Ulcerative colitis is a chronic condition characterized by inflammation affecting the colon. To objectively and integrally measure disease activity in patients with ulcerative colitis and thus optimize pharmacologic treatment, a novel integral disease index was created that includes the clinical, biochemical, endoscopic, and histologic characteristics necessary for achieving that task. The aim of the present study was to validate the novel integral disease index in patients with ulcerative colitis.
Materials and methodsA cohort study on a total of 222 patients with histologic confirmations of ulcerative colitis diagnosis was conducted. The variables included in the disease index were: number of bowel movements per day; values for hemoglobin, high-sensitivity C-reactive protein, and serum albumin; and endoscopic and histologic findings measured through the subscales of the Mayo and Riley scores, respectively. The data analysis was performed utilizing the STATA SE 11.1 statistics program.
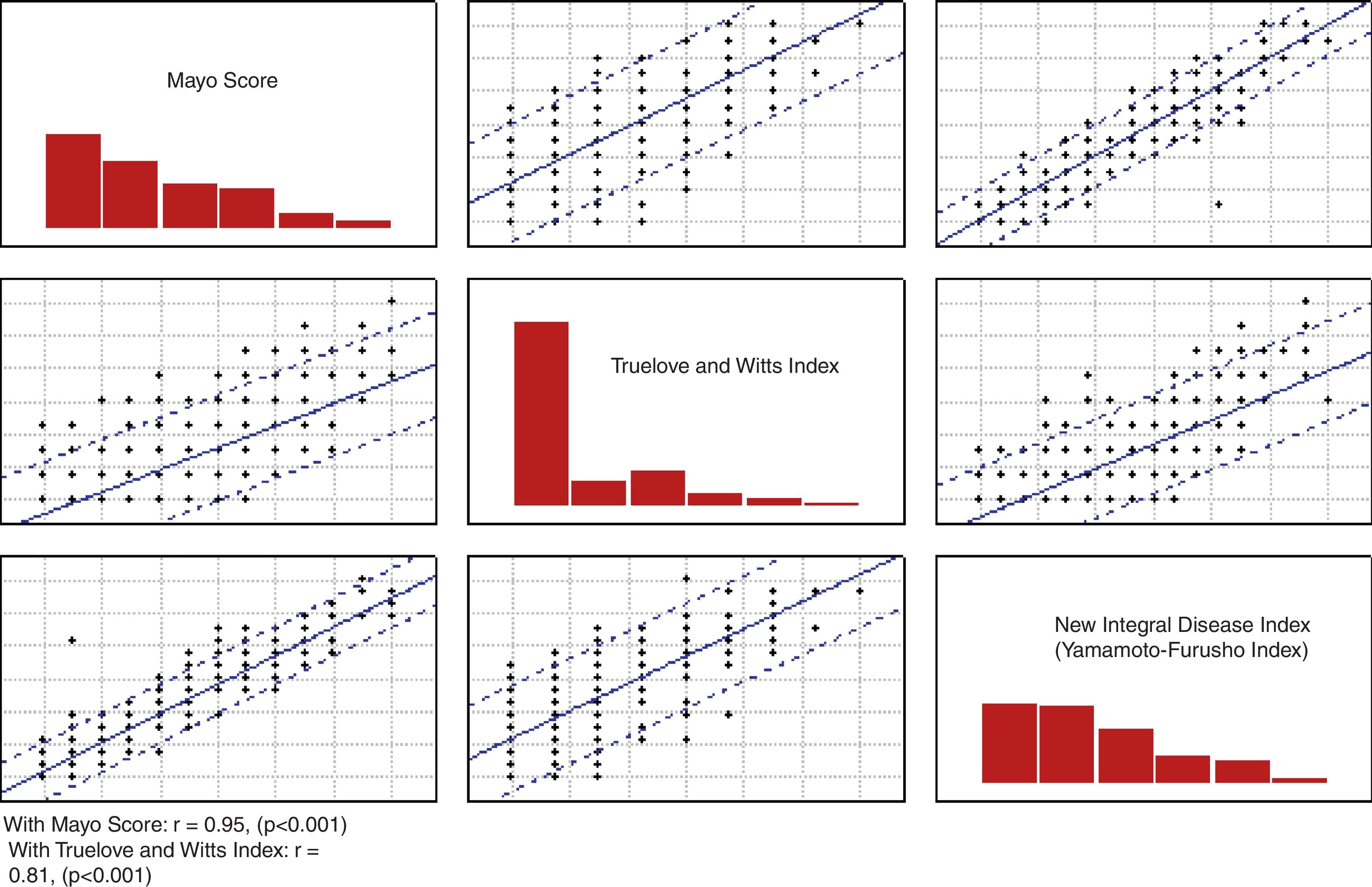
ResultsThe correlation of the novel disease index was very good (r=0.817, p <.001 with the Truelove and Witts criteria and r=0.957, p <.0001 with the Mayo score, respectively). Good internal consistency was found with a Cronbach's alpha coefficient of 0.78 and an acceptable mean inter-item correlation (r=0.47, p <.05). The total efficacy of the novel index was 87.2% correctly classified patients, with an AUC according to the three scenarios described of 0.93, 0.92, and 0.96, respectively.
ConclusionsThe novel integral disease index (Yamamoto-Furusho Index) provides an integral view of disease activity in patients with ulcerative colitis and is useful for optimizing pharmacologic treatment.
La colitis ulcerosa crónica idiopática (CUCI) es una condición crónica caracterizada por una inflamación que afecta al colon. Para medir objetiva e integralmente la actividad de la enfermedad en pacientes con CUCI y en consecuencia optimizar el tratamiento farmacológico, se creó un nuevo índice integral de enfermedad que incluye las características clínicas, bioquímicas, endoscópicas e histológicas necesarias para lograr dicho fin. El objetivo del presente estudio fue validar el nuevo índice integral de enfermedad en pacientes con CUCI.
Materiales y métodosSe realizó un estudio de cohorte con un total de 222 pacientes con confirmaciones histológicas de CUCI. Las variables incluidas en el índice de enfermedad fueron: número de evacuaciones por día; valores de hemoglobina, proteína C-reactiva de alta sensibilidad y albúmina en suero; hallazgos endoscópicos e histológicos medidos por medio de las puntuaciones en las subescalas de puntuación de Mayo y Riley respectivamente. El análisis de datos fue realizado con el programa de estadística STATA SE 11.1.
ResultadosLa correlación del nuevo índice de enfermedad fue muy bueno (r=0.817, p<0.001 con los criterios de Truelove y Witts y r=0.957, p <0.001 con puntuación de Mayo). Se encontró buena consistencia interna con un coeficiente de alfa de Cronbach de 0.78 y una media de correlación interelemento aceptable (r=0.47, p <0.05). La eficacia total del nuevo índice fue de 87.2% pacientes correctamente clasificados, con un área bajo la curva de 0.93, 0.92 y 0.96 respectivamente, de acuerdo a los tres escenarios descritos.
ConclusionesEl nuevo índice integral en enfermedad o índice Yamamoto-Furusho proporciona una visión integral de la actividad de la enfermedad en pacientes con CUCI y es útil para optimizar el tratamiento farmacológico.
Inflammatory bowel disease (IBD) is a group of diseases of the gastrointestinal tract that includes Crohn's disease and ulcerative colitis (UC).1 The etiology of IBD remains unknown, but patients with IBD may have an aberrant immune response that occurs in genetically susceptible individuals as the result of a complex interaction of environmental, microbial, and immunologic factors.2
UC is a chronic inflammatory condition that causes continuous mucosal inflammation of the colon, usually with no granulomas on biopsy. It affects the rectum, and to a varying extent, the colon, in a continuous fashion and is characterized by a relapsing and remitting course, with heterogeneous clinical, biochemical, and histologic features. A severe disease course is related to poor outcomes, such as inferior quality of life, disability, and mortality, and it could also be related to higher healthcare costs.3
Therapeutic advances in medical management have modified treatment targets for IBD.3 Consensus recommendations for clinical practice and trial endpoint support strive not only for symptom resolution, but also for endoscopic mucosal healing. However, those results do not necessarily reflect quiescent microscopic disease. The presence of persistent microscopic inflammation in patients with UC has been associated with an increased rate of relapse, hospitalization, colectomy, and risk for the development of dysplasia and colorectal cancer.2,4
It is important for the physician to be fully aware of disease activity because it influences therapeutic decisions (whether treatment should be oral, topical, or surgical), and provides valuable information on the risk of complications and patient prognosis.5 Clinical disease activity is a complex concept that relies on clinometric assessment tools that can be converted into a score to be objectively used to predict individual patient risk through variables obtained at a routine medical visit.6 However, the concordance of clinical evaluation utilizing the current index scores with the findings from objective diagnostic tools, such as serologic biomarkers, colonoscopy, and histology, can be poor.7 The addition of those parameters has been shown to improve risk stratification and treatment response. Fifty years ago, Sidney Truelove described an index that included clinical and biochemical endpoints in trials on UC,8 and it became one of the most widely used indexes in clinical practice for measuring disease activity. However, it does not integrally include important items for objectively describing disease activity, such as endoscopic and histologic parameters, and it did not go through a correct validation process.
An objective assessment of disease activity is an important clinical trial prerequisite for measuring the efficacy of novel drugs or medical treatment strategies.9 As new objectives are being developed for the global management of UC, leading to new treatment strategies aiming to enhance disease control, the availability of objective and user-friendly tools for evaluating disease activity and helping the clinician make treatment decisions is essential. Disease activity in patients with UC should be evaluated in an integral manner with clinical, biochemical, histologic, and endoscopic parameters.7,10 However, experts agree that an optimal index that includes histologic activity, along with the other parameters, still needs to be created. A better classification strategy could improve clinical decisions and therapeutic approaches.11
The aim of the present study was to validate the Novel Integral Disease Index (NIDI), or Yamamoto-Furusho Index, for measuring UC activity through clinical, biochemical, endoscopic, and histopathologic findings and to evaluate the overall diagnostic performance of the NIDI in Mexican patients presenting with UC.
Materials and methodsA prospective cohort study was conducted that included 222 patients with the definitive diagnosis of UC confirmed through histopathology. A total of 546 evaluations were analyzed at baseline and at a minimum of one follow-up visit for each patient. All patients were seen at the Inflammatory Bowel Disease Clinic of the Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán and were evaluated within the time frame of January 2014 to July 2016. Diagnosis of UC was based on medical history (chronic diarrhea with mucus and bloody stools, weight loss, and abdominal pain), endoscopic findings (ulcers, mucosa friability, loss of vascular pattern, and extension), histologic findings (lymphoplasmacytic infiltrates at the lamina propria, cryptitis, crypt abscess, destruction and ulceration of the glandular pattern) and the biochemical markers of erythrocyte sedimentation rate (ESR), hemoglobin, high-sensitivity C-reactive protein (hs-CRP), serum albumin, and leukocyte and platelet counts. The exclusion criteria were: diagnosis of indeterminate colitis, Crohn's disease, UC patients with proctocolectomy, concomitant infections, and patients with incomplete data in the clinical records from a minimum of two visits to the outpatient clinic.
Measurement of clinical activityEach patient was evaluated at least two times, using the NIDI or Yamamoto-Furusho index of disease activity, the Mayo score,12 and the Truelove and Witts index.8 The evaluation included clinical, biochemical, colonoscopic, and histopathologic findings. All parameters were measured within the span of one week to evaluate disease activity. The Mayo score12 includes four components: 1) number of bowel movements above the normal frequency; 2) presence of rectal bleeding; 3) global medical assessment, and 4) endoscopic appearance. The Truelove and Witts severity index includes several parameters: 1) number of stools per day; 2) quantity of rectal bleeding; 3) heart rate; 4) body temperature; 5) anemia severity, and 6) ESR level.
Biochemical parametersBiochemical markers were determined within 7 days of the colonoscopy and histopathologic evaluation to evaluate the presence of UC activity. The biomarkers used were: hemoglobin (Hb), albumin, hs-CRP, ESR, and platelet and white blood cell counts. The results were evaluated during the follow-up of at least two visits to the outpatient IBD Clinic.
Endoscopic findingsAll colonoscopies were performed within 7 days prior to the evaluation after the participants signed written statements of informed consent in accordance with the Declaration of Helsinki. All patients received standard bowel preparation with polyethylene glycol (washing solution) prior to colonoscopy. The endoscopic findings were based on the Mayo endoscopic subscore.13
Histologic UC evaluationAt least two colonic biopsies were taken from 6 segments that included the cecum, ascending colon, transverse colon, descending colon, sigmoid colon, and rectum. Each biopsy was evaluated according to the Riley index,14,15 which is composed of 6 variables: 1) acute inflammatory infiltrate (polymorphonuclear cells at the lamina propria); 2) chronic inflammatory infiltrate (lymphocytic infiltrate at the lamina propria); 3) cryptitis (abscesses); 4) loss of epithelial integrity; 5) mucin depletion, and 6) irregularities in the architecture of the crypts. Each variable is graded on a 4-point scale (no activity, mild, moderate, and severe) and the result is the average of two assessments made by independent observers.
The novel integral disease index of ulcerative colitis activity (NIDI), or Yamamoto-Furusho index
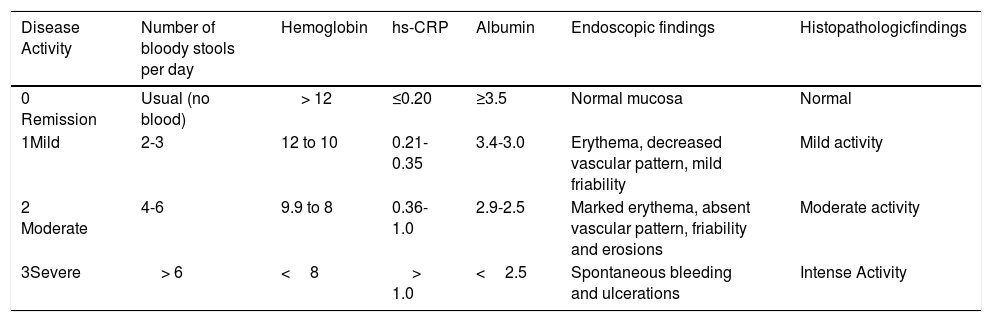
The NIDI, or Yamamoto-Furusho Index, takes six categories into consideration: 1) number of bloody stools per day; 2) hemoglobin; 3) high sensitivity C-reactive protein; 4) albumin; 5) endoscopic findings and 6) histologic findings. Each variable is graded on a score from 0 to 3 as shown in Table 1. The range of the NIDI (Yamamoto-Furusho Index) is from 0 to 18 points and is distributed into four categories: 1) remission (0 to 3 points); 2) mild activity (4 to 6 points); 3) moderate activity (7 to 12 points); and 4) severe activity (13 to 18 points).
Novel integral disease index of UC activity (NIDI) or Yamamoto-Furusho Index.
| Disease Activity | Number of bloody stools per day | Hemoglobin | hs-CRP | Albumin | Endoscopic findings | Histopathologicfindings |
|---|---|---|---|---|---|---|
| 0 Remission | Usual (no blood) | > 12 | ≤0.20 | ≥3.5 | Normal mucosa | Normal |
| 1Mild | 2-3 | 12 to 10 | 0.21- 0.35 | 3.4-3.0 | Erythema, decreased vascular pattern, mild friability | Mild activity |
| 2 Moderate | 4-6 | 9.9 to 8 | 0.36-1.0 | 2.9-2.5 | Marked erythema, absent vascular pattern, friability and erosions | Moderate activity |
| 3Severe | > 6 | <8 | > 1.0 | <2.5 | Spontaneous bleeding and ulcerations | Intense Activity |
A descriptive analysis of the demographic and clinical variables was performed. Correlations were made between the NIDI (Yamamoto-Furusho Index) and both the Truelove and Witts index and the full Mayo score with the Pearson coefficient. The validation and reliability analyses were carried out using the principal component analysis and the Cronbach's alpha coefficient for internal consistency and the average correlation of the individual items. Finally, we tested and compared all activity index scores with a receiver-operating characteristic (ROC)-based analysis to define their sensitivity, specificity, positive predictive value (PPV), negative predictive value (NVP), positive likelihood ratio (LR+), negative likelihood ratio (LR –), and area under the ROC curve (AUC), with 95% confidence intervals. We evaluated the performance of the NIDI (Yamamoto-Furusho Index) in several clinical scenarios and the disease spectrum was focused on severe disease according to histopathologic findings, which we used as a gold standard.
We classified our findings according to three possible scenarios of severe activity defined as follows: 1) severe activity defined only by colonoscopy findings, 2) severe activity defined only by histopathologic findings, and 3) severe activity defined only when both conditions were present. The performance of individual items was also tested. NIDI responsiveness was evaluated through its sensitivity to change in the 3 evaluations made for each patient, utilizing the Student's t test for paired samples and the test-retest analysis to evaluate the interclass correlation coefficient.
We considered a two-tailed p value level of <0.05 for all the hypothesis tests. The statistical analysis was performed using STATA SE 11.1 software.
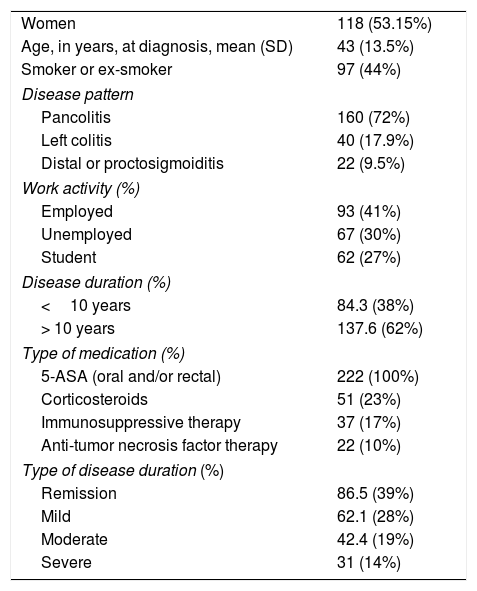
ResultsDemographics and baseline characteristicsWe included the data collected from 546 initial and follow-up evaluations belonging to 222 Mexican patients with the definitive diagnosis of UC. Clinical and demographic characteristics are summarized in Table 2.
Summary of demographics and baseline characteristics (n=222).
| Women | 118 (53.15%) |
| Age, in years, at diagnosis, mean (SD) | 43 (13.5%) |
| Smoker or ex-smoker | 97 (44%) |
| Disease pattern | |
| Pancolitis | 160 (72%) |
| Left colitis | 40 (17.9%) |
| Distal or proctosigmoiditis | 22 (9.5%) |
| Work activity (%) | |
| Employed | 93 (41%) |
| Unemployed | 67 (30%) |
| Student | 62 (27%) |
| Disease duration (%) | |
| <10 years | 84.3 (38%) |
| > 10 years | 137.6 (62%) |
| Type of medication (%) | |
| 5-ASA (oral and/or rectal) | 222 (100%) |
| Corticosteroids | 51 (23%) |
| Immunosuppressive therapy | 37 (17%) |
| Anti-tumor necrosis factor therapy | 22 (10%) |
| Type of disease duration (%) | |
| Remission | 86.5 (39%) |
| Mild | 62.1 (28%) |
| Moderate | 42.4 (19%) |
| Severe | 31 (14%) |
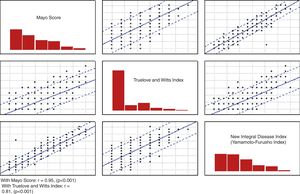
The correlation of the NIDI (Yamamoto-Furusho Index) was evaluated with the Truelove and Witts index and the full Mayo score (r=0.817, p <0.001 and r=0.957, p <0.001, respectively) as shown in Figure 1.
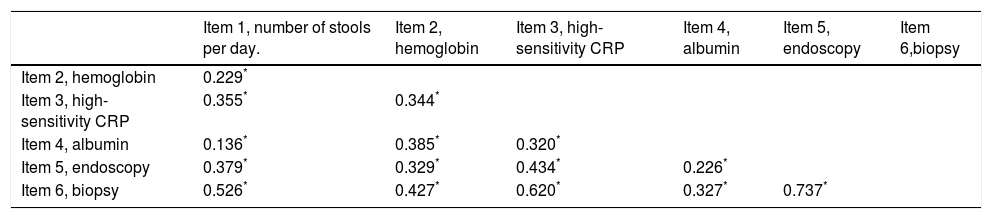
Validation analysis of the items and development of the NIDIThe main component analysis of the six items included in the NIDI (Yamamoto-Furusho Index) revealed a probable unidimensional scale, with 49.9% of the variance explained by a unique component. An adequate internal consistency was observed with a Cronbach's alpha of 0.78 and an acceptable average inter-item correlation (r=0.47, p <0.05). The correlation matrix is shown in Table 3. We decided to keep all items included in our index, since the minimal correction for the Cronbach's alpha was 0.02, when none of the items were removed.
Inter-item correlation matrix.
| Item 1, number of stools per day. | Item 2, hemoglobin | Item 3, high-sensitivity CRP | Item 4, albumin | Item 5, endoscopy | Item 6,biopsy | |
|---|---|---|---|---|---|---|
| Item 2, hemoglobin | 0.229* | |||||
| Item 3, high-sensitivity CRP | 0.355* | 0.344* | ||||
| Item 4, albumin | 0.136* | 0.385* | 0.320* | |||
| Item 5, endoscopy | 0.379* | 0.329* | 0.434* | 0.226* | ||
| Item 6, biopsy | 0.526* | 0.427* | 0.620* | 0.327* | 0.737* |
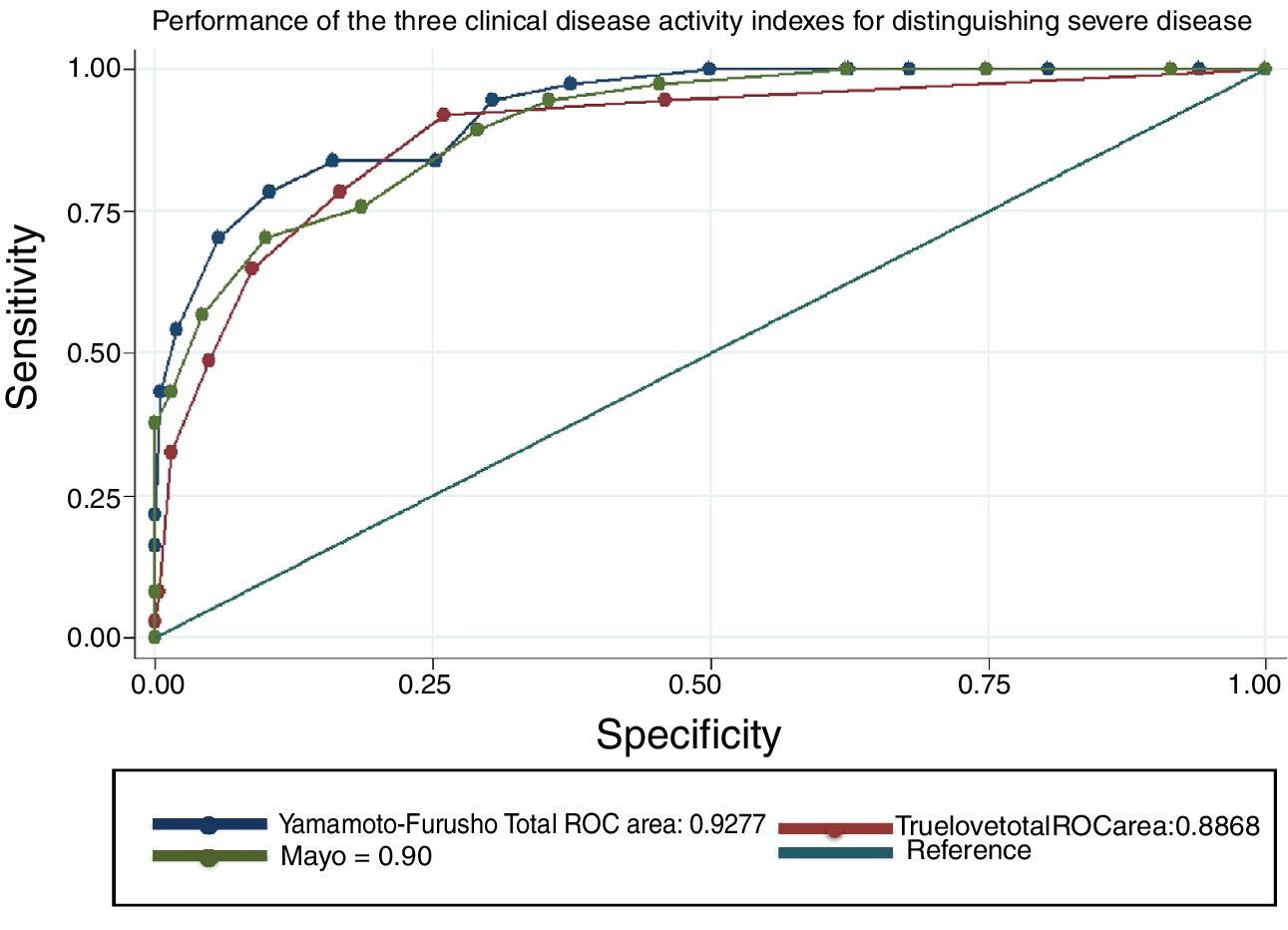
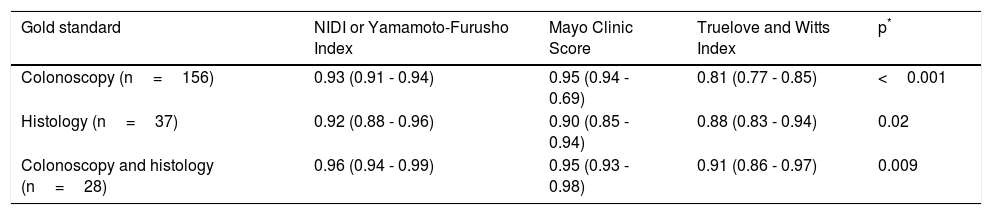
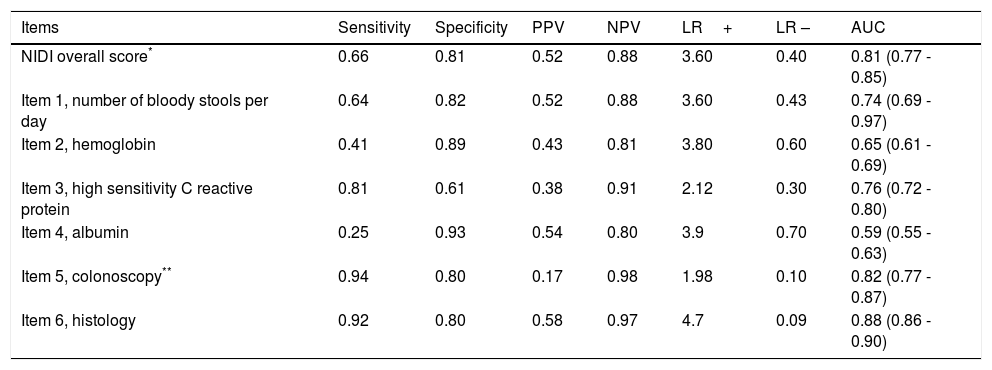
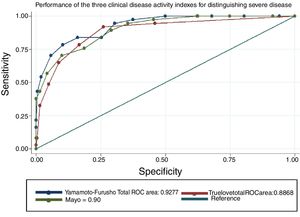
The overall efficacy of the NIDI (Yamamoto-Furusho Index) was 87.2% correctly classified patients. The AUC according to the three scenarios described was 0.93, 0.92, and 0.96, respectively. The comparison results among the three scores were similar, but the score we proposed showed better performance in regard to the endoscopic and histologic severity definitions, with an AUC=0.96, as shown in figure 2, compared with the Truelove and Witts and full Mayo score, whose AUCs were 0.95 and 0.91, respectively (p=0.0009). All items analyzed had a good discriminative capacity with a specificity range between 0.61 and 0.93 and a sensitivity range of 0.25 to 0.94. The single most accurate item was the histopathologic parameter, producing an AUC of 0.88. The detailed parameter comparison is shown in Tables 4 and 5.
Activity score performance in the detection of severe disease defined in various clinical scenarios.
| Gold standard | NIDI or Yamamoto-Furusho Index | Mayo Clinic Score | Truelove and Witts Index | p* |
|---|---|---|---|---|
| Colonoscopy (n=156) | 0.93 (0.91 - 0.94) | 0.95 (0.94 - 0.69) | 0.81 (0.77 - 0.85) | <0.001 |
| Histology (n=37) | 0.92 (0.88 - 0.96) | 0.90 (0.85 - 0.94) | 0.88 (0.83 - 0.94) | 0.02 |
| Colonoscopy and histology (n=28) | 0.96 (0.94 - 0.99) | 0.95 (0.93 - 0.98) | 0.91 (0.86 - 0.97) | 0.009 |
NIDI or Yamamoto-Furusho Index performance and its individual items for distinguishing severe activity.
| Items | Sensitivity | Specificity | PPV | NPV | LR+ | LR – | AUC |
|---|---|---|---|---|---|---|---|
| NIDI overall score* | 0.66 | 0.81 | 0.52 | 0.88 | 3.60 | 0.40 | 0.81 (0.77 - 0.85) |
| Item 1, number of bloody stools per day | 0.64 | 0.82 | 0.52 | 0.88 | 3.60 | 0.43 | 0.74 (0.69 - 0.97) |
| Item 2, hemoglobin | 0.41 | 0.89 | 0.43 | 0.81 | 3.80 | 0.60 | 0.65 (0.61 - 0.69) |
| Item 3, high sensitivity C reactive protein | 0.81 | 0.61 | 0.38 | 0.91 | 2.12 | 0.30 | 0.76 (0.72 - 0.80) |
| Item 4, albumin | 0.25 | 0.93 | 0.54 | 0.80 | 3.9 | 0.70 | 0.59 (0.55 - 0.63) |
| Item 5, colonoscopy** | 0.94 | 0.80 | 0.17 | 0.98 | 1.98 | 0.10 | 0.82 (0.77 - 0.87) |
| Item 6, histology | 0.92 | 0.80 | 0.58 | 0.97 | 4.7 | 0.09 | 0.88 (0.86 - 0.90) |
AUC: Area under the curve; LR –: negative likelihood ratio; LR+: positive likelihood ratio; NPV: negative predictive value; PPV: positive predictive value.
To measure responsiveness by means of sensitivity to change, we compared the first measurement with both the second measurement and the third measurement. In the comparison of the first measurement with the second measurement, the sensitivity to change was -0.7 (OF=3.5, p <0.05), and in the first measurement comparison with the third measurement, the sensitivity to change was -1.2 (OF=4.4, p=0.027). The interclass correlation coefficient of the first measurement against the second measurement was 0.640 (95% CI=0.52-0.73, p <0.05).
DiscussionTo the best of our knowledge, the present study provides the first validation of an integral index for objectively assessing the degree of disease activity in Mexican UC patients that includes clinical, biochemical, endoscopic, and histopathologic features. Previous scores have not been formally validated 8,12,16 due to the methodological designs for clinical trials employed, and thus did not undergo a validation process. Likewise, the majority of the scores do not include all the variables for quantifying UC activity (clinical, biochemical, endoscopic, and histopathologic).17 It is important to note that most of them were developed only as part of the evaluation in clinical trials and not for the purpose of assessing and validating an instrument that specifically measured disease activity. Consequently, the scores were not fully studied in the clinical settings of daily clinical practice.18
It is essential to have available validated UC scores to provide gastroenterologists with a global and objective evaluation that includes clinical, biochemical, endoscopic, and histologic parameters. As mentioned previously, numerous activity indexes are routinely used to classify patients according to disease activity, such as the Truelove and Witts index,8 the Mayo score,12 the Rachmilewitz index, 19 the Lichtiger index, 20 the Seo index,16 the Montreal index, 21 and the Pediatric Ulcerative Colitis Activity Index (PUCAI), 22 among others, but none of them includes histology as a component of their evaluation index.
The NIDI (Yamamoto-Furusho Index) has an advantage over the other scores because it provides a global and objective evaluation of disease activity that includes the clinical, biochemical, endoscopic, and histologic aspects of UC patients. Said integral index helps differentiate and objectively classify disease activity, to optimize medical treatment. We decided to compare the NIDI (Yamamoto-Furusho Index) with the Truelove and Witts index8 and the Mayo score12 because they are two of the most widely used indexes in clinical practice and trials. However, the Mayo score only uses clinical and endoscopic parameters and the Truelove and Witts index only evaluates biochemical and clinical aspects.
The advantage of the NIDI (Yamamoto-Furusho Index) is its objective evaluation of UC that includes histology as part of the patient evaluation, in addition to clinical, biochemical, and endoscopic findings. The importance of histologic activity in the evaluation of UC patients in relation to predicting the risk of relapse at 6 months, a worse disease prognosis, and true UC activity, has recently been described. 23–24
Based on the need to have a score that included the four areas to objectively assess those patients, we decided upon and designed the variables that were included in the NIDI (Yamamoto-Furusho Index). The correlation of our integral index, in comparison with the Truelove and Witts index8 and the Mayo score,12 was highly significant (r=0.817, p <0.001 and r=0.957, p <0.001 respectively), utilizing the histologic evaluation as a gold standard.25 In relation to the full Mayo score, we obtained a correlation close to 1, which was very good for the classification of the patients as being in remission or with mild, moderate, or severe disease activity (r=0.95).
The impact of adding endoscopic evaluation has been shown, compared with simple indexes that only assess signs and symptoms. In a study by Osada et al.,9 they reported a significant positive correlation between endoscopic and histologic scores (r=0.738), clinical activity and endoscopic scores (r=0.444), and clinical and histologic scores (r=0.557), and concluded that total colonoscopy may be indicated when CRP or ESR are elevated in UC patients with clinical remission.
In the present study, the NIDI (Yamamoto-Furusho Index) correlated very well (r=0.767, p <0.001) with the endoscopic activity of UC. Most of the endoscopic indexes that are used in clinical practice or clinical trials, such as the Ulcerative Colitis Endoscopic Index of Severity score (UCEIS),26 the Baron Score,27 the Ulcerative Colitis Colonoscopic Index of Severity (UCCIS),28 the Rachmilewitz Endoscopic Index,19 the Sutherland Index, 29 the Matts Score, 30 and the Blackstone Index, 31 share similar endoscopic variables.
In our study, we decided to include histologic activity as a gold standard 10,24 because it was the only variable that more accurately demonstrated disease activity (r=0.954, p <0.001). The addition of that parameter, which no other index has, enables a better estimation of disease activity, compared with other indexes that underestimate the severity of UC. That is especially true if the result of the endoscopic findings hides the grade of activity as “Mayo 0”, in the case of indexes that include a sub-endoscopic, or even worse, a noninvasive index.
We considered hs-CRP as a variable because it showed a good correlation with histologic activity in UC patients. 32–34 Finally, anemia severity was taken into account in the NIDI (Yamamoto-Furusho Index) because it objectively reflects systemic blood loss in the UC patient. 35–37
The Food and Drug Administration initiated an evaluation of composite indexes in clinical trials and proposed industry guidance for the development of PROs and PRO measures [PROMs] 38 to quantify patient perceptions of their own functional status and well-being. A PRO is any report that comes directly from a patient about his or her health condition or its treatment without interpretation of the patient's response by a clinician or anyone else. However, there is a consensus that the degree of inflammation should be measured using objective instruments such as endoscopy, histology, and/or cross-sectional imaging. It is necessary to correlate the findings in the physical examination and the laboratory and endoscopic studies to be able to make treatment modifications. The use of PROMS can be a support tool to become aware of patient treatment perception, but never to make absolute conclusions about disease activity based solely on clinical outcomes. The aim of the NIDI was to create an integral disease activity scale for patients diagnosed with UC, measured through the global analysis of the objective tools of clinical, histologic, endoscopic, and biochemical parameters, to have a more accurate approach to disease activity.
ConclusionThe NIDI, or Yamamoto-Furusho Index, provides an objective and integral evaluation of disease activity that includes clinical, biochemical, endoscopic, and histopathologic parameters in Mexican patients with UC. The NIDI (Yamamoto-Furusho Index) can possibly optimize medical treatment in patients that have no clinical evidence of disease activity, but present with biochemical, endoscopic, and histopathologic UC activity.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this study/article.
Conflict of interestDr. Jesús Kazuo Yamamoto Furusho is a Speaker, Opinion Leader, and Member of Advisory Board Committees for the following national and international laboratories: Abbvie, Ferring, Hospira, Janssen, Pfizer and Takeda. He is or has been a Speaker for the Almirall, Danone, Farmasa, Grunenthal, and UCB laboratories. He is or has been a lead researcher in international projects for the Abbvie, Allergan, Bristol, Ferring, Pfizer, Roche, Shire, and Takeda laboratories. He is currently the president of the Pan American Crohn's and Colitis Organisation (PANCCO).
The authors K.E. Bozada-Gutiérreza, A. Sánchez-Rodriguezb, F. Bojalil-Romanoa, R. Barreto-Zuñigac, and B. Martínez-Benitezd declare that there is no conflict of interest.
Please cite this article as: Yamamoto-Furusho JK, Bozada-Gutiérrez KE, Sánchez-Rodríguez A, Bojalil-Romano F, Barreto-Zuñiga R, Martínez-Benitez B. Validación de un nuevo índice integral de enfermedad para evaluar el grado de actividad en pacientes mexicanos con colitis ulcerosa: un estudio de cohorte prospectivo. Revista de Gastroenterología de México. 2019;84:317–325.