Hutchinson in 1986, Peutz in 1921, Jeghers in 1949, and Bruwer in 1954 were the first to describe Peutz-Jeghers syndrome (PJS)1, which is defined as a congenital autosomal dominant disorder, belonging to the hamartomatous syndromes2. Its incidence is rare, at 1:8300 to 1:12000003, with no predisposition to race or sex. PJS polyps can present at any level of the gastrointestinal tract.
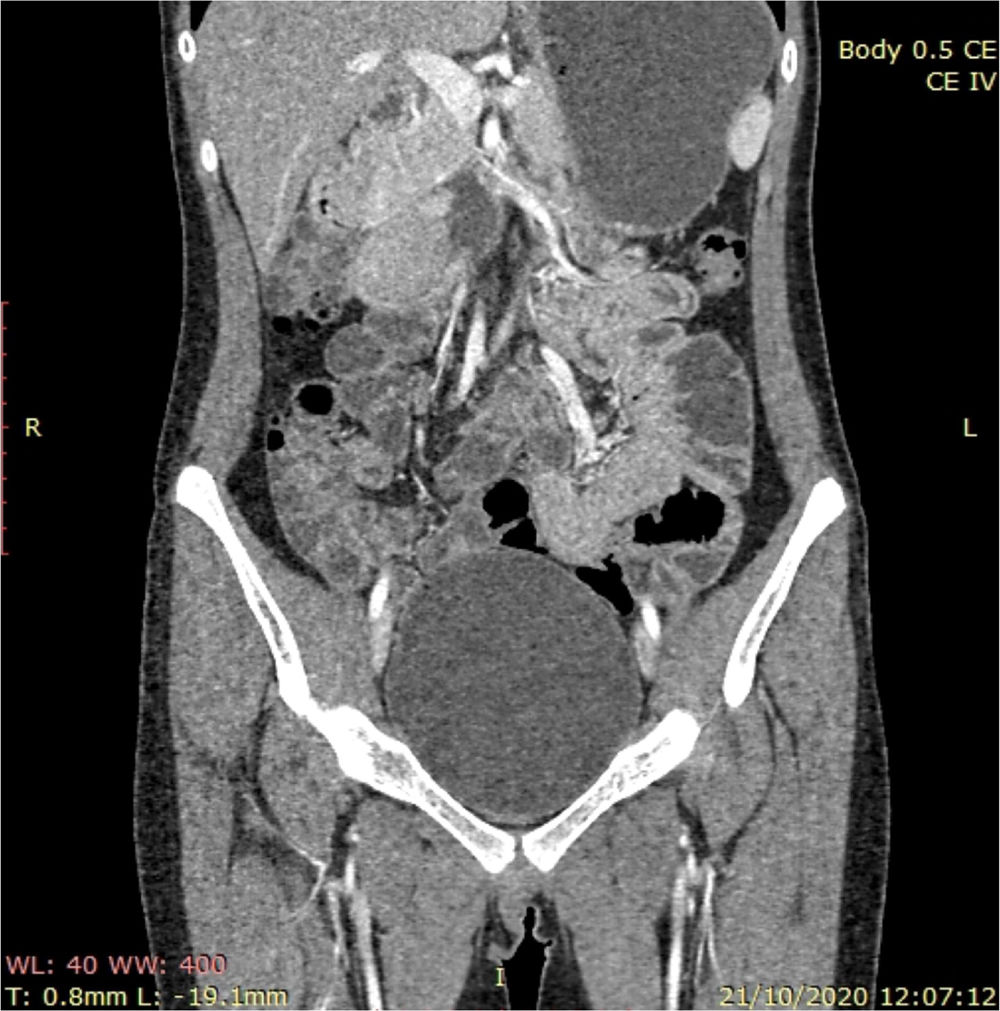
An 18-year-old woman was referred to the gastroenterology department due to abdominal pain, hematemesis, and intestinal intussusception on two occasions that required intestinal resection (80 cm of the jejunum and 50 cm of the ileum). She had unremarkable family, perinatal, and personal medical histories. Up to 3 descendant family lines were studied, with no evidence of tumoral, congenital, or neoplastic diseases. Physical examination revealed hyperpigmented macules on the lips and in the oral cavity. Her referral included numerous endoscopic and histopathologic reports, stating gastroduodenal polyposis (up to 25−40 mm), attenuated colonic polyposis (10–20) up to 40 mm, with suspicion of cancer. She had undergone multiple polypectomies. The histopathologic study reported serrated adenomas and tubulovillous adenoma, with high-grade and low-grade dysplasia in the colon. Imaging studies revealed hyperplastic gastroduodenal polyps, “advanced” tubulovillous adenoma larger than 1 cm, and duodenal hamartomatous adenoma (Fig. 1). Computed tomography (CT) enterography identified dilation of the second and third part of the duodenum, with a contrast-enhanced irregular mass of approximately 52 × 35 mm (Fig. 2). Head CT and a radiographic series for detecting bone metastases showed no lesions.
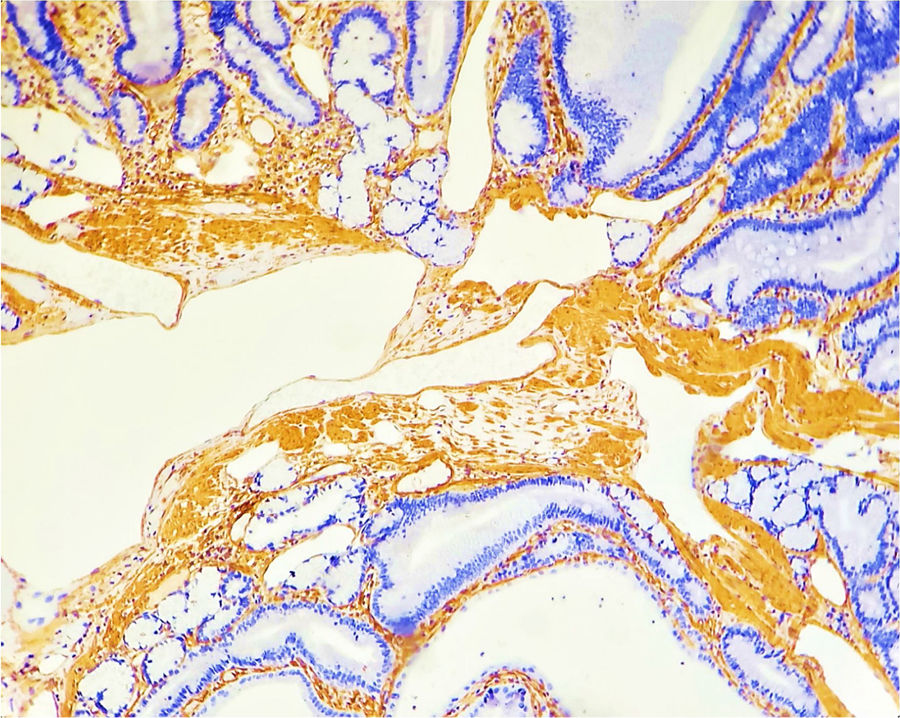
Attenuated familial adenomatous polyposis (FAP) was considered, with surgical implications (total colectomy and duodenal polypectomy vs pancreatoduodenectomy). Nevertheless, the last polypectomy was performed in the colon and the second part of the duodenum. The histopathologic study was conclusive for hamartomas in the duodenum and colon, through smooth muscle actin staining. Thus, any of the adenomatous polyposis syndromes, and even a mixed hamartomatous syndrome, were ruled out, meeting the PJS criteria. Magnetic resonance (MR) enterography and enteroscopy were performed, finding no evidence of polyps in the midgut that would need to be surgically resected.
PJS is a congenital autosomal dominant disorder that belongs to the polyposis syndromes2 and is characterized by the presence of gastrointestinal hamartomas and mucocutaneous hyperpigmentation4. It is a rare disease, with an incidence of 1:8300 and 1:1200000 and a prevalence of 1–1200000, affecting 55% of first-degree relatives, with no predisposition to race or sex3,4.
Mutations in the germline large-segment deletions in the STK11 gene (LKB1) are typical alterations in PJS5. STK11 located on chromosome 19p13.3 has different mutations, 64–80% of which are de novo mutations. STK11 is a tumor suppressor gene that encodes for a serine-threonine kinase, modulating cell growth and proliferation4,5, from AMP-activated protein kinase (AMPK) phosphorylation and the activation of cellular tuberin and the Wnt and GSK3B pathway, resulting in mTOR pathway inhibition5.
Hamartomatous polyps are rare, multilobed polyps with a papillary surface and branching bands of smooth muscle covered with glandular epithelium. There can be hundreds located in the gastrointestinal tract (small bowel 64%, colon 53%, stomach 49%, rectum 32%), gallbladder, bronchial tubes, bladder, and ureters4.
Anatomopathologically, and with immunohistochemical staining for desmin, smooth muscle fibers and epithelium that produce an “arborizing” aspect in 50% of polyps can be seen. In the mucosa and submucosa, the crypts are lobular, with central or peripheral dilations. Cell polarity is preserved6.
Mucocutaneous pigmentation is present in 95% of cases and is seen in the perioral region, the vermilion border of the lips, the nasal cavities, hands, feet, and perineum. Macules fade with age, are grayish blue, and measure 1−5 mm. They arise from melanocytic tumors, with low malignant potential1. Grayish-blue moles can present near the oral cavity4.
Intestinal intussusception presents in young patients with polyps in the small bowel. Its incidence is 47–69%7.
Diagnosis is made with two out of three criteria, including hamartomatous polyps, family history, and mucocutaneous pigmentation3.
The risk of malignant neoplasia in the gastrointestinal tract, pancreas, breast, testes, and uterine cervix is greater3,4, with a mean age of 42 years. The cumulative risk is 81, 66, and 32% for cancer in general, gastrointestinal cancer, and breast cancer, respectively8.
Surveillance is performed with colonoscopy/upper endoscopy in individuals at 8 years of age and repeated every 3 years, if there are significant polyps. If there are no significant polyps or there are symptoms, then surveillance is repeated at 18 years of age. At 50 years of age, surveillance is every 1 or 2 years8. Follow-up with video capsule endoscopy, MR enterography, or barium follow-through are reasonable alternatives2.
Magnetic resonance imaging or ultrasound of the breast should be carried out annually, from the ages of 25−40 years, and thereafter, through mammography. Pap smear should be performed every 2–3 years from 25 years of age3,9, and testicular ultrasound should be carried out, if abnormalities are found during physical examination. Surveillance for other cancers is not recommended2.
Complications are resolved through surgery. Endoscopic polypectomy reduces the risk for complications. Intraoperative enteroscopy should be performed in patients undergoing laparotomy. Drug therapy is not recommended2. Intense pulsed light or laser is used in cases of mucocutaneous pigmentation, if there is a psychologic impact10.
PJS is a rare, little-reported entity, whose surveillance is complicated. Adequate characterization of the polyps is essential, given that the characteristics and aggressiveness of the treatment to be followed (surveillance, colectomy, open polypectomy, pancreatoduodenectomy, etc.) depend on it. In addition, the recurrence of intestinal intussusception makes a comprehensive approach to the patient necessary, even including enteroscopic techniques.
Ethical considerationsThe authors declare that no experiments were conducted on humans or animals for this research and that approval by the institutional ethics committee was not required, given that no interventions were performed.
The authors declare that this article contains no personal information that could identify the patient and informed consent was not requested from her.
Financial disclosureNo specific grants were received from public sector agencies, the business sector, or non-profit organizations in relation to this study.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: García-García VH, López-Colombo A, Gutiérrez-Quiroz CT, Téllez-Cervantes JA. Síndrome de Peutz-Jeghers: toma de decisiones basada en la evidencia, acerca de un caso. Rev Gastroenterol Méx. 2022;87:263–265.