¿ Background
The mammalian intestinal tract contains a complex and diverse society of both pathogenic and non-pathogenic bacteria. While it is estimated that there are more than 400 bacterial species inhabiting the human intestinal tract, many of these are uncultivated microorganisms that have complex interactions with other microflora and their host.1 Recent advances in molecular biology and genetics will facilitate the identification of some unknown organisms through the science of meta-genomics. Subsequent analyses of genetic code may inform microbiologists how to create suitable culture conditions for these unknown organisms to facilitate further study and characterization.2 Although environmental factors and the genetic make-up of the host can modulate the distribution of microbial strains, diet appears to be a major factor in regulating the concentration of individual species of microorganisms that colonize the gut. In addition, several gastrointestinal diseases have been associated with imbalances in the endogenous microflora population.
Recent research has classified three distinct groups of intestinal microorganisms: (1) pathogenic, (2) neutral or innocuous, and (3) beneficial microorganisms. This review focuses on known microbiota imparting a protective or curative effect when the gut environment is challenged. These probiotic microorganisms have a potential therapeutic role in the maintenance of human health and the treatment of various gastrointestinal diseases.3 Through the examination of results obtained from high-quality open-label studies and controlled trials, the value of probiotic therapy in a variety of gastrointestinal diseases is discussed and areas for future research are identified.
¿ Bacterial Pathogenesis of Inflammatory Bowel Disease
Although we are closer than ever to understanding the pathogenesis of Crohn's disease (CD), ulcerative colitis (UC), and pouchitis, it is acutely apparent that there is a myriad of complex associations between an individual's genetics and his luminal microbial environment. New concepts, developed from this appreciation, suggest the involvement of dysfunctional mucosal and systemic immune cells that: (1) are unable to detect and/ or to eradicate potentially injurious microbes, and/or (2) detect normal enteric flora as 'foreign' and set up an inflammatory response that cannot be shut down, and/or (3) alter the enteric microbiota composition, thereby disrupting the otherwise platonic relationship with the host's tissues.
Appreciation of the transmetabolic functioning of the human intestinal tract is a relatively new development. Bacterial strain ratios in otherwise healthy humans are being linked to a range of whole-body manifestations, most notably obesity. With respect to inflammatory bowel disease (IBD), recent studies have identified significant strain variations between healthy and diseased individuals. For instance, patients with either CD or UC had 3-4 log greater amounts of Enterobacteriaceae spp in their tissues compared with healthy individuals. Similarly, a difference in both leukocyte count and the concentration of Faecalibacterium prausnitzii have also been identified between healthy and IBD subjects, and the robustness of the difference in these factors is such that they could be considered as non-invasive markers to identify IBD and help discriminate between CD and UC.
Currently, the exact role of bacteria in the pathogenesis of IBD is unknown, but emerging evidence confirms that the biome of the gut in IBD patients is measurably different from that in healthy individuals. Assuming that IBD does indeed occur as a consequence of an altered host response to mucosal microflora, it is conceivable that the introduction of probiotic organisms as therapeutic agents could change the luminal microflora and/ or enhance epithelial barrier function, and/or mollify the response of the mucosal and systemic immune systems in such a way as to attenuate the intestinal inflammatory response.
¿ Probiotics in the Treatment of Inflammatory Bowel Disease
A disturbance in the gastrointestinal microflora, or the host response to this flora, has been demonstrated to play a critical role in the pathogenesis of IBD. This has lead to attempts to modify the bacterial flora with probiotics, as reviewed in detail elsewhere.4,5 While differences in intestinal flora between healthy individuals and those with IBD have been well documented, it remains unclear whether regional patterns of colonization in the bowel of IBD patients might contribute to disease phenotype. Two recent studies comparing bacterial populations using 16S rRNA analysis from specific biopsy samples have advanced our understanding in that area.6,7 In the first study, by Bibiloni et al.,6 biopsy samples were taken from the intestine of healthy controls, as well as from both healthy and inflammed intestinal regions of UC and CD patients. Similar bacterial compositions were found in healthy and diseased regions within patients, a greater overall bacterial population in UC than CD, and a different variety of bacterial species between the IBD subgroups, with CD patients having a greater number of Bacteroidetes. The second study, by Zhang et al., looked solely at differences between bacterial populations in healthy and diseased regions of the colon of UC patients.7 While the dominant bacterial strain between regions within an individual was highly similar, compositions of Lactobacilli and Clostridium species varied considerably between diseased and healthy regions of an individual's colon. Taken together, results of these trials do not point to a single abnormality that can yet be targeted in IBD therapy, but do suggest that regional differences in bacterial populations may play a role in IBD pathogenesis, and may provide clues towards future therapeutic targets as the volume of available data increases.
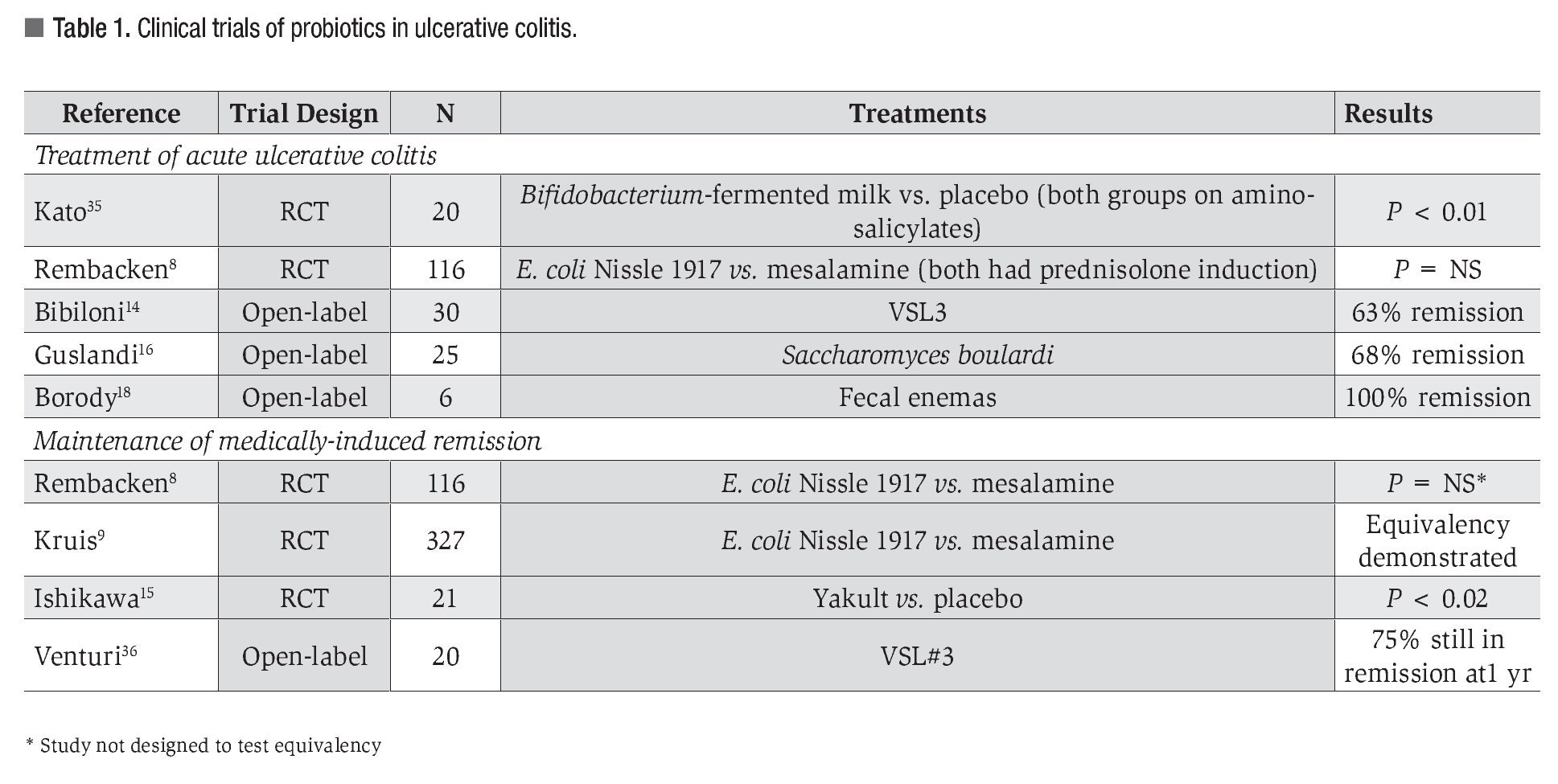
Ulcerative Colitis: A summary of the clinical trials with probiotics in UC is shown in Table 1. The largest study in treatment of active colitis enrolled 116 patients who were randomized to Escherichia coli Nissle 1917 or standard of care mesalamine therapy.8 There was no difference in clinical outcomes between groups, so the authors concluded equivalence between therapies. While this trial was not powered to detect equivalence, a later study of 327 patients with inactive UC assessed E. coli Nissle 1917 against mesalamine and established statistical equivalence.9 This was the best of all evidence to support the use of probiotics for UC therapy, but unfortunately, another study assessing maintenance of remission in 120 patients with E. coli Nissle 1917 failed to show any difference from placebo.10 In 2006, Matthes et al., reported a dose-dependent efficacy of the same probiotic, rectally administered, in the treatment of mild to moderate distal UC.11
In a smaller study, patients with UC received BIFICO capsules (Enterococci, Bifidobacteria, Lactobacilli) to maintain remission induced by sulfa-salazine.12 Patients receiving BIFICO demonstrated lower levels of pro-inflammatory cytokines and NF-kB, and increased levels of IL-10, compared with patients receiving placebo, and relapse in the BIFICO treated group was significantly less (20%) compared with placebo (93%) at 1 year.12 Another small, double blinded, randomized controlled trial used a synbiotic consisting of a prebiotic (Synergy) and a probiotic, Bifidobacterium longum to treat patients with active UC. After 1 month, patients receiving treatment demonstrated improvement in all clinical parameters.13 Uncontrolled pilot studies using VSL3 to treat patients with mild to moderate UC, suggested that this mixture of probiotic bacteria was effective in inducing remission.14 Promising preliminary findings have also been reported with the use of Bifidobacteria-fermented milk15 and Saccharomyces boulardii16 treatment. Randomized placebo-controlled trials using VSL3 to treat UC are currently ongoing in order to confirm the efficacy of this product in treating UC. The possibility of using probiotics to maintain steroid-induced remission was examined in 157 subjects. At the conclusion of the 1-year trial, no significant differences were noted between placebo and probiotic study groups that was in stark contrast with observations from earlier animal model studies.17
A novel protocol for probiotic administration, fecal flora donation from healthy adults, has had promising preliminary results in UC.18 Interestingly, at 1-13 years post-human fecal infusion, all patients were free of endoscopic and histologic evidence of UC.
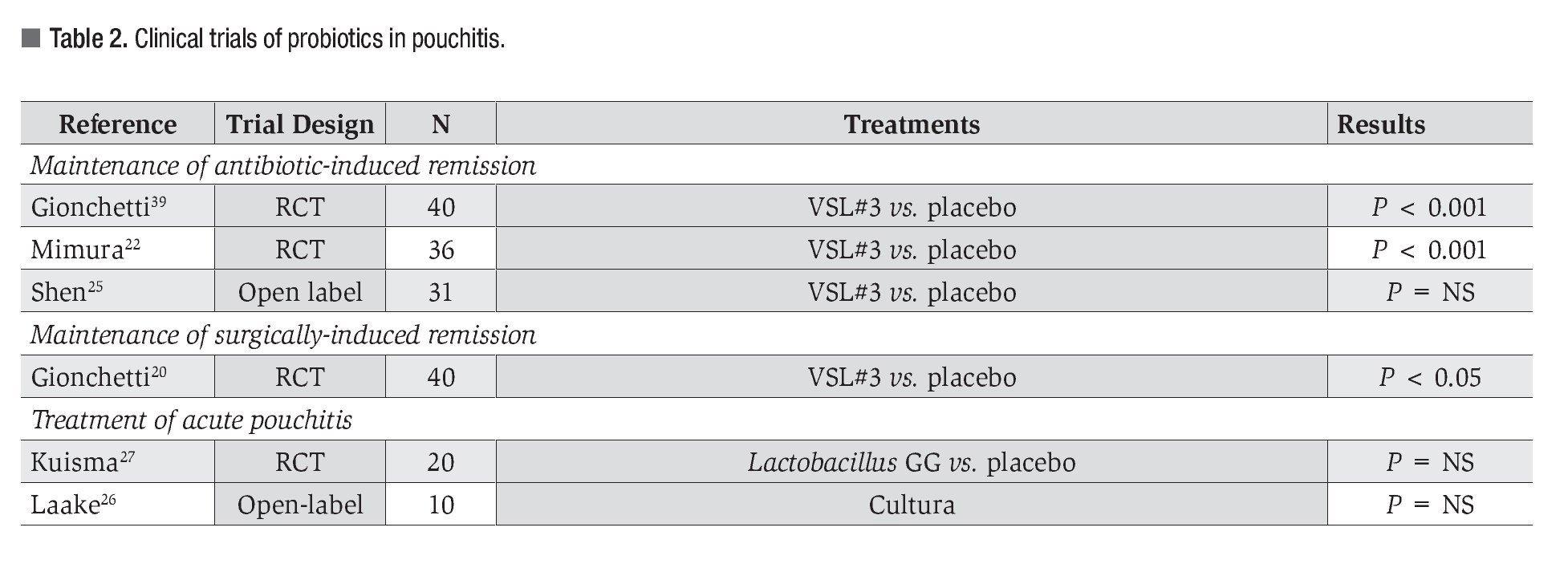
Pouchitis: Summaries of the clinical trials in pouchitis are shown in Table 2. Probiotics have been dramatically effective in the management of pouchitis defined as ileal inflammation following colectomy and ileal pouch formation. Randomized controlled trials have unequivocally shown that the preparation VSL3 is effective in the maintenance of antibiotic-induced remission of pouchitis, and in post-surgical prevention of pouchitis.19-22 In a small trial (n = 31) of subjects with antibiotic-dependent pouchitis, VSL3 was shown to be ineffective in maintaining remission.23-25 Trials using a fermented milk product, Cultura, containing Lactobacilli and Bifidobacteria, have also shown some benefit.26 However, the use of Lactobacillus GG for treatment of acute active pouchitis did not demonstrate efficacy.27
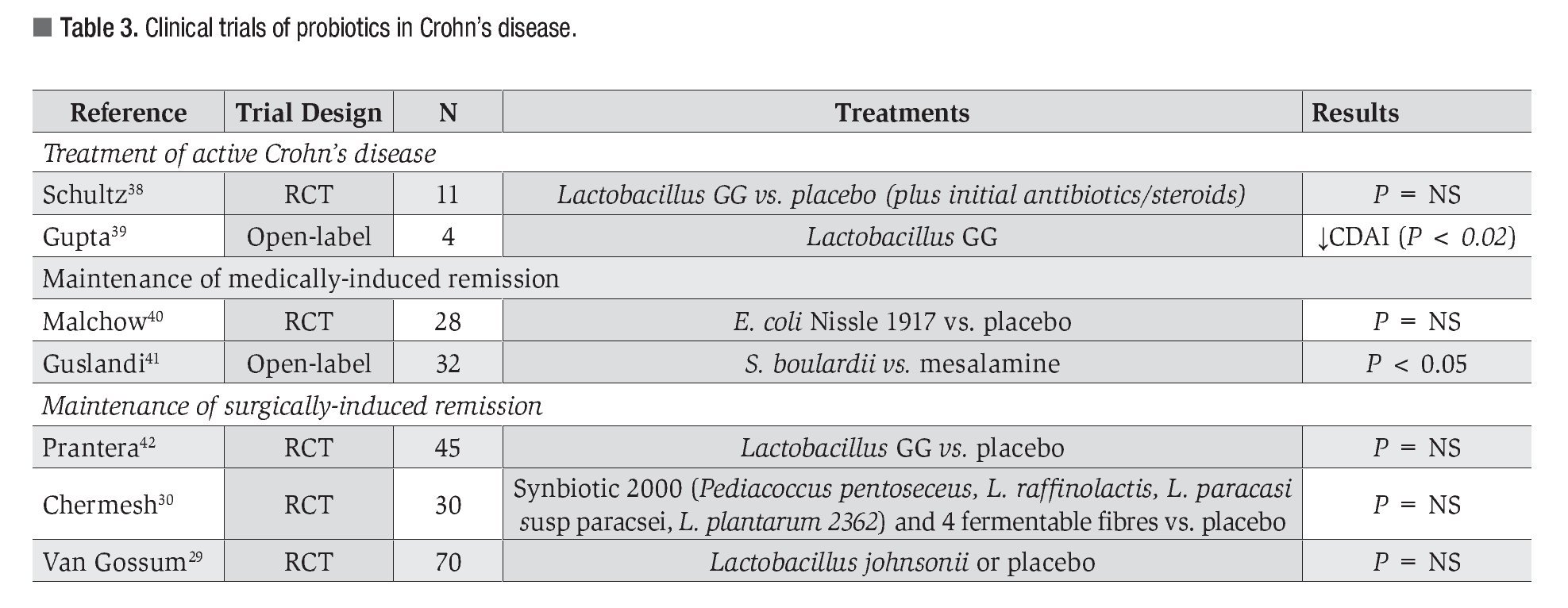
Crohn's Disease: A summary of the clinical trials in CD is shown in Table 3. Limited randomized controlled trials have examined the use of probiotics in the management of CD, and unfortunately, no strong evidence exists for the adoption of their use. A Cochrane review analyzing seven studies concluded that probiotic therapy did not significantly reduce the risk of relapse versus placebo or subsequent to either surgically- or medically-induced remission.28 In remission maintenance, no difference was observed between probiotics and either aminosalicylates or azathioprine. However, those who are intolerant to one or both of these drugs may now look to probiotic therapy before switching to other medicines that are associated with a list of side effects.
In keeping with previous studies, Van Gossum et al. used a single probiotic strain (Lactobacillus johnsonii) and examined its effect on the prevention of early post-operative recurrence of CD.29 Although no significant differences were noted between the two study groups, the underlying trend was that the placebo cohort performed better than the probiotic one. Only one study has investigated the effect of multiple strains of microorganisms (Synbiotic 2000) for the prevention of post-operative CD.30 However, this modification did not provide any beneficial effect on CD recurrence rates. The authors note that the Synbiotic 2000 dosage may need to be increased before observing any distinguishable differences from the placebo group.
As a whole, these trials are underpowered and the majority used a single strain of bacteria; thus, it remains possible that future larger trials with different multi-strain probiotic compounds and predetermined effective dosages will have more positive findings.
¿ Adverse Events
A review outlining the safety of current probiotic compounds has been published.31 Cases of infection due to Lactobacilli and Bifidobacteria are very rare and are estimated to occur at a rate of approximately 0.05% - 0.4% of all cases of infective endocarditis and bacteremia.31 In Finland, no increase in bacteremia caused by Lactobacillus species was seen during the period of 1990 to 2000 despite an increased consumption of Lactobacillus rhamnosus GG.32 Nevertheless, case reports have identified fungemia in two immunosuppressed patients33 and exacerbation of diarrhea in two patients with UC34 who consumed Saccharomyces boulardii.
¿ Conclusion and Future Needs
It is clear that probiotics have beneficial effects. High quality, randomized controlled trial evidence is now available for the therapeutic use of probiotics in selected inflammatory bowel disorders. Nevertheless, not all probiotics are similar and not all probiotics have similar efficacy. Clinical practice must align itself with best evidence and be guided by appropriately controlled randomized clinical trials. Tables 1 through 3 summarize the current published literature on clinical research of probiotics in the treatment of IBD.
Correspondence: 1-10 Zeidler Ledcor Centre 112th Street and 85th Avenue Edmonton, Alberta Canada T6G 2X8
Phone 780 492 6941 Fax 780 492 8121.
E mail:Richard.fedorak@ualberta.ca. http://www.departmentofmedicine.ualberta.ca/gastro/