Esophagorespiratory fistulas (ERFs) are rare, and even though the majority have a malignant origin (>80%), benign causes include chronic infections and those secondary to radiotherapy, trauma, or surgery.1 Symptoms are nonspecific and can range from chronic cough to pulmonary infection, sepsis, and potentially to death.2,3 Treatment can be endoscopic or surgical, the latter utilized only when endoscopy fails, given that there is a risk for death of up to 15.4%.4
Endoscopic treatment includes fibrin application (efficacy < 40%),5 endoluminal stent placement (better efficacy in malignant etiology vs. benign etiology [42 vs. 22%]),2 and hemoclip and OVESCO clip (OTSC®, Ovesco Endoscopy AG, Tübingen, Germany) application (25-55% efficacy).6 Other treatments with endoscopic suturing and endoscopic submucosal dissection (ESD), as well as other modified methods with a potential efficacy above 80%,7–10 have been used, with resolution related to size and time.1–10
A 55-year-old woman presented with dysphagia, chest pain, and heartburn of 2-year progression. Evaluation protocol was begun and the diagnosis of “hypercontractile esophagus” was documented. Due to medical treatment failure, the decision was made to carry out peroral endoscopic myotomy. The entrance site was 20 cm from the superior dental arch, the submucosal tunnel continued up to 42 cm, and the myotomy of the inner circular muscle layer of the esophagus was carried out up to 40 cm, sparing the lower esophageal sphincter. The procedure was performed with no incidents. However, 15 days later, the patient sought medical attention due to chest pain, fever, tachycardia, and oral feeding intolerance. A chest tomography scan confirmed esophageal perforation in the upper third of the esophagus and endoscopic examination revealed an orifice with a 10 mm diameter. Local lavage was performed, and a 28 mm × 23 mm × 15 cm fully covered esophageal stent (Wallflex™, Boston Scientific, Marlborough, MA, USA) was placed. It was fixed with 2 hemoclips (Resolution clip™, Boston Scientific, Marlborough, MA, USA) and an OVESCO GC clip (Ovesco Endoscopy AG, Tübingen, Germany). The patient presented with clinical improvement, no longer having signs of systemic inflammatory response, and was discharged. The stent was removed 6 weeks later and contrast medium instillation confirmed an ERF located 22 cm from the superior dental arch in the right bronchial region. An OVESCO T clip (Ovesco Endoscopy AG, Tübingen, Germany) was placed, achieving initial endoscopic closure of the fistula.
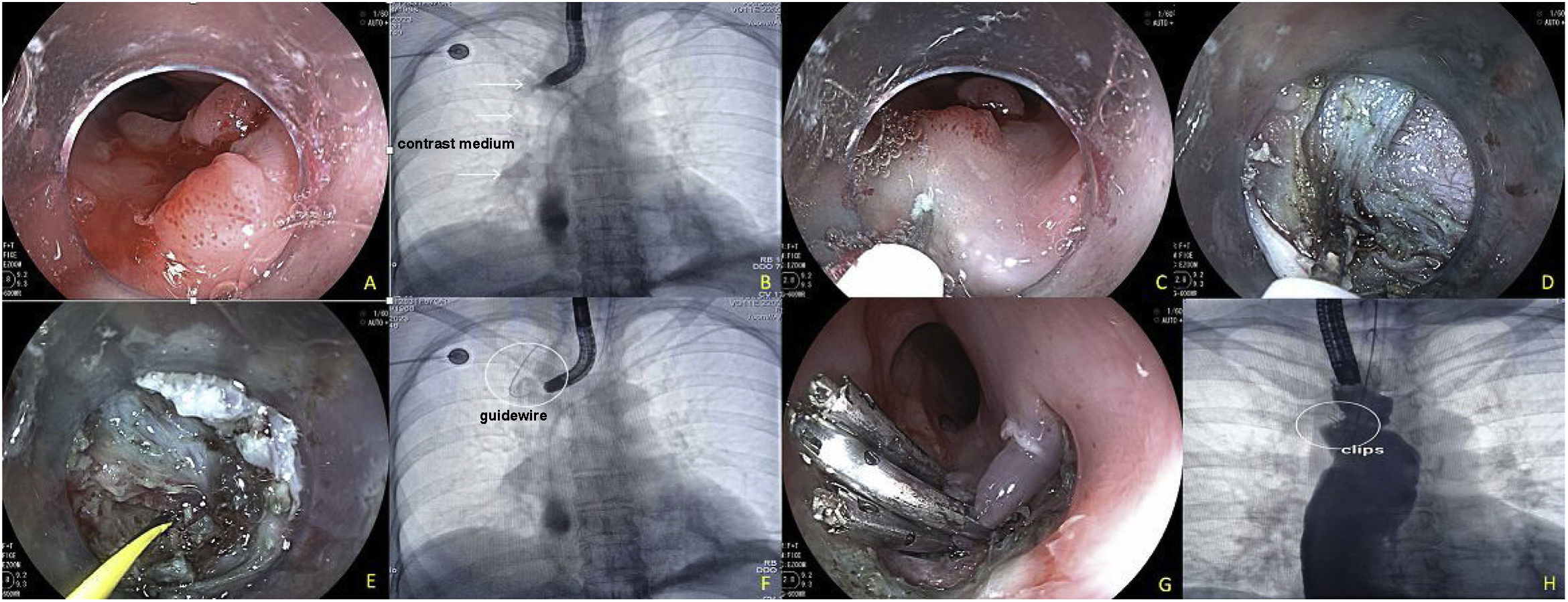
One week after clip placement, the patient presented with hemoptysis on 2 occasions, as well as fever. Endoscopic examination revealed that the OVESCO clip had partially migrated, and so it was removed. Persistence of the fistula with a 2 mm diameter was confirmed. Endoscopic closure combining the ESD technique and hemoclip application was decided upon. To carry it out, a submucosal injection 3 mm proximal to the ERF was applied, enabling the dissection of an approximately 10 mm circumferential area, with an I knife (ERBE, Tübingen, Germany). Once the ERF was denuded and isolated, passage of contrast medium into the right bronchopulmonary region, utilizing a 0.035″ guidewire (Jagwire™, Boston Scientific, Marlborough, MA, USA), was confirmed. Fistulectomy was performed, using Endocut mode I in effect 3, to eliminate the fistulous epithelium, at an approximate depth of 5 mm in that region. Closure was satisfactorily carried out with 3 hemoclips and confirmed through contrast medium instillation (Fig. 1).
Esophagobronchial fistula closure technique with endoscopic submucosal dissection combined with hemoclips. A) Persistence of the fistula after removal of the OVESCO T clip, 22 cm from the superior dental arch. B) Confirmation of the fistula through contrast medium injection. C) Start of the dissection, with submucosal injection 3 mm proximal to the fistula. D) Complete endoscopic fistulectomy, including dissection at the level of the muscularis propria of the esophagus. E) Passage of the guidewire through the fistula. F) Fluoroscopic image showing the guidewire in the bronchopulmonary region. G) Placement of 3 hemoclips for definitive closure. H) Contrast medium instillation confirming complete closure of the fistula and no signs of contrast medium passage into the bronchial region.
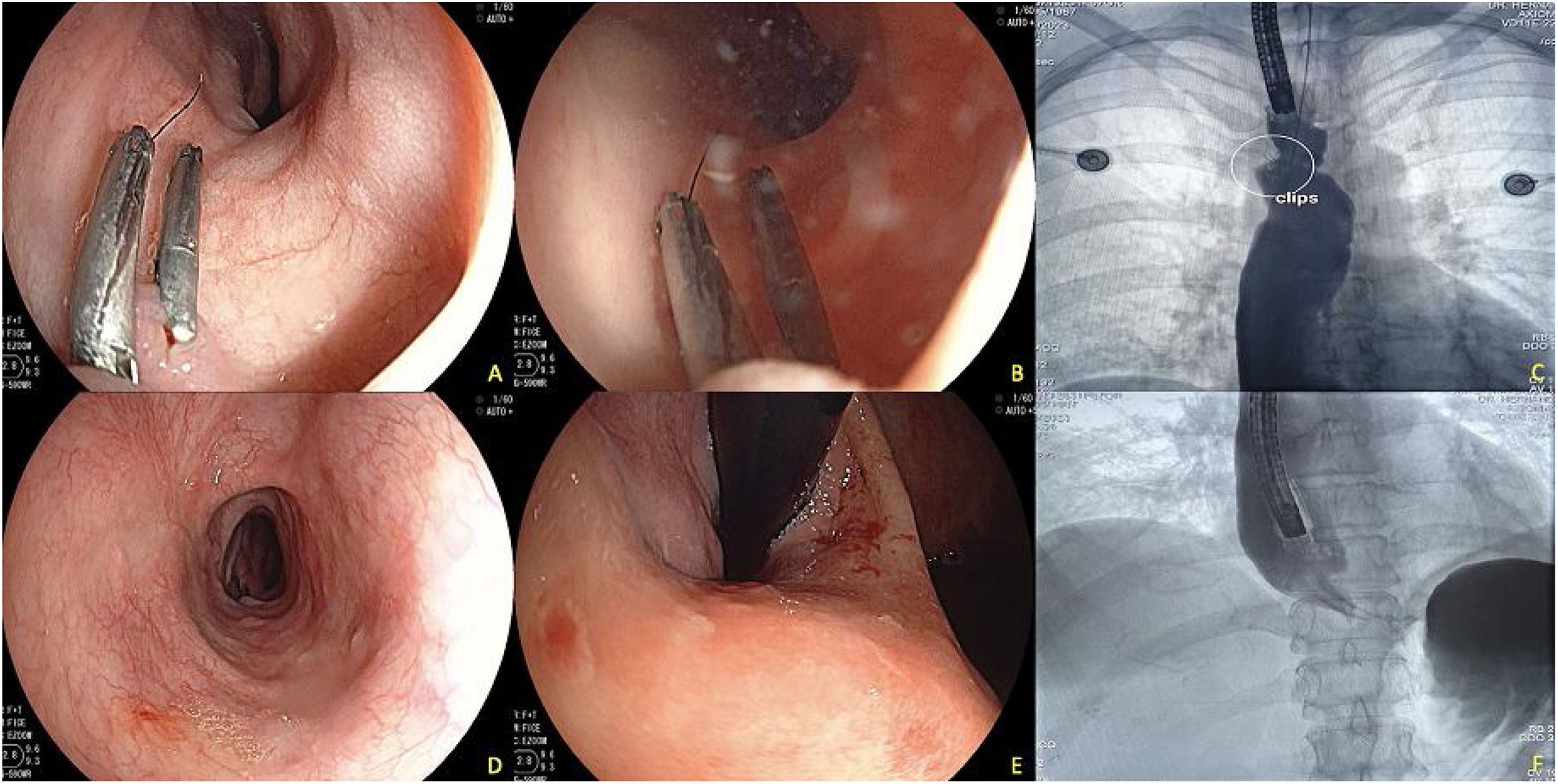
The patient’s clinical progression was satisfactory; she no longer presented with cough or hemoptysis during the ensuing weeks and a new examination 3 months later showed the persistence of the 2 clips in the region, placed prior to the ESD. The absence of esophageal pulmonary fistula was confirmed through contrast medium instillation (Fig. 2).
Endoscopic control at 3 months after fistula closure with dissection and hemoclips. A) Image showing the persistence of 2 hemoclips in the dissected region, revealing completely healed tissue around the region. B) Endoscopic contrast medium instillation. C) Fluoroscopic image showing the continued persistence of the 2 clips and absence of contrast medium passage into the pulmonary region. D) Middle and distal esophagus, with no lesions in the mucosa of that region. E) Changes at the level of the gastroesophageal junction after the peroral endoscopic myotomy, with adequate opening of said junction. F) Contrast medium instillation in the distal esophagus showing adequate passage into the gastric cavity and no signs of leaks.
Unfortunately, there is a poor response (20-54%) to endoscopic treatment of ERFs, whether utilizing single or combined methods,2,4–7,9,10 with a high recurrence risk (55-70%).6–9 Endoscopic suturing has shown good effectiveness in some cases for ERF closure,7 but it is a costly option with poor availability. ESD is an alternative with good results (ERF closure > 80%); it has the advantage of enabling total fistulectomy prior to mechanical closure, or even modifying it by creating a traction line, as described by Bertrand et al.9 In our patient, ESD was performed as a third option, after the failure of other treatments, and with the advantage of enabling a deeper approach to the ERF and the performance of a controlled fistulectomy for good epithelization, prior to mechanical closure. Recurrence is a problem, but the confirmation of closure after 3 months in our patient suggests definitive closure. There are other novel methods that utilize special stents, especially for larger fistulas, but their availability is poor.10 In conclusion, ESD combined with mechanical closure can be a safe and effective alternative for the treatment of ERFs.
Ethical considerationsThe authors declare that the corresponding signed statement of informed consent was requested, that the images presented preserve patient anonymity.
Financial disclosureNo specific grants were received from public sector agencies, the business sector, or non-profit organizations in relation to this study.
Conflict of interestThe authors declare that there is no conflict of interest.