Carcinoid tumors were first described more than 100 years ago, by Lubarsch, who found multiple tumors in the distal ileum of 2 patients, during autopsy.1 They account for 2 to 5% of all gastrointestinal cancers. Histologically, diagnosis is confirmed through positive chromogranin A (CgA) and synaptophysin immunohistochemical staining.2 The Ki-67 labeling index and the mitotic index are correlated with cell proliferation and are useful for classifying neuroendocrine tumors (NETs) (Table 1).3
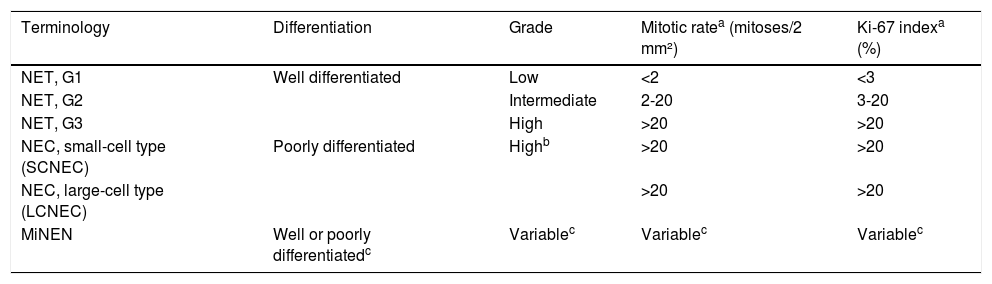
Classification and grading criteria of neuroendocrine tumors of the gastrointestinal tract and the hepatobiliary organs, World Health Organization (WHO), 2019.
| Terminology | Differentiation | Grade | Mitotic ratea (mitoses/2 mm²) | Ki-67 indexa (%) |
|---|---|---|---|---|
| NET, G1 | Well differentiated | Low | <2 | <3 |
| NET, G2 | Intermediate | 2-20 | 3-20 | |
| NET, G3 | High | >20 | >20 | |
| NEC, small-cell type (SCNEC) | Poorly differentiated | Highb | >20 | >20 |
| NEC, large-cell type (LCNEC) | >20 | >20 | ||
| MiNEN | Well or poorly differentiatedc | Variablec | Variablec | Variablec |
LCNEC: large-cell neuroendocrine carcinoma; MiNEN: mixed neuroendocrine–non-neuroendocrine neoplasm; NEC: neuroendocrine carcinoma; NET: neuroendocrine tumor; SCNEC: small-cell neuroendocrine carcinoma.
Mitotic rates are to be expressed as the number of mitoses/2 mm² (the equivalent of 10 high power fields with ×40 magnification and a field of view of 0.5 mm), as determined by counting in 50 fields of 0.2 mm² (i.e., in a total area of 10 mm²); the Ki-67 proliferation index value is determined by counting at least 500 cells in the regions of highest labelling (hot-spots), which are identified at scanning magnification; the final grade is based on whichever of the two proliferation indexes places the neoplasm in the higher grade category.
In most MiNENs, both the neuroendocrine and non-neuroendocrine components are poorly differentiated, and the neuroendocrine component has proliferation indices in the same range as other NECs, but this conceptual category allows for the possibility that one or both components may be well differentiated; when feasible, each component should therefore be graded separately.
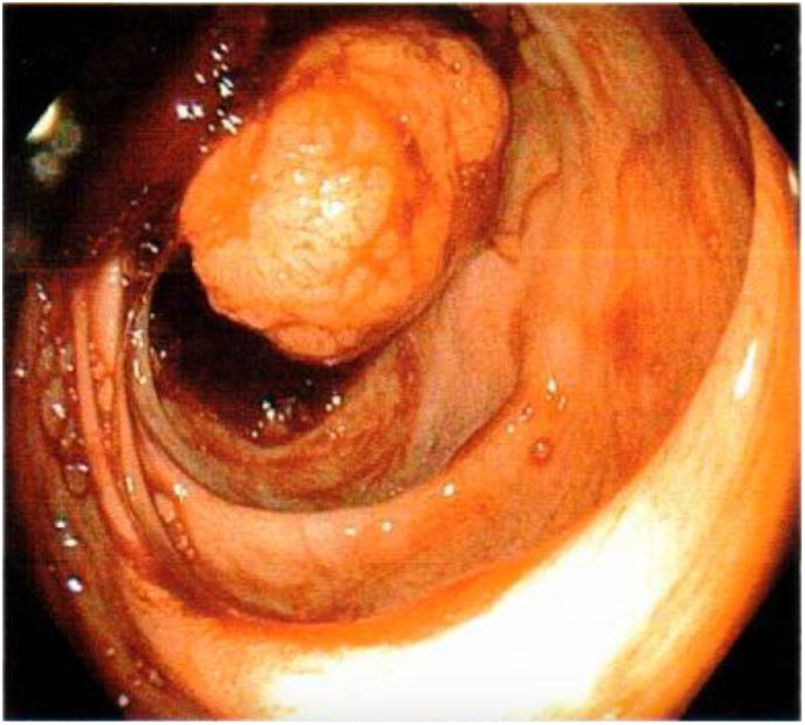
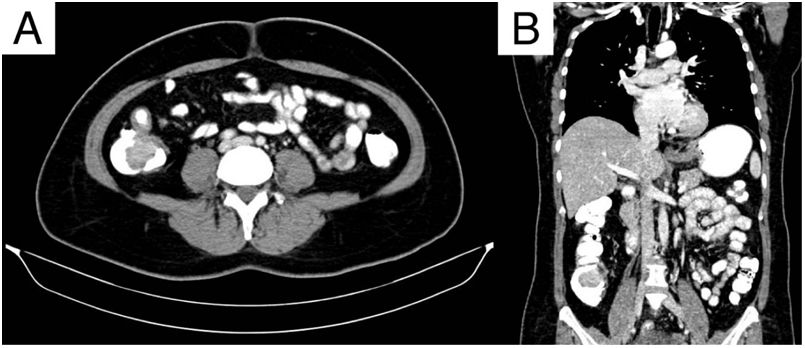
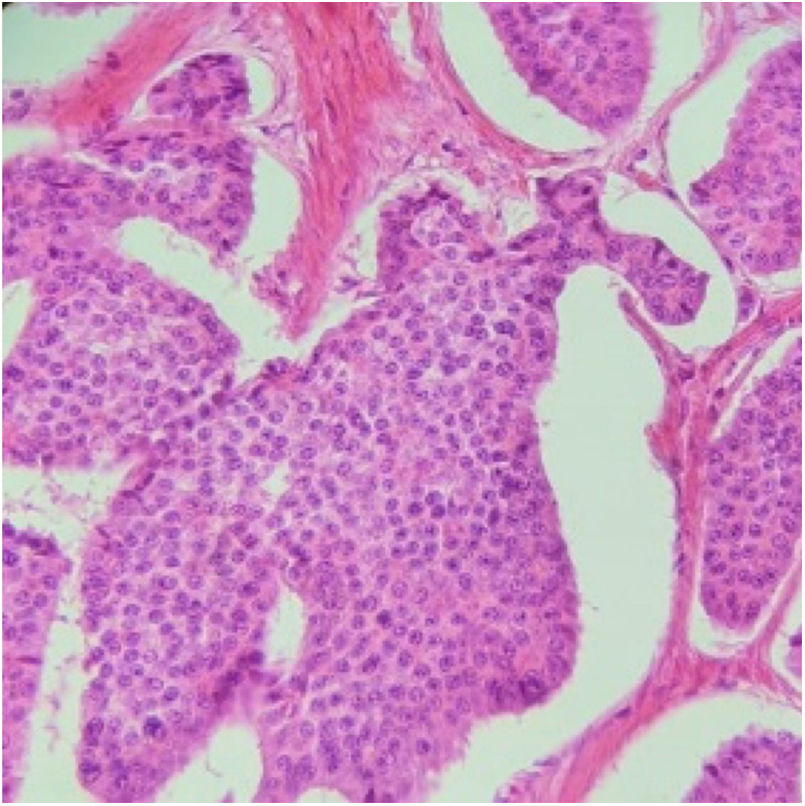
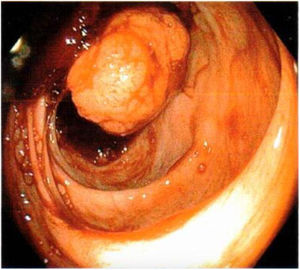
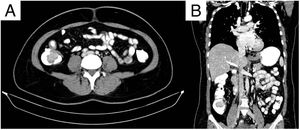
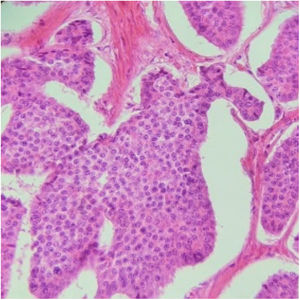
A 53-year-old woman had a past surgical history of a right oophorectomy secondary to a nonspecified benign tumor, 30 years prior, and hysterectomy secondary to endometrial polyps, 10 years earlier. Her current illness began with 4 episodes of hematochezia, over the past 10 months, change in stool consistency, asthenia, and adynamia. She received numerous blood transfusions due to acute bleeding. Upon admission to the emergency department, the patient presented with symptoms of anemia. Laboratory work-up results reported normocytic hypochromic anemia of 6.4 g/dl, lymphopenia of 0.65, PTT of 18.1 s, glucose of 117.9 mg/dl, and total serum proteins of 6.5 g/dl. During her hospital stay, she was transfused with 3 units of packed red blood cells and then underwent diagnostic colonoscopy. A sessile polyp was found in the right colon, measuring approximately 4 cm in diameter, and occluding 30% of the intestinal lumen (Fig. 1). Biopsy results reported a polyp with a morphologic pattern consistent with a carcinoid tumor. Serum carcinoembryonic antigen level was 0.79 ng/mL. A double-contrast computed tomography (CT) scan revealed a filling defect at the level of the cecum, from a 31 × 40 × 30 mm, hypodense image that was enhanced upon contrast medium application (Fig. 2). A right oncologic laparoscopic hemicolectomy, with mechanical ileotransverse anastomosis, was performed, and a cecal tumor was found, measuring approximately 3 × 4 × 3 cm, with wall adhesions and mesenteric lymph nodes. The patient’s postoperative progression was favorable, requiring no transfusion, and she resumed oral diet on day 3. The pathology study of the specimen, corresponding to the ascending colon, terminal ileum, and cecum, reported a 3 × 4 × 3 cm, well differentiated NET, located at the ileocecal valve, with invasion through the muscle and into the subserous tissue; lymph nodes with follicular lymphoid hyperplasia (Ki-67: 2%); and unaltered cecal appendix, stage pT3, pN0, pMX (Fig. 3). The patient was released on postoperative day 5, continuing the oncology protocol. A positron emission tomography (PET) scan identified signs of metastatic disease at the level of the liver, in the bone system (T3 vertebral body), and in the proximal third of the humerus. The patient is currently receiving treatment with octreotide and strict surveillance.
Axial (A) and coronal (B) views of double-contrast computed tomography, showing an exophytic hypodense lesion in the cecum, dependent on the medial wall, measuring 31 × 40 × 30 mm, with poor enhancement, after contrast medium administration. The lesion can be seen infiltrating the ileocecal valve and the base of the cecal appendix.
Histopathologic slice of a well differentiated neuroendocrine tumor. A proliferation of small cells with monotonous round nuclei can be seen, with salt-and-pepper chromatin and eosinophilic cytoplasm, with signs of mitosis that are forming solid nests, with peripheral palisading. Immunohistochemistry with immunophenotyping: synaptophysin-positive, chromogranin-positive, CD56-positive, CDX-2-positive, and CKAE1/AE3-positive, with a 2% proliferation index.
Neuroendocrine neoplasms (NENs) arise from the neuroendocrine cell system. The term NEN encompasses well differentiated NETs and poorly differentiated neuroendocrine carcinomas.4 The term “carcinoid” is used in the literature, in reference to well differentiated NETs, but it is currently not included in their classification. They are found more frequently in the gastrointestinal tract (54.5%) and the lungs (30.1%).5 The incidence of NETs is 5.25 for every 100,000 inhabitants.6 The most frequent locations in the gastrointestinal tract are the small bowel (44.7%) and the rectum (19.6%), and they account for only 10.6% in the colon.5
The clinical manifestations of carcinoid tumors depend on the location, and the presence of metastasis or carcinoid syndrome. The most common symptoms are diarrhea, abdominal pain, gastrointestinal bleeding, or weight loss. Late manifestation, as large tumors, is common and metastasis is often present at the time of diagnosis. They are generally incidental findings during routine endoscopy performed for a different reason or secondary to rectal bleeding.7
Imaging studies are essential for the diagnosis, prognosis, and treatment of NETs. Diagnostic methods include CT, magnetic resonance imaging (MRI), ultrasound, scintigraphy, and PET. CT is the predominant imaging modality for NETs. Gastrointestinal endoscopy is the procedure of choice for diagnosing gastric, duodenal, and colorrectal NETs.8
Colonic NETs arise from Kulchitsky cells or enterochromaffin cells located inside the colonic crypts of Lieberkühn. Patients generally present with abdominal pain due to the effect of the mass or desmoplastic reaction induced by the tumor, gastrointestinal bleeding, and weight loss. At diagnosis, the mean size of colonic NETs is 5 cm and the majority present with distant or local metastases.2 They have the worst 5-year survival rate of all the gastrointestinal NETs, at 40 to 70%, depending on the site and stage, compared with other sites of the gastrointestinal tract. NETs located in the rectum have a 5-year survival rate of 74 to 84%. Survival for localized, regional, and distant disease is 261, 36, and 5 months, respectively. Right-sided colonic NETs are more frequent (70%) and have often metastasized at the time of diagnosis.2,9
The primary treatment goal is to increase survival, control symptoms, and improve quality of life, which is possible through wide surgical resection. Said procedure is recommended in cases of locoregional and advanced disease, and in cases with imminent obstruction.10 Surgical extension depends on the size and extension of the tumor in the gastrointestinal tract. Due to late detection and the poor prognosis associated with colonic NETs, the following is recommended: endoscopic resection in tumors < 1 cm; transanal excision in tumors between 1-2 cm or after endoscopic resection with positive margins; and partial colectomy with regional lymph node dissection in tumors > 2 cm and in invasive tumors.11 The low therapeutic response of chemotherapy (< 30%) is not very encouraging.12 The most common sites of metastasis, in order of frequency, are the liver, bone, and lungs.13
The prognosis and follow-up of patients depend on their clinical status at diagnosis and the therapy they have received. Triple-phase CT of the abdomen and pelvis or MRI is recommended every year for the first 3 years, and then every 1 to 2 years, for at least 10 years. Markers in blood or urine (including CgA and 5-HIAA) are not recommended for follow-up, given that there is no evidence showing the value of their routine testing.14
NETS are rare entities in the lower gastrointestinal tract, compared with the rest of the cell lines of colon cancer, specifically in the ileocecal valve. The term carcinoid is currently inadequate and the incidence of those tumors, in Mexico, is not precisely known. There should be a multidisciplinary approach, with respect to surgical treatment and postoperative follow-up, given that the advent of diagnostic tools, such as PET and minimally invasive surgery of the colon, enable directed treatment.
Financial disclosureNo financial support was received in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Ethical considerationsThe authors declare that no experiments were conducted on humans or animals for the present study, that they have followed the protocols of their work center on the publication of patient data, and that they have preserved patient anonymity at all times. A written statement of informed consent was obtained and all personal data that could identify the patient was omitted.
Please cite this article as: Maldonado-García EL, Álvarez-Barragán AF, Acevedo-Aguirre CE, Galicia-Alemán B, Arellano-Gutiérrez G. Tumor neuroendocrino de válvula ileocecal: reporte de un caso y revisión de la literatura. Rev Gastroenterol Méx. 2022;87:261–263.