A 64-year-old man with no comorbidities presented with symptoms for 7 days of jaundice, asthenia, fever, calf muscle pain, mild upper abdominal pain, and reduced urine output. Upon admission to a hospital in Lima, Peru, he was hemodynamically stable, had a score of 15 on the Glasgow coma scale, and had slight abdominal pain in the right hypochondrium and epigastrium. Laboratory work-up reported hemoglobin at 11.8 g/dL, leukocytes at 19,800, platelets at 30,000, urea at 206, creatinine at 5.7, amylase at 684 (normal value < 110 U/L), lipase at 1,283 (normal value < 300 U/L), and altered liver profile (Table 1). Chest x-ray was normal. No gallstones or bile duct alterations were identified in the abdominal ultrasound study.
Liver profile.
| Day 1 | Day 4 | Day 6 | Day 8 | Day 10 | Day 14 | Day 17 | Day 28 | |
|---|---|---|---|---|---|---|---|---|
| Total bilirubin (mg/dL) | 22.4 | 40.2 | 49.3 | 43.5 | 36.5 | 24.9 | 6.6 | 3.0 |
| Direct bilirubin (mg/dL) | 20.9 | 36.8 | 44.8 | 40.3 | 33.6 | 23.6 | 5.6 | 2.4 |
| AST (U/L) | 263 | 172 | 114 | 101 | 83 | 61 | 62 | 48 |
| ALT (U/L) | 126 | 132 | 95 | 69 | 49 | 33 | 26 | 57 |
| ALP (U/L) | 186 | 235 | 325 | 384 | 356 | – | – | 283 |
| GGT (U/L) | 132 | 245 | 308 | 369 | 293 | – | – | 75 |
| Albumin (g/dL) | 2.7 | – | – | – | 2.4 | – | – | – |
The initial diagnostic focus was acute kidney failure secondary to severe acute pancreatitis and probable severe cholangitis. Treatment was started with fluid resuscitation, ceftriaxone, and intravenous metronidazole. The next day the patient was admitted to the intensive care unit. An abdominal tomography scan revealed no significant alterations in the pancreas or liver and the antibiotic was changed to intravenous meropenem for 14 days. Due to the presence of fever, calf muscle pain, marked direct bilirubinemia and no other alterations in the liver profile, and imaging studies that were negative for biliary, pancreatic, or neoplastic disease, an infectious etiology was suspected. Upon broadening the clinical history, it was discovered that the patient had been exposed to rodents, when cleaning a water cistern, 2 weeks prior to his hospital admission. A microscopic agglutination test was performed due to high suspicion of leptospirosis, confirming the diagnosis. Urea and creatinine levels continued to increase, and so hemodialysis was started on day 3. Liver involvement also worsened (Table 1).
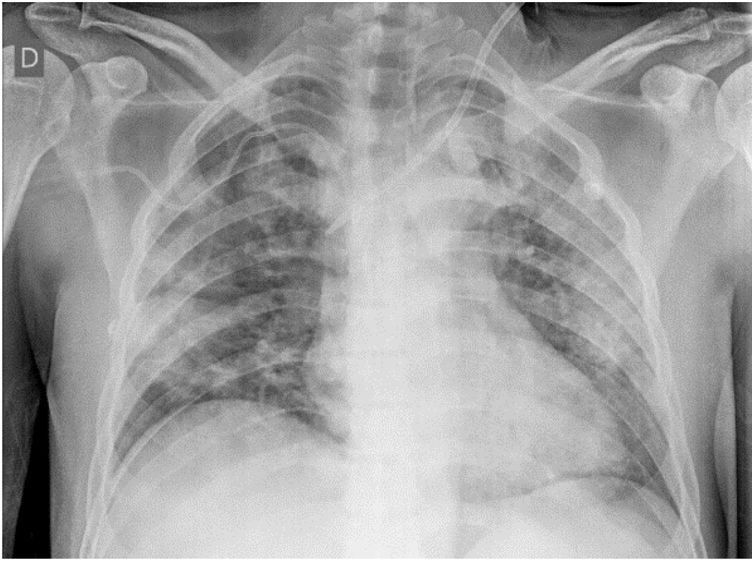
One week later, the patient showed improved liver involvement but continued to undergo hemodialysis and developed respiratory failure due to diffuse alveolar hemorrhage (Fig. 1), with a drop in hemoglobin to 5 g/dL. He was given supplemental oxygen through a face mask with a reservoir bag and did not require mechanical ventilation. The patient finally presented with favorable progression and was discharged on day 31 of hospitalization, continuing hemodialysis as an outpatient.
Leptospirosis is a zoonotic infection with worldwide distribution, that is produced by spirochetes from the genus Leptospira. It predominates in tropical regions, especially during the rainy season, in areas with scant resources and the presence of rodents.1 In Peru, an incidence of 1.55 to 7.11 cases per 100,000 inhabitants was reported for the period from 2017 to 2021.2 In 2021, an additional 513 cases and one death were reported. Approximately 80% of the cases were reported by the departments of Madre de Dios and Loreto (jungle), whereas Lima (coast) reported less than 1.5%2. The patient described herein was from Lima.
Leptospira is maintained in nature due to chronic kidney infection in domestic and wild carriers (rodents, among other small mammals, as well as cattle and domestic dogs and cats) that colonize the brush border of the proximal renal tubule.1
Infection can be produced through direct or indirect contact with the urine or tissues of infected animals. Leptospira spirochetes enter the organism through cuts or abrasions in the skin, through the mucous membranes, or through the inhalation of aerosols, and are disseminated in the blood. Leptospiremia and the associated vasculitis explain the broad spectrum of its clinical presentation.3
The incubation period is 10 days (5-14 days). Leptospirosis can present as a subclinical disease, a self-limited systemic disease (90% of infections), or a severe disease. It can involve 2 phases: an initial septicemic phase in the first 5-7 days, characterized by the presence of Leptospira organisms in the blood or cerebrospinal fluid that cause high fever, headache, muscle pain that is predominant in the calves and lumbar area, abdominal pain, nausea, vomiting, diarrhea, cough, and conjunctival suffusion (red eyes with no secretions). The posterior immune phase lasts 4-30 days, in which circulating IgM antibodies and leptospiruria are produced, and patients can present with jaundice, kidney failure, difficulty breathing, hemoptysis, aseptic meningitis, heart arrythmias, and pancreatitis.3,4 The most characteristic severe form is Weil’s disease. It can appear after the acute phase and is characterized by altered liver function, and pulmonary and renal involvement. The mortality rate in patients with untreated severe disease reaches 40%.5
Patients present with a characteristic liver profile: marked direct hyperbilirubinemia (up to 80 mg/dL), associated with a slight increase (2-3 times the normal value) in other enzymes that indicate cholestasis (ALP, GGT) and a slight increase in transaminases (<200 U/L).6,7Leptospira spirochetes infiltrate the space of Disse and damage the hepatocytes, altering their intercellular binding and causing bile leakage from the bile canaliculi into the sinusoids, which explains the very high levels of direct bilirubin, with no major alterations in ALP or GGT. In addition, due to the fact that there is no hepatocellular necrosis, transaminase levels are not elevated.1
Renal involvement varies in severity, ranging from mild, nonoliguric acute kidney injury to established acute kidney failure. The majority of patients that survive recover their kidney function but some present with lasting chronic kidney disease.3
Some patients present with multifactorial hyperlipasemia and hyperamylasemia that is generally secondary to acute kidney failure and the critical status of the patients. Nevertheless, cases of acute pancreatitis due to leptospirosis, that even include fatal outcomes, have been reported.8 The mechanism of pancreatitis is not clearly defined but it has been postulated that the vasculitis and associated ischemic injury cause the activation of proteolytic enzymes and autodigestion.8 In the present case, the patient had hyperlipasemia and hyperamylasemia that were not associated with typical abdominal pain or contributory imaging studies, and so the diagnosis of acute pancreatitis was ruled out.
Direct and indirect detection methods are available. The direct detection methods include dark-field microscopy and the polymerase chain reaction technique, in both blood and urine. The standard indirect method is the microscopic agglutination test, in which live antigens react with antibodies in the blood sample, with 77.4% sensitivity and 97.6% specificity. Currently in Peru, the microscopic agglutination test is exclusively performed at the Instituto Nacional de Salud, at no cost to the patient.9
When leptospirosis is suspected, antibiotic treatment should be started, albeit, according to a recent meta-analysis, its efficacy continues to be controversial.10 Mild disease is treated with oral doxycycline and severe disease with intravenous penicillin or ceftriaxone.10 Support treatment is essential. Patients that present with acute kidney injury should receive intense intravenous fluid resuscitation and if they progress to oliguric kidney failure, the early start of hemodialysis reduces mortality.11 In addition, due to the associated vasculitis, the use of corticosteroids has been suggested in severe cases that have no pulmonary or renal involvement, but in a systematic review, Rodrigo et al. concluded that there is not sufficient evidence for suggesting their use.12
In patients with acute kidney failure and acute marked direct hyperbilirubinemia, with no major alterations in the remaining liver function tests and with imaging studies that are negative for obstructive biliary disease, rodent exposure should be searched for and leptospirosis added to the differential diagnosis, for the consequent study and early management of the disease, resulting in a decrease in mortality, as occurred in the case of our patient. Likewise, the fact that these patients can present with hyperlipasemia and hyperamylasemia secondary to acute kidney failure and the critical status of the patient should be kept in mind.
Ethical disclosuresInformed consent was not required for the publication of the present case because the article contains no personal data that could identify the patient. In addition, the present work meets the current bioethical research norms.
Financial disclosureNo specific grants were received from public sector agencies, the business sector, or non-profit organizations in relation to this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Flores LE, Carlin A, García CA. Enfermedad de Weil como diagnóstico diferencial en síndrome ictérico: reporte de un caso. Rev Gastroenterol Méx. 2023;88:78–80.