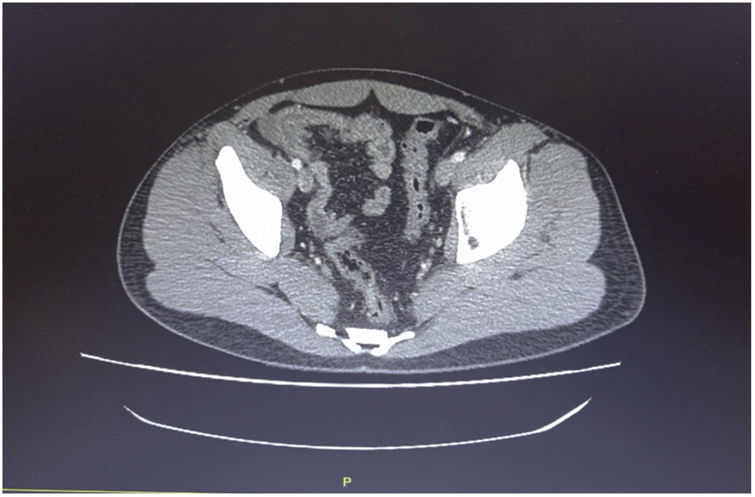
Although the symptoms and imaging features of coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), predominantly involve the respiratory system, manifestations of the disease and its possible complications in extrathoracic organs, such as the gastrointestinal (GI) tract and musculoskeletal system, can be found.1
We present herein a case of terminal ileitis related to reactive ileal lymphoid hyperplasia (RILH) that occurred in a patient with underlying COVID-19. A 29-year-old man, a smoker, with an unremarkable past medical history, was admitted to the hospital during Spain’s Omicron coronavirus wave, presenting with acute abdominal pain in the right lower quadrant (RLQ), of 2-day progression, unaccompanied by diarrhea, respiratory symptoms, or fever. He had received an mRNA COVID-19 vaccine 8 months before admission. A nasopharyngeal swab specimen was positive for RT-PCR SARS-CoV-2, with low cycle threshold values. Respiratory auscultation was normal and blood test results showed increased acute phase reactants. No other laboratory test abnormalities were detected. Chest X-ray was normal. Abdominal computed tomography (CT) scan (Fig. 1) revealed marked circumferential bowel wall thickening of the terminal ileum that was suggestive of Crohn’s disease (CD). Because of the suspected diagnosis of CD and/or an associated bacterial co-infection, the patient was empirically started on a broad-spectrum regimen with ceftriaxone, metronidazole, and methylprednisolone at admission, with good clinical outcome. Ileocolonoscopic exploration of 15 cm of the terminal ileum identified slightly edematous and erythematous areas of mucosa, suggestive of indeterminate ileitis. Biopsy taken at the terminal ileum suggested RILH of the lamina propria. No enzyme immunoassays detected other viral or bacterial agents (norovirus, adenovirus, rotavirus, Salmonella spp., Shigella spp., Yersinia enterocolitica, Campylobacter spp. and Clostridioides difficile antibodies) or autoimmune antibodies (antinuclear antibodies [ANA] or antineutrophil cytoplasmic antibodies [ANCA]). IgM serology was negative for SARS-CoV-2 and IgG serology was positive for the virus. Thus, there were no clear clinical (no diarrhea), endoscopic (indeterminate ileitis), or histologic (normal architecture) features that enabled a diagnosis of CD or other conditions, such as infectious ileitis, microscopic enteritis, or autoimmune vasculitis.
Angiotensin-converting enzyme 2 (ACE2) has been established as the functional and major host receptor for entry of the SARS-CoV-2 spike glycoprotein (S) into cells. The high expression of ACE2 in GI epithelial cells, including the ileum, has correlated with digestive symptoms (anorexia, diarrhea, vomiting, and abdominal pain) and abnormal gut microbiota. In a recent meta-analysis, Mao et al. demonstrated that 10–21% of patients with COVID-19 had GI manifestations.2 Tullie et al. showed 9 cases of isolated ileal involvement detected through abdominal ultrasound or CT scan attributed to COVID-19 in children diagnosed by a positive nasopharyngeal swab test. Neither ileal biopsy nor testing on stool samples was carried out on those subjects. No similar cases have been demonstrated in adults.3
Interestingly, RILH is a rare condition of the GI tract that is predominant at the terminal ileum and rectum, and its etiology is unknown. The disease is common in children but rarely occurs in adults. It is characterized by the growth of submucosal lymphoid tissue and a mucosal response to different types of noxious stimuli, has been described in association with infectious agents, and is hypothesized to be the result of an antigenic response. In the present case, we found no source of infection other than SARS-CoV-2. RILH can be misdiagnosed as acute appendicitis because of the abdominal pain in the RLQ, and its abdominal CT features can mimic CD or other pathologic entities,4 as occurred in our patient. The malignant potential of this condition in adults is still unclear and our case showed no signs of malignancy. The common presentation is abdominal pain, with or without loose stools or gastrointestinal bleeding.
With respect to our search for SARS-CoV-2 infection-associated RILH, several considerations have to be taken into account. First, although no similar cases have been described in adults, it is possible that subtle GI features were not documented or were underestimated. Second, causality between SARS-CoV-2 infection and ileitis related to RILH is often impossible to prove, but we cannot ignore the fact that it may have a role as a direct consequence of viral injury involvement and the onset of an overlapping cell-mediated immune phenomenon of RILH.
In conclusion, SARS-CoV-2 may exclusively occur with intestinal features. Future well-conducted studies will provide superior evidence-based answers.
Ethical disclosuresThe authors declare that no experiments were conducted on humans or animals for the present work, that they have followed the protocols of their work center on the publication of patient data, that they have preserved patient anonymity at all times. Informed consent was not required for the publication of the present case because the article contains no personal data that could identify the patient.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Qanneta R, Feliu-Masgoret M, García-Pardo G et al. Hiperplasia linfoide ileal reactiva relacionada con infección por SARS-CoV-2 como característica clínica singular parecida a la enfermedad de Crohn. Rev Gastroenterol Méx. 2023;88:80–82.