are transmitted in the form of wavelengths through a plastic tube that connects that device with the plate. The plate is a passive driver, 10cm in diameter, that is placed at the liver and it transmits the wavelengths to the organ. The plate is secured to the body with an elastic band.
The information generated by the wavelengths that pass through the parenchyma is processed by elastography software that displays the data on a working station screen in the form of images in 4 different presentations (Table 1):
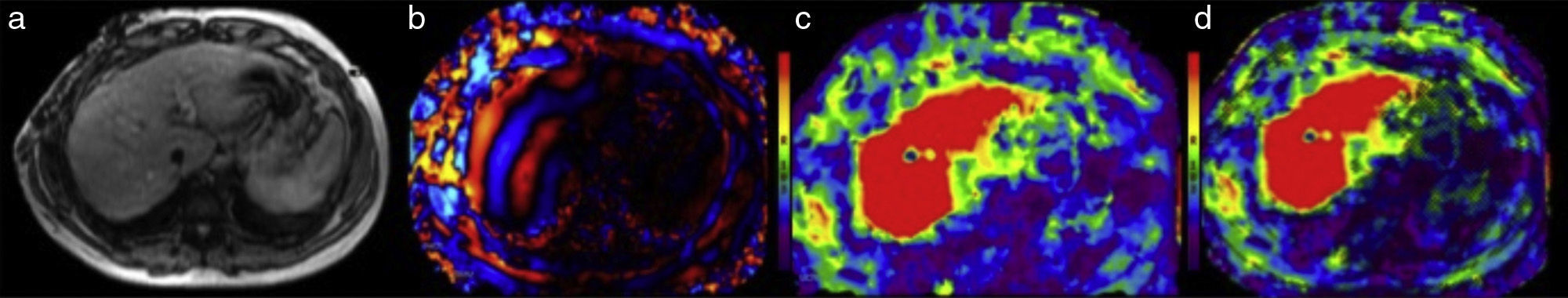
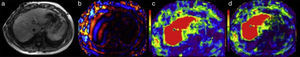
The first, is a low-resolution gray-scale reference image whose only function is to select the areas of the liver parenchyma that are to be measured, not including inadequate zones such as the portal vessels, large fissures, the gallbladder, etc. (Figure 2a).
The second image corresponds to a map of the wavelengths in color and motion that shows the advance of the wavelengths into the liver tissue (Figure 2b).
The third image, called an elastographic map, is a color map of the stiffness of the liver. A colorimetric scale is attached to each elastographic map (Figure 2c).
The elastography software creates a fourth image called the “confidence map” that outlines the zones of the liver that can be reliably measured (Figure 2d).
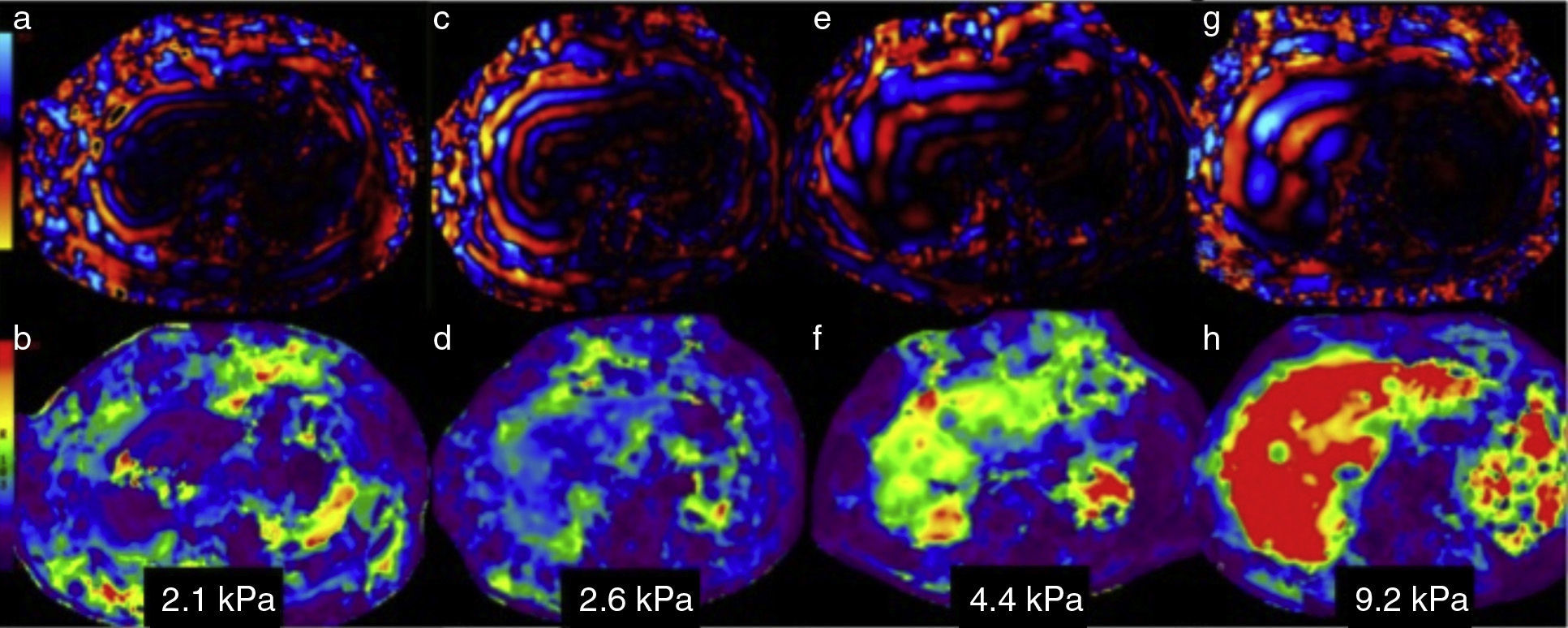
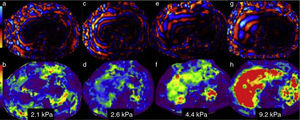
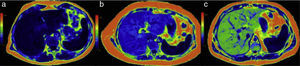
Image analysis and liver stiffness measurementWavelength mapWavelength analysis can be done statically in a fixed image or in a video showing the wavelength motion. It verifies that the wavelengths sent by the passive driver penetrated the liver tissue and were adequately distributed. Wavelengths are thin and parallel in the normal liver (Figure 3a). As the grade of fibrosis increases, the wavelengths become thicker and faster (figures 3c, 3e, and 3 g).
Information analysis. The wavelength map in 4 patients with different grades of stiffness is in the upper row. The lower row corresponds to the elastographic or color map of each of them. Figures a, c, e, and g show the increase in thickness and the irregularity of the wavelengths as the grades of fibrosis increase. The elastographic maps show the changes in color of the liver and their respective stiffness measurements: b) purples for 2.1kPa=F 1; d) greens and blues for 2.6kPa=F2; f) greens and yellows for 4.4kPa=F3; and h) red for 9.2kPa=F4. Compare the colors of the liver with the color bar on the left side.
This color map makes it possible to make a rapid visual evaluation of normality or abnormality of liver parenchymal stiffness. To make this assessment, the colors of the liver are compared with the colorimetric scale accompanying each image. This affords a first appreciation of the grade of fibrosis as absent, incipient, moderate, or intense (figures 3b, d, f, and h).
In our equipment, blue and purple tones are located at the lower end of the scale and correspond to minimum stiffness. Red is at the upper end of the scale and represents maximum stiffness, whereas yellow and orange tones correspond to intermediate stiffness values. The color map provides a visual appreciation of whether the fibrosis distribution is homogeneous or heterogeneous.16
The radiologist carries out the stiffness measurements by manually placing the electronic cursors on the reference image (taking care to avoid the portal vessels and gallbladder) or on the elastographic map in the zones of the so-called “confidence area”.
Automatic quantification programs have recently been developed that need no human intervention. Given that the elastographic map covers the entire circumference of the abdomen, it also makes it possible to recognize whether there is an increase in spleen stiffness in the same image (Figure 3 h).
Normal liver hardness values in magnetic resonance elastographyThe normal liver is soft and elastic with a mean stiffness of 2.05 to 2.44kPa and a range of 1.54 to 2.87kPa.15,16 The cutoff point for detecting fibrosis with MRE varies between 2.4 and 2.9kPa with 98% sensitivity and 99% specificity for the value of 2.93kPa.13,16,17
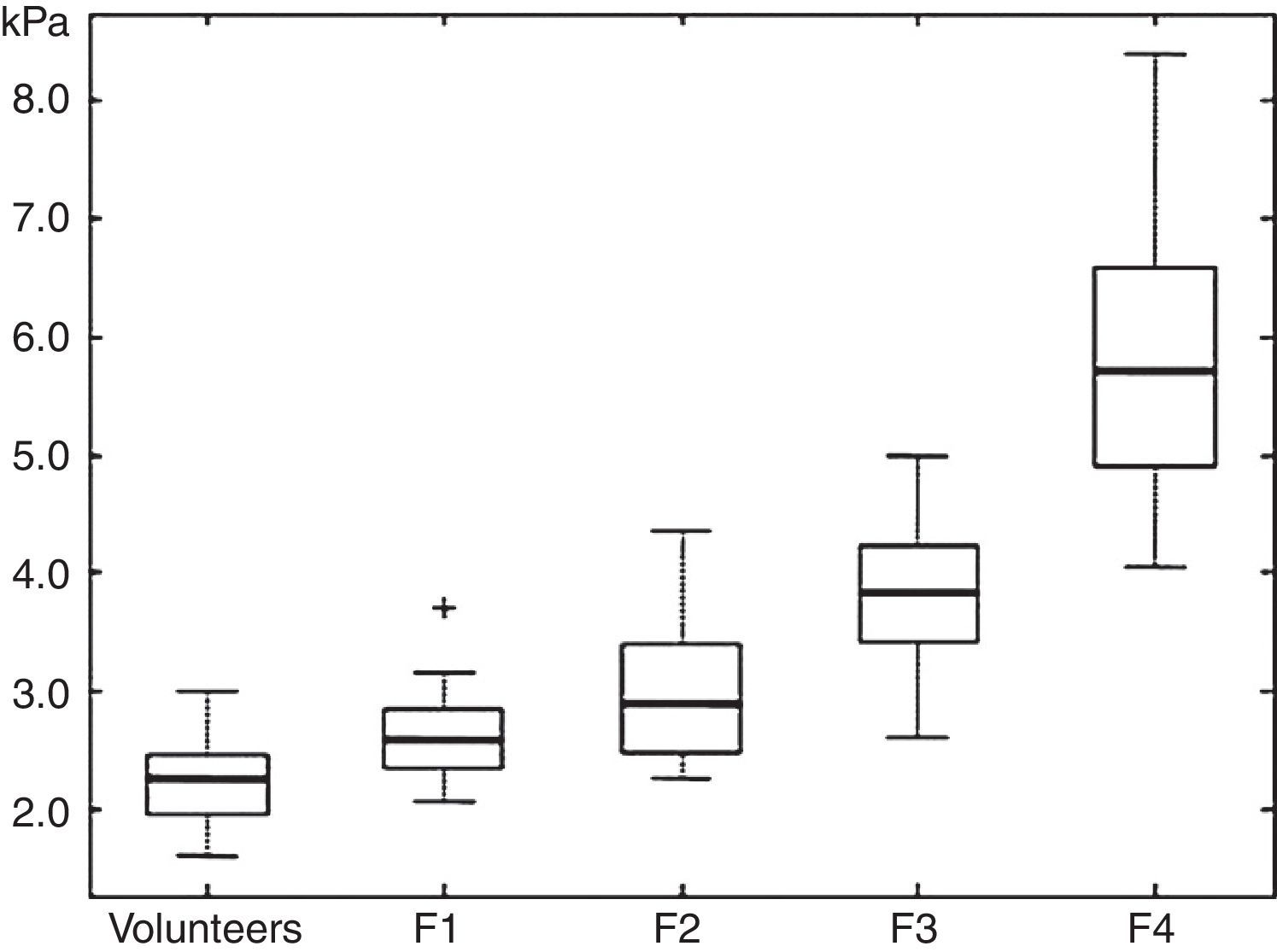
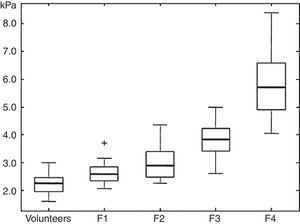
Huart et al. have correlated the kPa values with the METAVIR scale from F0 to F4.13 This scale was modified by Asbach et al., who formulated the table that we use in our practice to report the results of the study (Figure 4).14
Diagram showing the low, medium, and high quartiles of the shear modulus for fibrosis stages F1-F4, compared with volunteers.
(Reproduced with permission). Source: Asbach et al.14.
It is important to know that the kPa scale in MR is different from the one used in US. This is due to the fact that in US, the values are processed with the Young's modulus, which evaluates the longitudinal wavelengths on a scale from 2.5 to 75kPa. In contrast, in MRE, the values are processed with the transverse modulus of elasticity, or shear modulus, which measures the transverse wavelengths that are distributed in the parenchyma, also in kPa, but on a scale from 0 to 8kPa.
Clinical follow-up of fibrosis utilizing magnetic resonance elastographyDifferent studies have shown that MRE can detect liver stiffness modifications over the course of time. It is a repeatable and reproducible study that is not very operator-dependent and thus is a useful, noninvasive marker for monitoring the increase or possible decrease of liver fibrosis during clinical follow-up.15–17
Spleen stiffness measurementThe techniques of elastography employed for measuring liver stiffness have also been used to explore the spleen, under the hypothesis that the increase in splenic stiffness can be related to the development of portal hypertension and can be useful for classifying cirrhosis.
Measuring with ultrasoundSpleen stiffness can be measured with USE or MRE in a manner similar to that when measuring stiffness of the liver. The first USE studies were performed using transitory elastography equipment. However, recent studies report on the use of equipment that utilizes acoustic radiation force impulses (ARFIs) to generate shear wave images. This equipment makes it possible to measure the spleen under gray-scale and real time ultrasound image guidance, without measuring the splenic hilum vessels that can be prominent when there is portal hypertension, thus obtaining a more precise measurement.18–23
Measuring with magnetic resonanceA growing number of publications report on the usefulness of MRE for measuring the stiffness of the spleen with favorable results, compared with those of USE.24–30
It is possible to study the spleen during MRE of the liver, due to the fact that the elastographic map covers the entire circumference of the abdomen, which includes both organs.
The spleen can also be explored in the study area by placing the pulse-emitting plate directly on the splenic area.
Normal stiffness of the spleen measured by MRE varies from 2.35 to 5.6kPa, with a measurement of 3.6kPa in normal volunteers.27 Ongoing studies should provide greater knowledge on this subject matter.
Advantages of magnetic resonance elastographyMRE obtains information from the vectors of the wavelengths that are propagated in 2 or 3 dimensions in the liver parenchyma. The liver tissue volume analyzed in 2D MRE is calculated to be approximately 250cm3, compared with the volume evaluated through ultrasound methods, which varies from 0.5cm3 to 20cm3 (Table 2). Therefore, the volume of tissue analyzed in MRE is much more representative of parenchymal infiltration and reduces errors due to possible fibrosis heterogeneity.2 The MRE study is not limited by the presence of ascites or closed intercostal spaces. RM equipment utilization time and the cost of the study are considerably reduced when a specific elastography study is carried out.
Tissue volume quantified by the USE and MRE methods22
| Elastography method | Manufacturer | Volume |
|---|---|---|
| 1. Transitory elastography (Fibroscan®) | Echosens, FR | 4cm3 |
| 2. US ARFI pSWE | Siemens,Philips | 0.5-1.0cm3 |
| 3. US ARFI 2DSWE | Supersonics, Toshiba, General Electric (GE) | 20cm3 |
| 4. RM elastography | General Electric (GE), Siemens, Philips | 250cm3 |
The most important limitation is the presence of excessive iron deposition in the parenchyma due to hemosiderosis or hemochromatosis that can coexist with fibrosis in patients with CLD. When there is iron overload, the wavelengths are transmitted into the parenchyma well, but the signal emitted into the liver is too low (especially in 3T equipment) and measurement can be imprecise. Excessive obesity, bile duct obstruction, claustrophobia, and the inability to hold one's breath are causes that can limit the examination.
SteatosisMR offers various techniques for detecting and quantifying fat content in the liver parenchyma (Table 3). They are based on the different precession frequencies -resonance- of the hydrogen protons (1H+) in water and fat.31–33
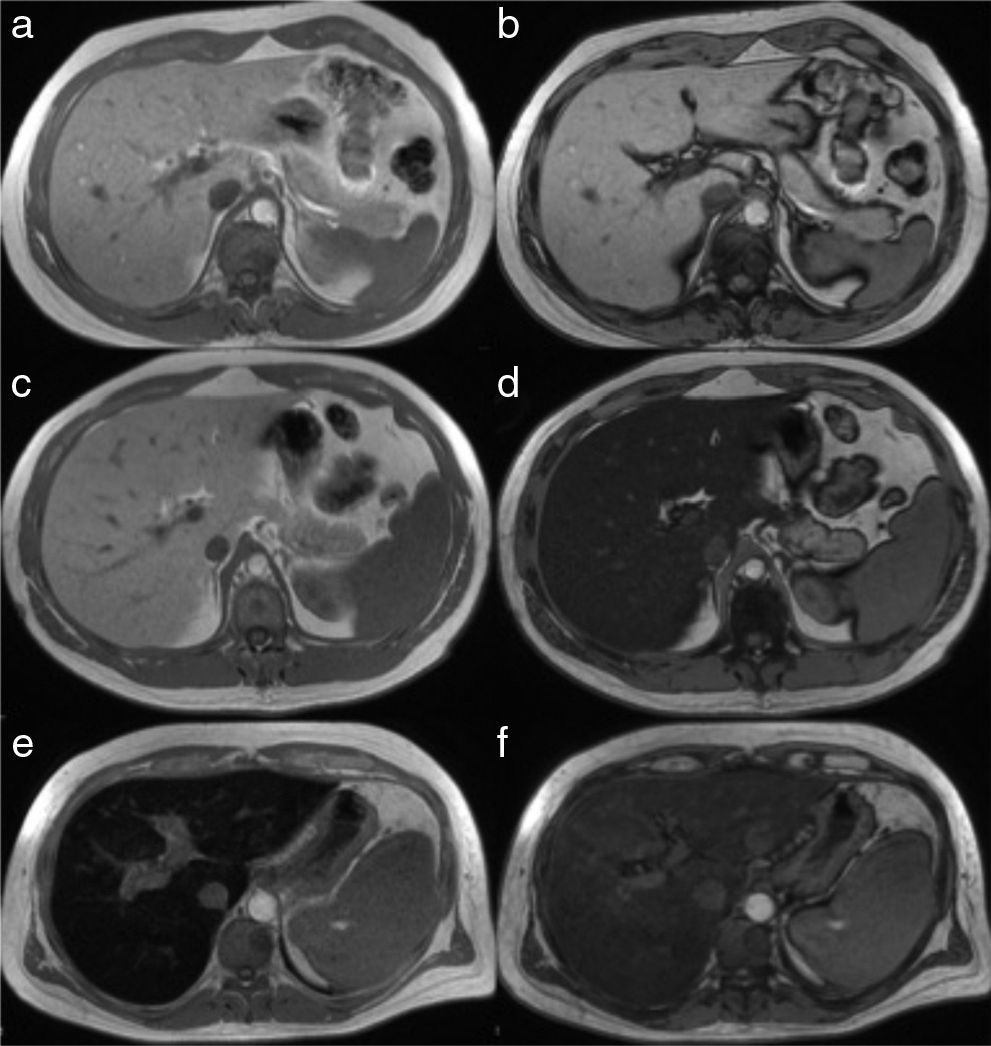
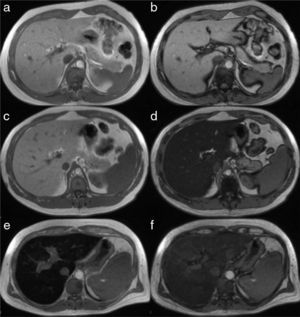
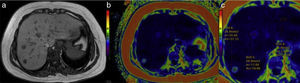
The simplest technique is the visual analysis of the image of the liver obtained in the dual echo T1-weighted sequence that makes up part of all MR liver protocols. It provides a pair of good quality anatomic images of the liver, “weighted to T1” and in 2 distinct acquisition phases: one “in-phase” (IP) and the other “out-of-phase” (OP) that enable the visual and qualitative detection of fat. In the normal patient, the liver parenchyma has the same signal in both the in-phase and out-of-phase images (Figure 5a and 5b). In patients with steatosis, signal decay in the “out-of-phase” images is produced, making the image of the liver turn darker, the greater the quantity of triglycerides (Figure 5c and d), enabling the fat fraction to be calculated, as described further ahead.
Image of the liver in the in-phase (left column) and out-of-phase (right column) T1 sequence in 3 different patients. In a normal patient, the signal is similar in the a) in-phase and b) out-of-phase sequences. The images in the center are those of a patient with steatosis in whom the intensity of the parenchymal signal is normal in the “in-phase” sequence (c) and it markedly decays in the “out-of-phase” sequence, where it acquires a tone of black due to the greater fat content (d). The bottom row corresponds to a patient with hemochromatosis. The signal decays in the “in-phase” sequence (e), due to the increase in iron saturation, compared with the “out-of-phase” sequence (f).
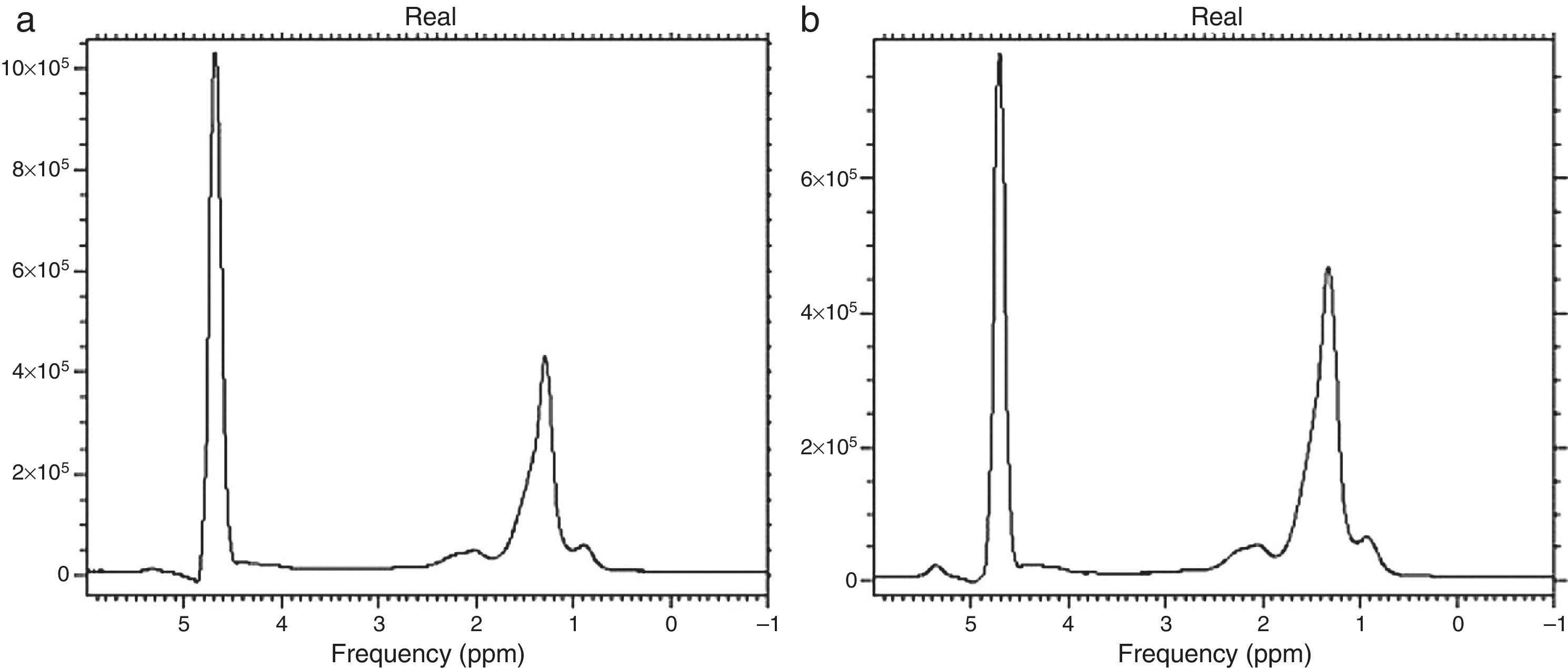
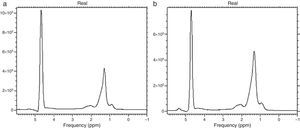
The most precise quantitative technique today for quantifying fat is MR spectroscopy (1H-RM). It separates the water curve spectrum from those of the lipid curves, providing an exact quantification. In the spectral curve, each metabolite is detected according to its resonance frequency, measured in parts per million (ppm). The water peak can be distinguished from various peaks produced by triglycerides and occasionally by other metabolites (Figure 6a and b). Spectroscopy is the noninvasive reference technique for diagnosing steatosis.32,34,35 Nevertheless, it is the most complex, slow, and costly. It requires specialized software and therefore is used only in academic and research studies.
Spectroscopy of lipids. Graphs a and b show 2 main peaks: on the left, an elevated peak that corresponds to water and on the right, a lower peak that corresponds to triglyceride concentration. Note that the triglyceride peak is higher in image b in a patient with a greater lipid concentration.
The most widely used technique is the one described by Dixon36 that measures the chemical shift between fat and water protons. It utilizes the dual echo T1-weighted sequence: the IP sequence contains the sum of the water and lipid protons and the OP sequence contains the absolute value of the difference of water minus lipids (IP-OP).
The fat content is calculated with the following equation:35
Where: FSF is the fat signal fraction; SFat is the fat signal; SWater is the water signal; SIP= is the “in-phase” signal, and SOP= is the “out-of-phase” signal
The current recommendation is to use the more recent techniques from the same family, such as the multi-echo 3D proton density sequences that provide the most precise fat fraction calculation, separating it from water and with a good histologic correlation. These are more precise, especially when steatosis coexists with iron deposits in the parenchyma that can falsify the reading.
The multi-echo sequences make it possible to eliminate the effect of iron, providing a more precise corrected fat quantification.32,34,35,37
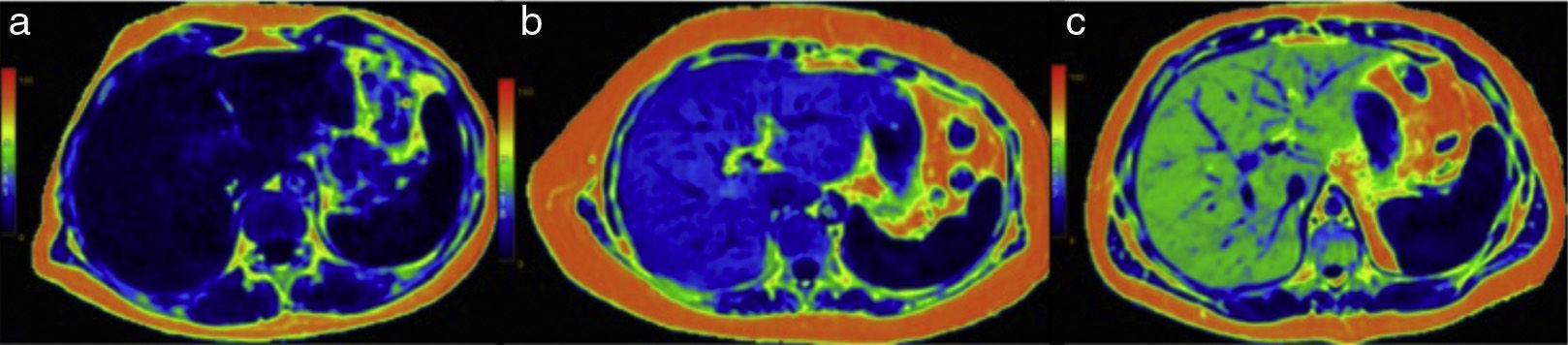
Steatosis quantification in color cartographyLiver fat quantification can also be carried out through cartography or a color map. It provides rapid visual appreciation of the existence of steatosis and the measurements are made directly on the image (Figure 7a, b, and c).
Color maps in 3 patients with different steatosis grades: a) patient with normal fat content: the dark blue of the liver corresponds to the lower part of the colorimetric scale and to a quantification below 6% (normal); b) in this patient the light blue is higher on the scale and the fat content is increased to an average 17%; c) the image in the third patient assigned green to the liver, which is indicative of a high fat content and was quantified at 33%.
The different colors of the map correspond to the distribution and intensity of the infiltrate that can be diffuse or regional (“geographic steatosis” or “in patches”), or even be present in a focal lesion, such as occurs in lipomas and some adenomas and hepatocellular carcinomas.
Cartography facilitates the analysis of fat content by comparing the colors of the colorimetric bar that is attached to the image. In our equipment, dark blue tones correspond to the lowest lipid concentration. Light blue and green tones indicate larger quantities of infiltrate, and yellow and red tones appear in the highest concentrations. The cartography image covers the entire circumference of the abdomen, and thus the subcutaneous fat and the peritoneal and perirenal spaces are also highlighted in red tones.
To quantify the liver infiltrate, the radiologist utilizes electronic devices that can choose the regions in which the percentage of the existing fat is to be calculated. The presence of fat within a focal lesion of the liver aids in its characterization (Figure 8).
Liver adenoma with fat. a) The T1 sequence shows 3 small focal lesions in segments 7 and 8 of the liver. The color map for quantifying fat shows that 2 of them have blue tones in the center (b), in which the lipid concentration was 37 and 19%, respectively (c). The histologic diagnosis was HNF1 adenoma.
MR can identify triglyceride infiltrates as low as 6-15%. The fat percentage figures obtained in the parametric image are reproducible in time, making them useful in the diagnosis, as well as in the follow-up of the progression of steatosis under treatment.
In 2 comparative, prospective studies, MR results have been more accurate than those of US and CT studies for the diagnosis and quantification of steatosis.38–40
Hepatic siderosisMR also has special sequences for studying the iron content in the liver parenchyma and in other organs, such as the pancreas, spleen, and the heart. It can accurately detect and quantify the amount of iron deposited in the liver, with reproducibility, and therefore is a useful tool for diagnosis and for monitoring treatment response. When there is a large amount of iron accumulation in the liver tissue, it distorts the magnetic field and produces signal decay in the T1, T2, and T2* sequences.
The dual echo T1 sequence can make a rapid qualitative appreciation of the increase in the hepatic iron content due to the fact that in this sequence the decay presents in the IP image (Figure 5e and f), in contrast to what occurs in the case of steatosis, as described above.
The most well-known quantitative technique in our environment, and the most widely used in our department, is that of Gandon et al., from the University of Rennes, France, published on the university website: http://www.radio.univrennes1.fr/Sources/EN/Hemo.htlm The study can be performed on various magnetic resonance machines, adapting the protocol to the magnitude of the available magnetic field, either at 0.5, 1.0, or 1.5 T. The calculation is made “online” with a Java application provided by the university.41
The method is efficacious for ruling out the existence of small parenchymal iron overloads, even when values are below 60¿mol Fe/g. In contrast, the efficacy of the technique can be lost when the deposits are very intense, because they cause complete loss of the resonance signal due to the effect that hepatic iron produces on the magnetic field.
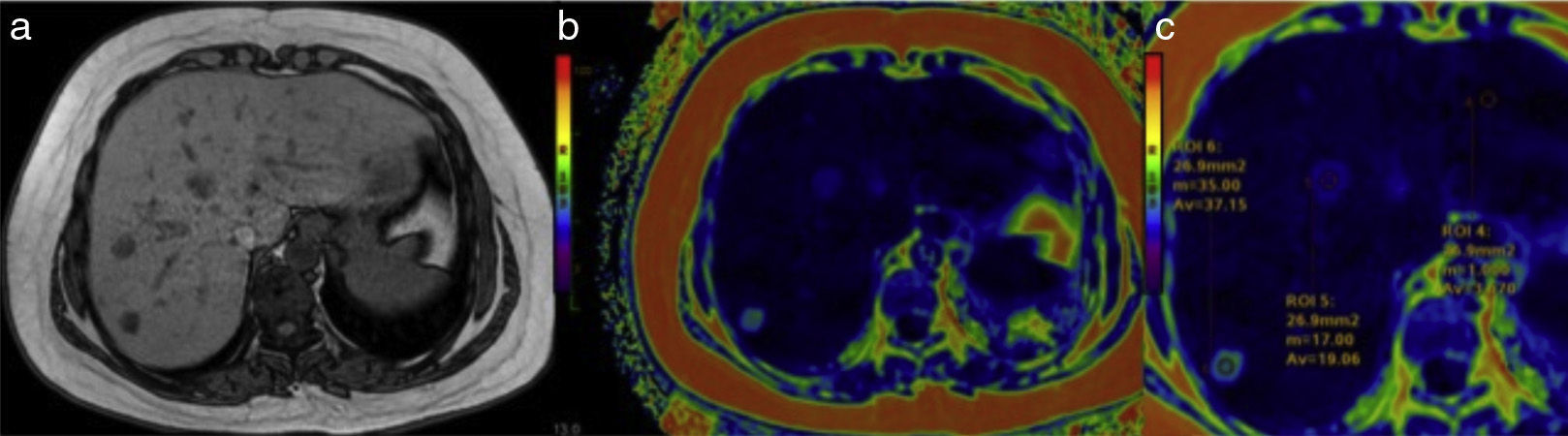
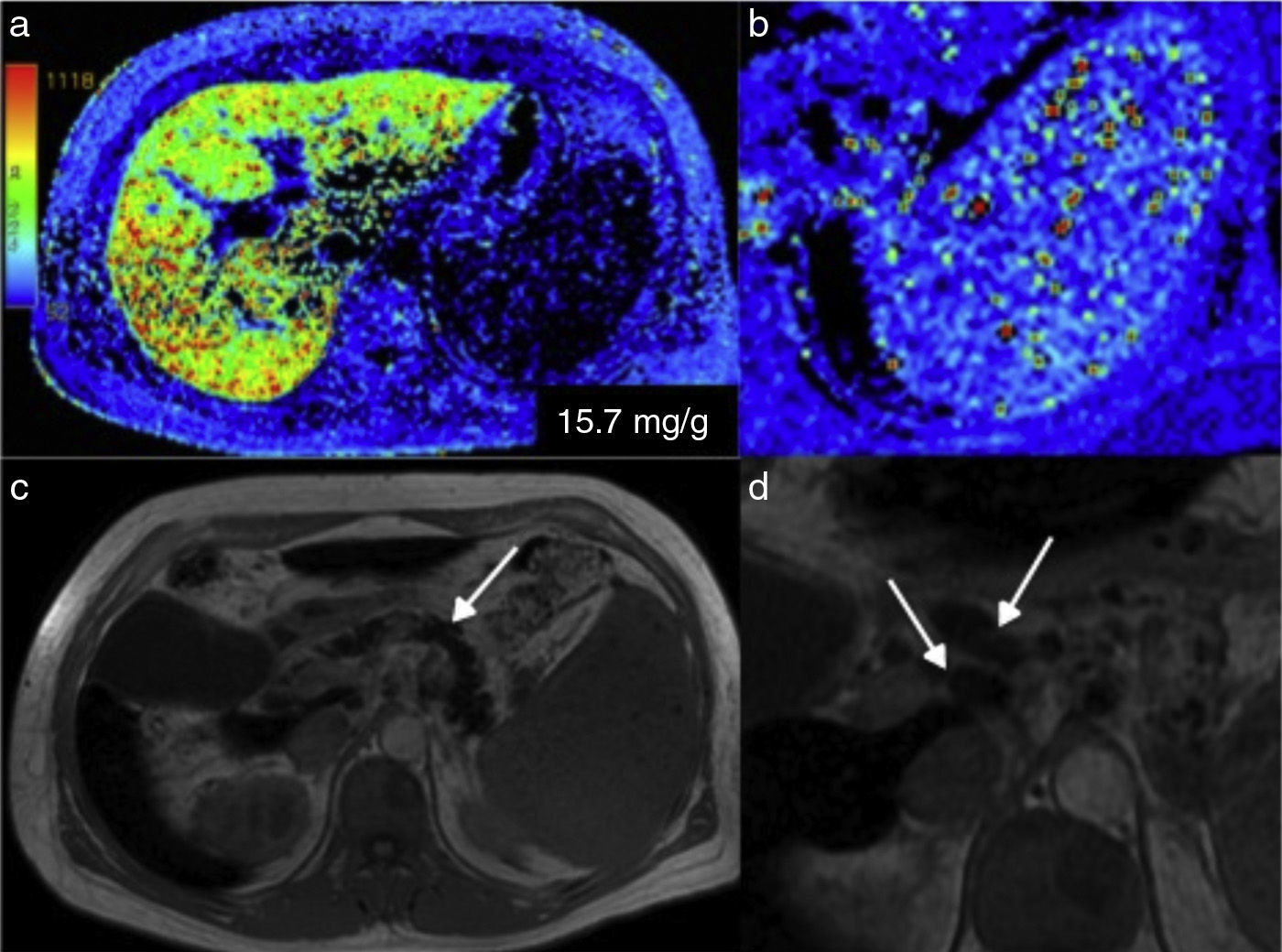
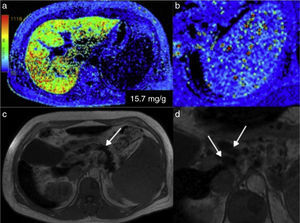
There are other techniques for evaluating parenchymal iron. Among them, the so-called MRI R2 and R2* technique can provide parametric images that reflect the iron content in the liver and spleen (Figure 9). The results of comparisons with liver biopsy have been dissimilar due to the heterogeneity of the iron deposits. They are generally similar, but MR has the obvious advantages in treatment follow-up and patient preference because of its noninvasiveness.42,43
Hemochromatosis. a) In the colorimetric map, the reds correspond to the higher part of the scale, indicating greater iron concentration in the liver parenchyma. b) Also note the red dot in the amplified image of the spleen produced by the Gamna-Gandy siderotic bodies. c) In addition, the anatomic image in T1 of this patient showed decay of the parenchymal signal of the pancreas (arrow) and the presence of enlarged peripancreatic lymph nodes (arrows) in relation to the hemochromatosis (d).
With the technique described by Gandon et al., values of 40 to 100¿ml/ml represent a slight iron overload in liver tissue, whereas values of 100 to 200¿ml/ml correspond to moderate overload, and those over 300¿ml/ml indicate major overload, which can saturate the sequences and falsify the readings.41 The MRI R2-R2* measures the parenchymal iron concentration in mg of Fe/g.42,43
Anatomic imageThe protocol utilized in our imaging department includes the presentation of 2 additional non-contrast enhanced MR anatomic sequences that are clinically useful: the abovementioned T1-weighted sequence with IP and OP images that also serve the purpose of qualitatively evaluating fat and iron, plus an axial T2 sequence. They provide an excellent anatomic image of the liver, the organs of the upper abdomen, and the retroperitoneum, including the gallbladder and bile ducts, the spleen, intestine, pancreas, pancreatic duct, kidneys, adrenal glands, aorta, and vena cava. They can detect benign or malignant focal lesions of the liver, in which case broadening the study to include sequences with endovenous contrast medium, whether gadolinium or gadoxetic acid (Primovist®), should be evaluated.
Discussion and commentsThe need for precise, reproducible, noninvasive methods that are capable of detecting the different stages of CLD derives from various facts, among which the following stand out: the increase in disease prevalence (which in the case of NAFL has been qualified as epidemic), improved knowledge of the pathology, and the greater therapeutic opportunities (that have also made the decisions that the clinician must make more complex).44,45
FibrosisEven though there are still few opportunities for cure in advanced stages of cirrhosis, with the exception of transplantation, early or intermediate stages of fibrosis are becoming entities that are treatable through preventive means and other possibly curative ones that can stop the progression or enable the regression of fibrosis, such as the new direct antiviral agents and certain experimental drugs.46–50
The diagnosis of fibrosis as the central factor in CLD pathogeny acquires greater significance in this context. Recent AASLD-IDSA guidelines point out that the correct evaluation of fibrosis is essential for evaluating treatment indication, and in some cases, its duration.51 In the consensus meeting on the use of different elastography techniques held in Denver, Colorado, in October 2014 that included infectologists, hepatologists, radiologists, pathologists, biomedical engineers, and FDA representatives, it was established that the priority for giving antiviral treatment to patients with hepatitis B and C viruses is currently determined by the presence or absence of moderate-to-intense fibrosis (F3 or higher).22
The MRE technique developed at the Mayo Clinic in Rochester has enriched our diagnostic capacity by providing the clinician with a wide range of information on fibrosis that other technologies cannot. Among them are:
- a)
The sample of tissue volume examined through MRE is significantly larger than that analyzed by ultrasound methods: Fibroscan®=approximately 4cm3; ARFI pSWI=0.5 to 1cm3; and ARFI 2D SWE=20cm3, compared with approximately 250cm3 of the MRE22 (Table 2).
- b)
A kinetic image is simultaneously obtained that confirms the passage of wavelengths in the amplitude and depth of the organ.
- c)
The presentation of a color elastographic map enables the visual evaluation of stiffness grade and the homogeneous or irregular distribution of fibrosis so that measurements directed by the image to the zone of interest can be made.
- d)
MRE and USE, whether ARFI pSWE or ARFI 2D SWE, provide the anatomic images of the liver and the organs of the upper abdomen in the same procedure, which is not the case with transitory elastography through Fibroscan®.22
- e)
The possibility of quantifying spleen stiffness is an extra advantage, and how this measurement might be a predictor of portal hypertension is discussed further ahead.52
The specific fibrosis study protocol can be broadened during the same MRE session with 2 additional sequences for quantifying the concentration and distribution of the fat infiltrate, and if so desired, the measurement of the iron content of the parenchyma, which frequently coexist in patients with CLD.
Unlike the USE techniques that have differing results depending on the equipment manufacturer, making it impossible to compare their equivalence, the 3 MRE manufacturers currently approved by the FDA (General Electric, Siemens, and Phillips) use the same shear wave management methods, processing algorithms, and image display, making their results comparable.22,53
This set of parametric data obtained noninvasively qualifies MR as the method that provides the most information, compared with the other diagnostic procedures, including biologic tests, USE, and even biopsy, with which it has shown excellent concordance.12,22,44,54
MRE is also the method that guarantees the greatest performance success: in a case series of 141 patients, Huwart et al. obtained successful studies with MRE in 133/141 (94%), compared with 118/141 (84%) with transitory elastography, in addition to obtaining greater diagnostic efficacy.13 Similar results have been reported in the recent study by Ichikawa et al.55
The efficacy and correlation of MRE with liver biopsy has been the motivation for an increasing number of studies. The 2012 meta-analysis by Wang et al. showed that the efficacy of MRE for diagnosing fibrosis provided figures of 0.95, 0.98, 0.98, and 0.99 under the AUROC curve.56 These authors also stated that liver stiffness measured with MRE increased in parallel with the grade of fibrosis: when there was greater stiffness, there was more fibrosis. Huwarth et al. reported that in 141 patients in whom liver biopsy was performed, the MRE areas under the AUROC curve were: 0.994 for F ≥ 2; 0.985 for F ≥ 3; and 0.998 for F=4.13
A meta-analysis carried out in 2014 by Su et al. that included 989 patients in 13 studies, showed great diagnostic efficacy of MRE for the detection, quantification, and staging of liver fibrosis. The mean sensitivity and specificity figures for: F ≥ 1, F ≥ 2, F ≥ 3, and F ≥ 4 were: 0.87, 0.92, 0.87, and 0.92, respectively.57
Another meta-analysis conducted by Sing et al. in 2015 that included 697 patients, 92.1% of whom had less than a one-year interval between MRE and biopsy, showed that the mean values of the area under the AUROC curve (with a 95% confidence interval) for the diagnosis of any stage: negative fibrosis (≥ stage 1), significant fibrosis (≥ stage 2), advanced fibrosis (≥ stage 3), and cirrhosis, were: 0.84 (0.76-0.92), 0.88 (0.84-0.91), 0.93 (0.90-0.95), and 0.92 (0.90-0.94), respectively.58 In addition, the percentage of procedure failures in the 697 patients collected in the same meta-analysis was 4.3%, a very low figure compared with other techniques. The authors concluded that MRE is highly efficacious for the diagnosis of significant or advanced fibrosis and that the result is independent of body mass and CLD etiology.
MRE is an efficacious tool for following the progression of patients with CLD and its measurements have been proven to be reliable, repeatable, and reproducible.59–61 The possibility of MRE replacing liver biopsy for diagnosing fibrosis has been expressed in several studies, emphasizing the fact that the tissue volume analyzed in MRE is substantially greater than that in liver biopsy, which is only 0.2% of the parenchyma, and there is no information about whether the distribution of the infiltrates is homogeneous or heterogeneous.13,16,43,44,59
Portal hypertensionExamination of the liver by MRE has also awakened the interest in studying splenic viscoelasticity and its possible relation to the development of portal hypertension. The correlation between the increase in stiffness of the spleen and the pressure gradient of the hepatic veins (HVPG) has shown promising results for the detection of severe portal hypertension and the presence or absence of esophageal varices.
The experimental studies carried out by Nedredal et al. and Yin et al.24,25 with MRE on animals found that there was a significant correlation between spleen stiffness measured with MRE and the HVPG. In their preliminary study on 38 patients with CLD, Talwalkar et al. demonstrated a significant correlation between the stiffness of the spleen and that of the liver, with increased splenic stiffness in the more advanced stages of liver fibrosis (Figure 3 h). A mean stiffness ≥ 10.5kPa has been considered a marker of high-grade esophageal varices.24,25,28
The majority of those studies have been based on the measurement of stiffness of the liver and spleen. Ronot et al. recently utilized a three-dimensional MRE technique (3DMRE), measuring 3 parameters: stiffness, elasticity, and viscosity of the spleen. They concluded that 3DMRE is useful for: a) noninvasively estimating the increase in HVPG, b) detecting high-risk esophageal varices, and c) recognizing advanced portal hypertension.29 Likewise, Shin et al. found a linear correlation with the endoscopic grade of esophageal varices, using the 3DMRE techniques.30 The abovementioned works constitute progress in the knowledge of portal hypertension and even though they do not replace endoscopy or HVPG measurement, they represent an advance that can be useful in the selection of patients that require treatment, and it should continue to be studied.23
SteatosisAnother area of renewed interest in diagnosis through parametric imaging has been the diagnosis of steatosis. This interest has been brought about by the increase in obesity in the general population, which is the most frequent cause of fat infiltrate in the liver. The rise in obesity has been qualified as epidemic and is estimated to currently affect 15 to 30% of the world population. Mexican studies in 2008 put the figure at 14%, stating that the prevalence could reach up to 45% in the Hispanic population residing in the United States.33
A strong increase in the prevalence of NAFL within the next few decades is also estimated, paving the way for a severe public health problem.3
Steatosis diagnosis through imaging methods can be performed with US, CT, and MR. Real time ultrasound is the most widely used, because it is also the most accessible. However, the information provided by this method is qualitative and reported in “grades” that are not measurable. US has little interobserver reproducibility. Its operator-dependent character makes it poorly reliable for follow-up, when the studies are performed by different persons and with different equipment and transducers.
Non-contrasted CT is more precise, since it provides quantitative information in Hounsfield units. Steatosis is usually an incidental diagnosis with this method during a study for a different indication. Ionizing radiation limits its indication for the sole purpose of diagnosis and it is not recommended for steatosis follow-up.
MR spectroscopy is currently the most precise technique for quantifying lipids in the liver. 3T equipment has been reported to have 100% exactness for a cutoff value of fat concentration of 7.48%,33,62 but the complexity of spectroscopy limits its routine use.
In daily practice, the techniques that analyze the phenomenon of chemical shift are used. They are solid, reproducible, and quantitative. The result is reported numerically as a fat infiltration percentage.31,32,58,63,64 The development of a parametric color map makes it easier to observe the quantity and distribution of fat in the parenchyma.38 This technique can even identify fat in the interior of some liver tumors.
MR is also a precise method for NAFL patient follow-up. The quantification of steatosis can be routinely carried out with MR of the liver for another cause or it can be done as a specific multiparametric protocol as the one described above.
It is a known fact that simple steatosis can remain stable and not be modified over time. Nevertheless, 10 to 20% of the patients with simple steatosis progress to NALD and 10 to 15% of those patients can progress to cirrhosis.61 The risk for developing hepatocellular carcinoma is also greater in patients with cirrhosis.63,64
An important question is whether MR can differentiate simple steatosis from NALD in patients with NAFL. Some authors have suggested that NALD can be identified in its initial stages if the fat quantification study is combined with MRE, given that simple steatosis does not alter liver stiffness. In contrast, the development of fibrosis that occurs in NALD does increase stiffness and can be identified by combining parametric studies of fat and MRE.13,16,59,61,65 There have been reports that the inflammation that occurs in the initial stages of NALD can increase stiffness and aid in identifying steatohepatitis, even before the appearance of fibrosis.66
Another indication for steatosis quantification is in the protocol of donor selection for liver transplantation:59 an infiltrate above 20% rules out the donor liver.67
The main limitation for diagnosing steatosis through the chemical shift technique is the coexistence of iron and fat in the liver parenchyma, because the signal is reduced and can falsify the results, hence the importance of quantifying that element in the multiparametric study and utilizing correction protocols: the routine use of the Dixon multi-echo technique is recommended.
Hepatic siderosisThe measurement of iron with the different MR techniques has shown results at least similar to those of liver biopsy, with the advantage for the patient of being noninvasive. Iron quantification with MR has low interstudy variability, making it a reliable study and a valuable tool for evaluating the result of chelation treatments. Patients also prefer it over periodic biopsy in the follow-up.68
Duration of the study, protocols, and cost of magnetic resonance elastographyIn many articles and conferences, MRE is described as an expensive study.
This is true when elastography is added to a complete MR protocol of the liver and non-contrasted and contrasted with gadolinium, which itself is costly.
In contrast, the cost is considerably reduced if the study is specifically directed to only investigate fibrosis, fat, or iron.
The multiparametric protocol used in our department is performed in 25min in a manner similar to that described in the literature by other authors.22,44
Our service has 3 different specific protocols that can be performed in accordance with the clinical necessity of each patient:
- a)
elastography alone
- b)
elastography and lipids
- c)
multiparametric protocol of elastography and lipid and iron quantification
Not only are these protocols applicable for diagnosis, but also for the follow-up of patients during treatment.44 The performance of specific MR studies significantly reduces cost, making them more accessible for both the patient and the institution.
ConclusionsMRE is an efficacious method that provides the gastroenterologist and hepatologist with broader and more precise information for diagnosing liver fibrosis. One of its greatest advantages is the fact that it can carry out MR sequences for the diagnosis and quantification of fibrosis, steatosis, and hepatic siderosis in the same session, making it a multiparametric diagnostic procedure that has excellent concordance with liver biopsy. Today, the precise determination of the grade of fibrosis in patients with hepatitis C or B virus is an indispensable element for selecting and giving priority to patients that may require antiviral treatments. The epidemic increase in metabolic diseases and NAFL also makes MRE an important indication in clinical practice. The possibility of detecting early changes in NALD should be investigated through more studies, but there are already reports that the increase in liver stiffness in patients with NAFL can reveal the presence of early fibrosis and even inflammatory states that precede the development of NALD.
The addition of 2 anatomic sequences in our protocol enriches the information by making it possible to detect morphologic alterations of the liver and the organs of the upper abdomen. The anatomic and functional information provided by this study is useful for the evaluation and follow-up of progression of the CLDs. The biggest innovation of the multiparametric protocol described is obtaining the diagnosis and quantification of fibrosis, steatosis, and hepatic siderosis during the same 25min study, making it clinically and economically viable for the patient and the hospital center.
Financial disclosureNo financial support was received in relation to this study.
Conflict of interestM. Stoopen-Rometti, E.R. Encinas-Escobar, C.R. Ramirez-Carmona, E. Wolpert-Barraza, E. Kimura-Hayama, L.A. Sosa-Lozano, Y. Kimura-Fujikami, J.A. Saavedra-Abril, A.Loaeza-Del Castillo declare that there is no conflict of interest.
R. Favila is a specialist in advanced applications of MR for GE Healthcare.
The authors wish to thank Dr. Rocío Pérez-Johnston and Dr. Véronique Barois-Boullard for their revision of the manuscript.
Please cite this article as: Stoopen-Rometti M, Encinas-Escobar ER, Ramirez-Carmona CR, Wolpert-Barraza E, Kimura-Hayama E, Sosa-Lozano LA, et al. Diagnóstico y cuantificación de fibrosis, esteatosis y hepatosiderosis por medio de resonancia magnética multiparamétrica. Revista de Gastroenterología de México. 2017;82:32–45.