Double-balloon enteroscopy has been improving the visualization of the entire intestine for more than a decade. It is a complementary method in the study of intestinal diseases that enables biopsies to be taken and treatments to be administered. Our aim was to describe its main indications, insertion routes, diagnostic/therapeutic efficacy, and complications.
Materials and methodsAll patients referred to our unit with suspected small bowel pathology were included. The insertion route (oral/anal) was determined through diagnostic suspicion. The variables measured were: insertion route, small bowel examination extent, endoscopic diagnosis/treatment, biopsy/histopathology report, complications, and surgical findings.
ResultsThe study included 28 double-balloon enteroscopies performed on 23 patients, of which 10 were women and 13 were men (mean age of 52.95 years). The oral approach was the most widely used (n=21), the main indication was overt small bowel bleeding (n=16), and the general diagnostic rate was 65.21%. The therapeutic intervention rate was 39.1% and the procedure was effective in all the cases. The most widely used treatment was argon plasma therapy (n=7). The complication rate was 8.6%; one patient presented with low blood pressure due to active bleeding and another had deep mucosal laceration caused by the argon plasma.
ConclusionsDouble-balloon enteroscopy is a safe and efficacious method for the study and management of small bowel diseases, with an elevated diagnostic and therapeutic rate.
La enteroscopia de doble balón (EDB) tiene más de una década mejorando la visualización de la totalidad del intestino. Es un método complementario en el estudio de enfermedades intestinales, permite tomar biopsias y efectuar tratamientos. El objetivo fue describir las principales indicaciones, el tipo de abordaje, la eficacia diagnóstica/terapéutica y sus complicaciones.
Material y métodosSe incluyó a todos los pacientes referidos a nuestra unidad con sospecha de patología de intestino delgado. El tipo de abordaje (oral/anal) fue determinado por la sospecha diagnóstica. Las variables medidas fueron: tipo de abordaje, alcance de intestino delgado, diagnóstico/tratamiento endoscópico, toma de biopsias/reporte histopatológico, complicaciones y hallazgos quirúrgicos.
ResultadosSe incluyeron 28 EDB realizadas a 23 pacientes, 10 mujeres y 13 hombres (media de edad 52.95 años). La vía de abordaje más usada fue la oral (n=21). La principal indicación fue sangrado de intestino delgado evidente (n=16). La tasa diagnóstica general fue del 65.21%. La tasa de intervención terapéutica fue del 39.1%, la cual fue eficaz en todos los casos. El tratamiento más utilizado fue terapia de argón plasma (n=7). La tasa de complicaciones fue del 8.6%. Un paciente presentó hipotensión por hemorragia activa y uno sufrió laceración profunda de la mucosa por aplicación de argón plasma.
ConclusionesLa EDB es un método eficaz y seguro para el estudio y manejo de enfermedades originadas en el intestino delgado, con una elevada tasa diagnóstica y terapéutica.
The technologic advances emerging over the last decades have enabled the endoscopic methods of small bowel examination to be perfected, as illustrated by video capsule endoscopy and device-assisted enteroscopy (DAE). The latter include the balloon-guided endoscope (NaviAid device; SMART Medical Systems Ltd, Ra’anana, Israel), spiral enteroscopy, single-balloon enteroscopy, and double-balloon enteroscopy (DBE).1 The patent application for DBE and ethics committee authorization for its use in humans were carried out in Japan in 1998,2 and in 2001, the report by Yamamoto et al.,2,3 the first on this new method, was published. Double-balloon enteroscopy consists of an endoscope coupled with an overtube with a hydrophilic coating that facilitates the movement of the two components and the intestinal lumen. Latex balloons are mounted at the distal ends of each component. The balloons are coordinatedly inflated and deflated to facilitate both the advancement into the small bowel and its examination. DBE insertion routes can be either anterograde (oral), retrograde (anal), or both,2–4 and they can be performed on the same day.5 DBE has been in use for more than a decade, and it enables the entire intestine to be viewed.1–4,6,7 Video capsule endoscopy is currently considered the initial method of choice in studying small bowel diseases (small bowel bleeding, tumors, polypoid syndromes, Crohn's disease, and celiac disease), because it is minimally invasive, and DBE is considered a complementary method (because it is invasive, requires sedation, and can have serious complications) that allows different types of lesions in that part of the digestive tract to be biopsied and treated.1,5,7 DBE is also indicated when there are surgical alterations of the gastrointestinal tract in patients undergoing endoscopic retrograde cholangiopancreatography (ERCP) or technical difficulties in patients undergoing colonoscopy.4–7
Finally, DBE can play a primary role in the study of small bowel diseases when video capsule endoscopy is contraindicated or unavailable.1
Few hospital centers in our environment are equipped with this endoscopic method for studying small bowel diseases. It has been available at our hospital since the beginning of 2015, and therefore we decided to report on our experience with its use.
The aim of our study was to describe the main indications, type of approach, diagnostic/therapeutic yield, and complications involved in DBE.
Materials and methodsAll the patients referred to the Western Endoscopy Unit of the Hospital Versalles de Guadalajara with the suspicion of a small bowel disease were included in the study.
The sex, age, referral diagnosis, and when available, the results of previous video capsule endoscopy, upper and lower gastrointestinal endoscopy, and radiologic studies were recorded for each patient. The insertion route (oral or anal) was determined by lesion location suspicion according to the clinical manifestations (oral, when there was melena, and anal, when there was hematochezia) and the findings from previous studies in each case. The DBE data obtained were: type of approach (oral, anal, or both), depth to which the small bowel was explored, diagnosis made and endoscopic treatment carried out, biopsies and histopathologic report, and complications and surgical findings, depending on the case. All the patients were adults and gave their informed consent. No pregnant women were included in the study.
None of the patients had contraindications for undergoing the procedure, nor did they have previous DBE studies. Preparation consisted of fasting for 8h before the anterograde (oral approach) examination and fasting for 8h plus standard bowel preparation (4 l of polyethylene glycol) the afternoon prior to undergoing the retrograde (anal approach) study. Bowel preparation was considered adequate based on the Boston scale that divides the colon into 3 segments (right, transverse, and left), with a score of 0 to 3 for each segment. The minimum score is 0 (unprepared colon) and the maximum score is 9 (perfectly clean colon with no residual liquid).8
A Fujinon double-balloon enteroscope (Fujinon EN-450T5, Fujinon Inc, Saitama Japan) was employed. It is composed of a high-resolution video processor, a 200cm-long insertion tube, and a 140cm-long overtube with an external diameter of 13.2mm. The 2 latex balloons (one at the end of the endoscope and the other at the end of the overtube) were controlled by an electronic pump (Fuji Balloon Controller PB-20, Fujinon Inc, Japan).
The enteroscopies were performed at the endoscopy unit with no fluoroscopic control and with the patient under monitored conscious sedation. The depth of the final insertion and the location of the lesions were calculated by adding the individual distances of each advance of the enteroscope (between 0 and 40cm) from the pylorus, in the anterograde procedures, and from the ileocecal valve in the retrograde procedures. The intestinal mucosa was tattooed with organic carbon dye for medical use at the most distal site reached during the examination, along with lesions that would be easily located for future interventions. Each procedure was ended once the lesion justifying the referral diagnosis was found, the most distal site possible from the intestinal lumen was reached, or when a complication arose. Endoscopic treatment or biopsy were carried out on all lesions that required one or the other.
All the procedures were performed by a single endoscopist that adhered to the guidelines recommended by national and international associations for privileging, credentialing, and proctoring to perform enteroscopy and other endoscopic procedures.9,10
The data were analyzed using the Windows SSPS version 20.0 database.
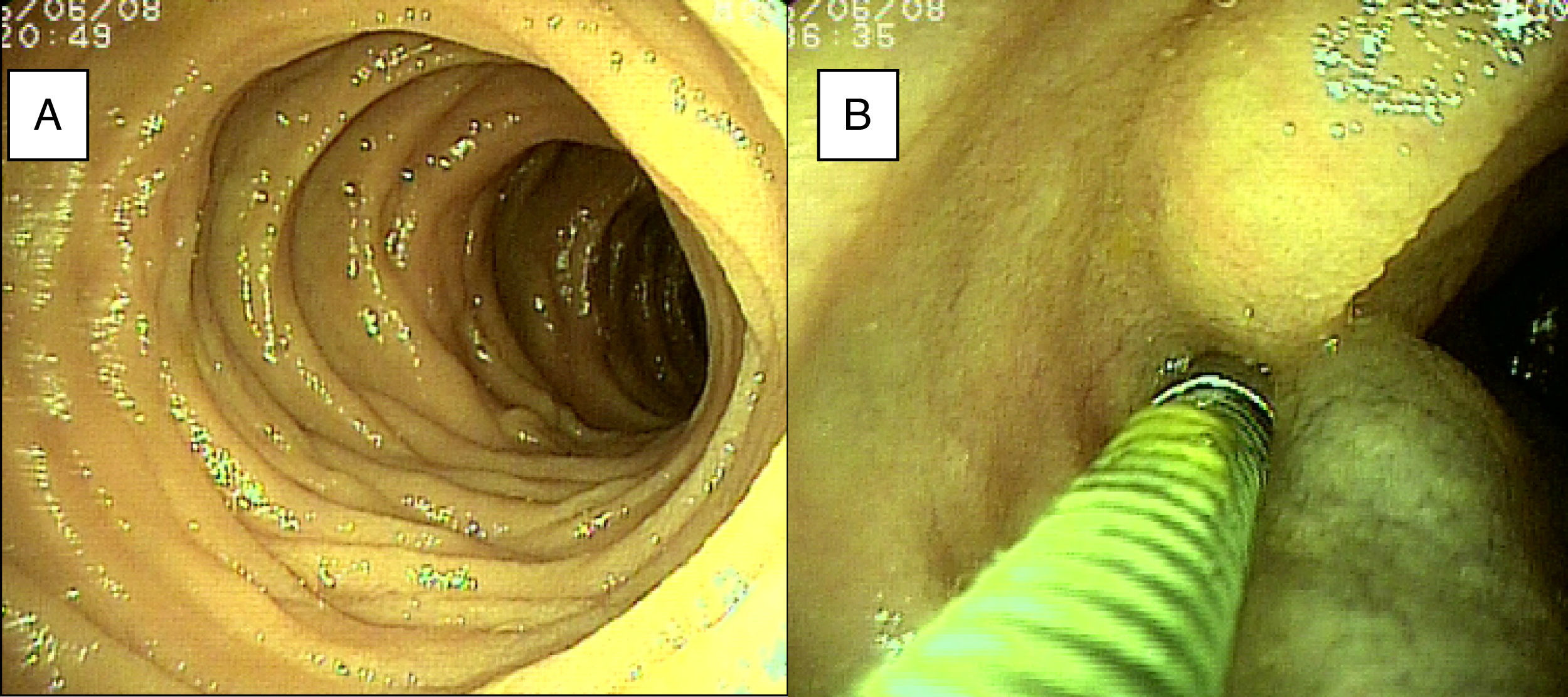
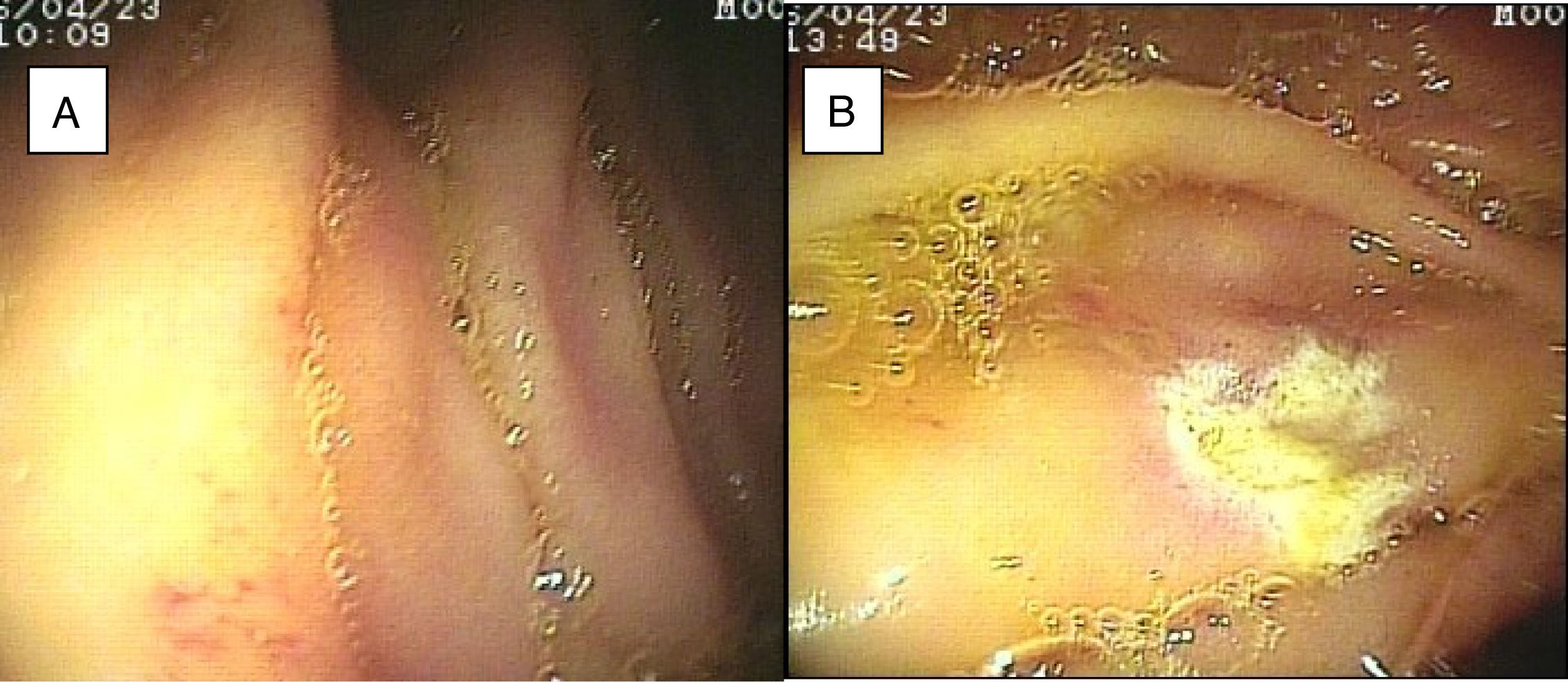
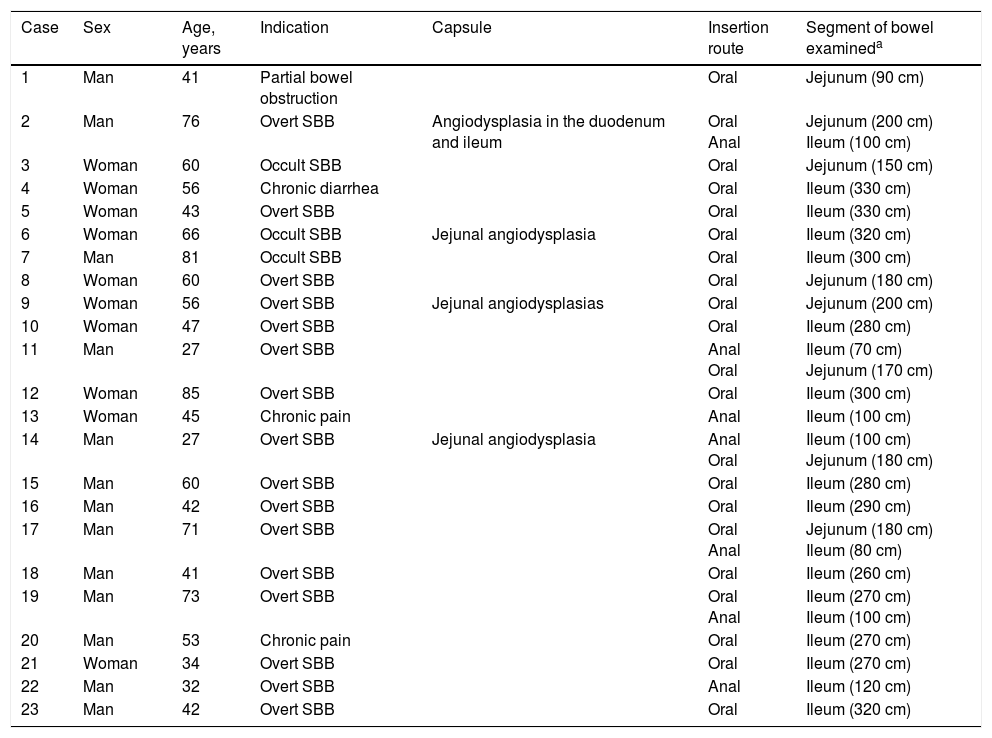
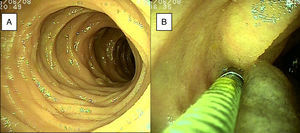
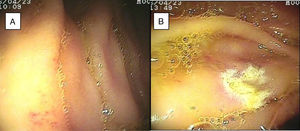
ResultsTwenty-eight double-balloon enteroscopies were performed on a total of 23 patients,10 women and 13 men. They were between 23 and 85 years of age (mean age 52.95 years) and were referred from tertiary care hospitals within the time frame of January 28, 2015 and April 2, 2016. The insertion route in 16 patients was oral, in 2 patients it was anal, and 5 patients (10 procedures) had both the oral and anal approaches. Bowel preparation was adequate in all the patients with the anal insertion route (≥ 6 points on the Boston scale), except for one, that had massive bleeding that prevented lesion identification in the small bowel. The main indications (Table 1) for the procedure were: 19 patients with small bowel bleeding (SBB), 16 of whom had the referral diagnosis of overt SBB and 3 had occult SBB, manifested by anemia with a positive fecal occult blood test; 2 patients with chronic abdominal pain; one patient with chronic diarrhea diagnosis; and one patient diagnosed with intermittent partial small bowel obstruction. All the patients had upper and lower gastrointestinal endoscopies with no abnormal findings to justify their referral. All the patients with small bowel bleeding had abdominal tomography scans as part of their evaluation and they were normal. Only 3 patients diagnosed with overt SBB and one with occult SBB had a previous capsule endoscopy study (Table 1). Figure 1 shows normal mucosa of the duodenum (A) and jejunum (B) tattooed with organic carbon dye.
Main demographic characteristics, indications, insertion route, and length in cm of examination.
| Case | Sex | Age, years | Indication | Capsule | Insertion route | Segment of bowel examineda |
|---|---|---|---|---|---|---|
| 1 | Man | 41 | Partial bowel obstruction | Oral | Jejunum (90 cm) | |
| 2 | Man | 76 | Overt SBB | Angiodysplasia in the duodenum and ileum | Oral Anal | Jejunum (200 cm) Ileum (100 cm) |
| 3 | Woman | 60 | Occult SBB | Oral | Jejunum (150 cm) | |
| 4 | Woman | 56 | Chronic diarrhea | Oral | Ileum (330 cm) | |
| 5 | Woman | 43 | Overt SBB | Oral | Ileum (330 cm) | |
| 6 | Woman | 66 | Occult SBB | Jejunal angiodysplasia | Oral | Ileum (320 cm) |
| 7 | Man | 81 | Occult SBB | Oral | Ileum (300 cm) | |
| 8 | Woman | 60 | Overt SBB | Oral | Jejunum (180 cm) | |
| 9 | Woman | 56 | Overt SBB | Jejunal angiodysplasias | Oral | Jejunum (200 cm) |
| 10 | Woman | 47 | Overt SBB | Oral | Ileum (280 cm) | |
| 11 | Man | 27 | Overt SBB | Anal Oral | Ileum (70 cm) Jejunum (170 cm) | |
| 12 | Woman | 85 | Overt SBB | Oral | Ileum (300 cm) | |
| 13 | Woman | 45 | Chronic pain | Anal | Ileum (100 cm) | |
| 14 | Man | 27 | Overt SBB | Jejunal angiodysplasia | Anal Oral | Ileum (100 cm) Jejunum (180 cm) |
| 15 | Man | 60 | Overt SBB | Oral | Ileum (280 cm) | |
| 16 | Man | 42 | Overt SBB | Oral | Ileum (290 cm) | |
| 17 | Man | 71 | Overt SBB | Oral Anal | Jejunum (180 cm) Ileum (80 cm) | |
| 18 | Man | 41 | Overt SBB | Oral | Ileum (260 cm) | |
| 19 | Man | 73 | Overt SBB | Oral Anal | Ileum (270 cm) Ileum (100 cm) | |
| 20 | Man | 53 | Chronic pain | Oral | Ileum (270 cm) | |
| 21 | Woman | 34 | Overt SBB | Oral | Ileum (270 cm) | |
| 22 | Man | 32 | Overt SBB | Anal | Ileum (120 cm) | |
| 23 | Man | 42 | Overt SBB | Oral | Ileum (320 cm) |
SBB: Small bowel bleeding
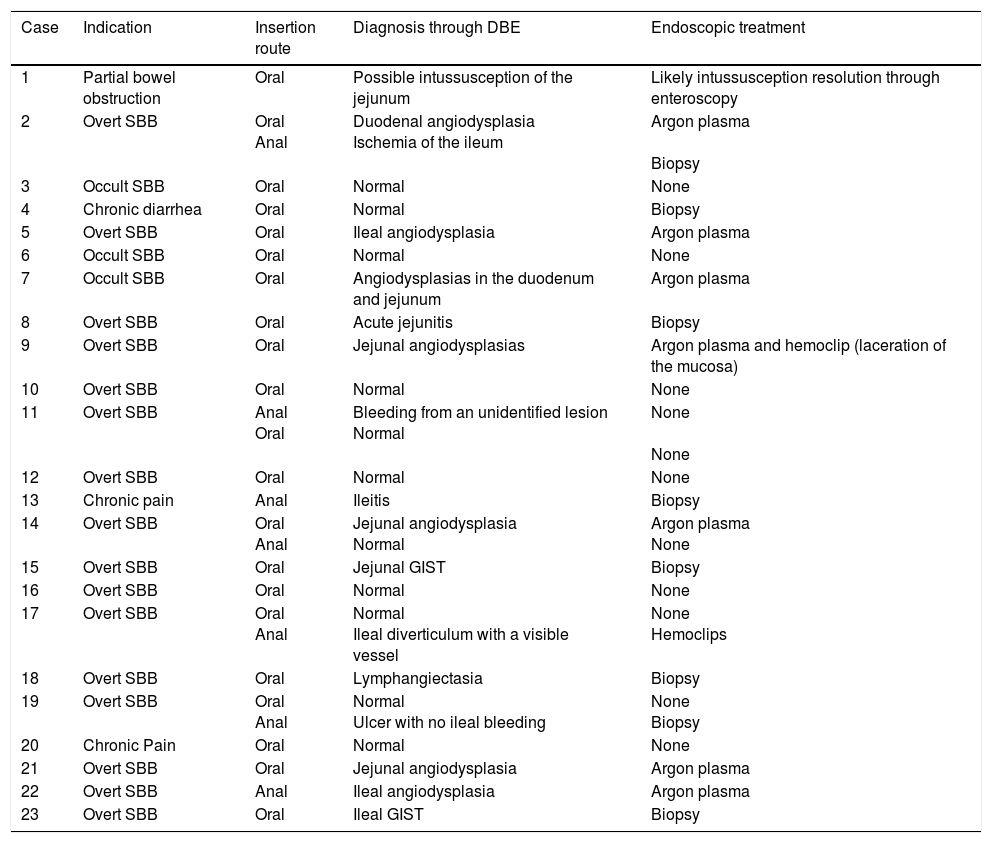
Table 1 describes the segment of the small bowel examined in each procedure. The mean length of insertion was 246.19cm (range of 90-330cm) beyond the pylorus and 95.71cm (range of 70-120cm) beyond the ileocecal valve. It was not possible to examine the entire small bowel in any of the cases. In one patient with the oral approach, the examination reached 50cm from the ileocecal valve, which was corroborated by locating the tattoo during the resection of the segment of the intestine affected by a jejunal gastrointestinal stromal tumor (GIST) located during the DBE. Complete examination of the small bowel was not possible in the 5 patients with both approaches (oral and anal) either, because the tattoo from the first study was not observed. The oral and anal approaches were performed in the same event in 4 patients. In the last patient, the anal insertion route was first employed, but the patient presented with such significant low blood pressure (due to severe gastrointestinal bleeding) that the examination had to be stopped. Two days later, the oral insertion route was employed. This approach was chosen because in the first study, upon reaching the ileum, there were abundant remains of fresh blood and coagulates that did not allow the bleeding site to be identified, suggesting a more proximal one, making the oral route the better option. Table 2 shows the diagnoses found and the corresponding treatments.
Diagnosis through double-balloon enteroscopy and endoscopic treatment.
| Case | Indication | Insertion route | Diagnosis through DBE | Endoscopic treatment |
|---|---|---|---|---|
| 1 | Partial bowel obstruction | Oral | Possible intussusception of the jejunum | Likely intussusception resolution through enteroscopy |
| 2 | Overt SBB | Oral Anal | Duodenal angiodysplasia Ischemia of the ileum | Argon plasma Biopsy |
| 3 | Occult SBB | Oral | Normal | None |
| 4 | Chronic diarrhea | Oral | Normal | Biopsy |
| 5 | Overt SBB | Oral | Ileal angiodysplasia | Argon plasma |
| 6 | Occult SBB | Oral | Normal | None |
| 7 | Occult SBB | Oral | Angiodysplasias in the duodenum and jejunum | Argon plasma |
| 8 | Overt SBB | Oral | Acute jejunitis | Biopsy |
| 9 | Overt SBB | Oral | Jejunal angiodysplasias | Argon plasma and hemoclip (laceration of the mucosa) |
| 10 | Overt SBB | Oral | Normal | None |
| 11 | Overt SBB | Anal Oral | Bleeding from an unidentified lesion Normal | None None |
| 12 | Overt SBB | Oral | Normal | None |
| 13 | Chronic pain | Anal | Ileitis | Biopsy |
| 14 | Overt SBB | Oral Anal | Jejunal angiodysplasia Normal | Argon plasma None |
| 15 | Overt SBB | Oral | Jejunal GIST | Biopsy |
| 16 | Overt SBB | Oral | Normal | None |
| 17 | Overt SBB | Oral Anal | Normal Ileal diverticulum with a visible vessel | None Hemoclips |
| 18 | Overt SBB | Oral | Lymphangiectasia | Biopsy |
| 19 | Overt SBB | Oral Anal | Normal Ulcer with no ileal bleeding | None Biopsy |
| 20 | Chronic Pain | Oral | Normal | None |
| 21 | Overt SBB | Oral | Jejunal angiodysplasia | Argon plasma |
| 22 | Overt SBB | Anal | Ileal angiodysplasia | Argon plasma |
| 23 | Overt SBB | Oral | Ileal GIST | Biopsy |
DBE: Double-balloon enteroscopy; GIST: Gastrointestinal stromal tumor; SBB: Small bowel bleeding.
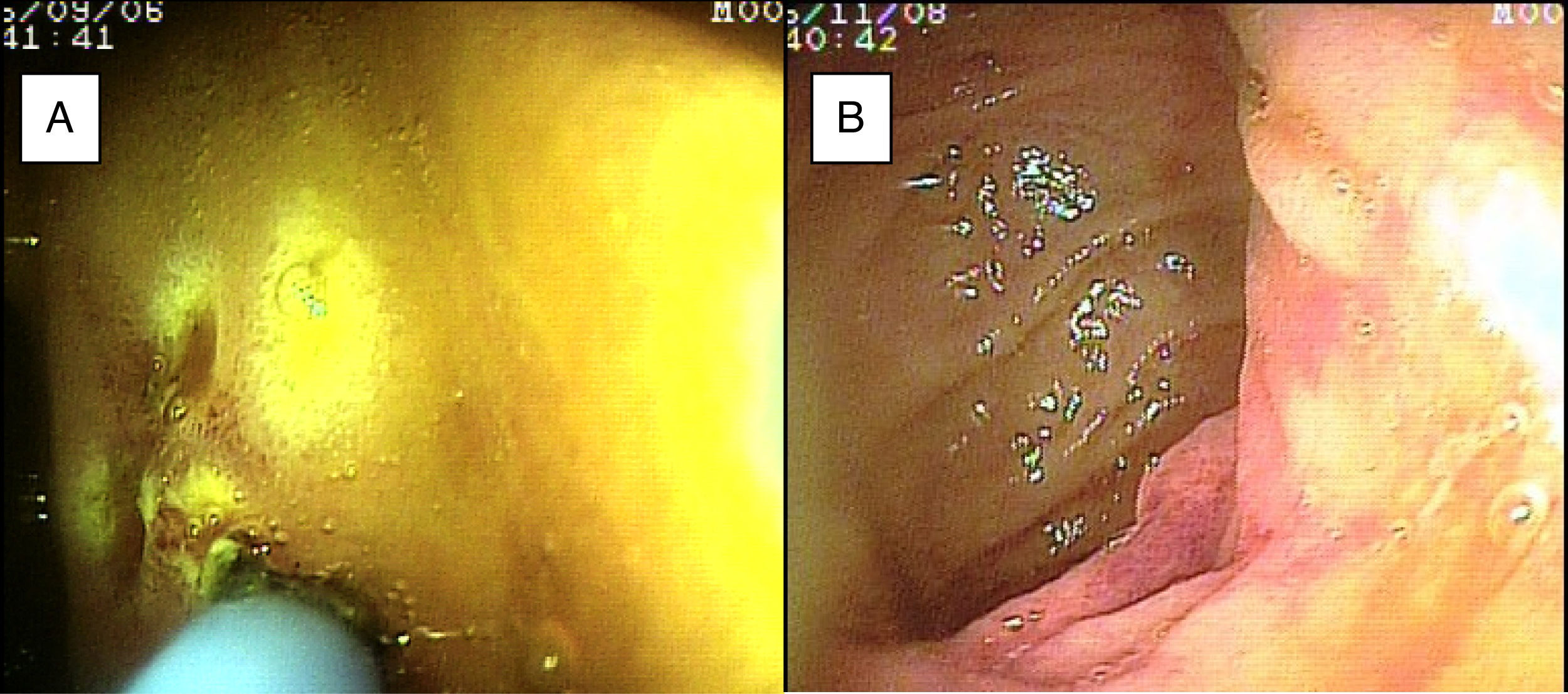
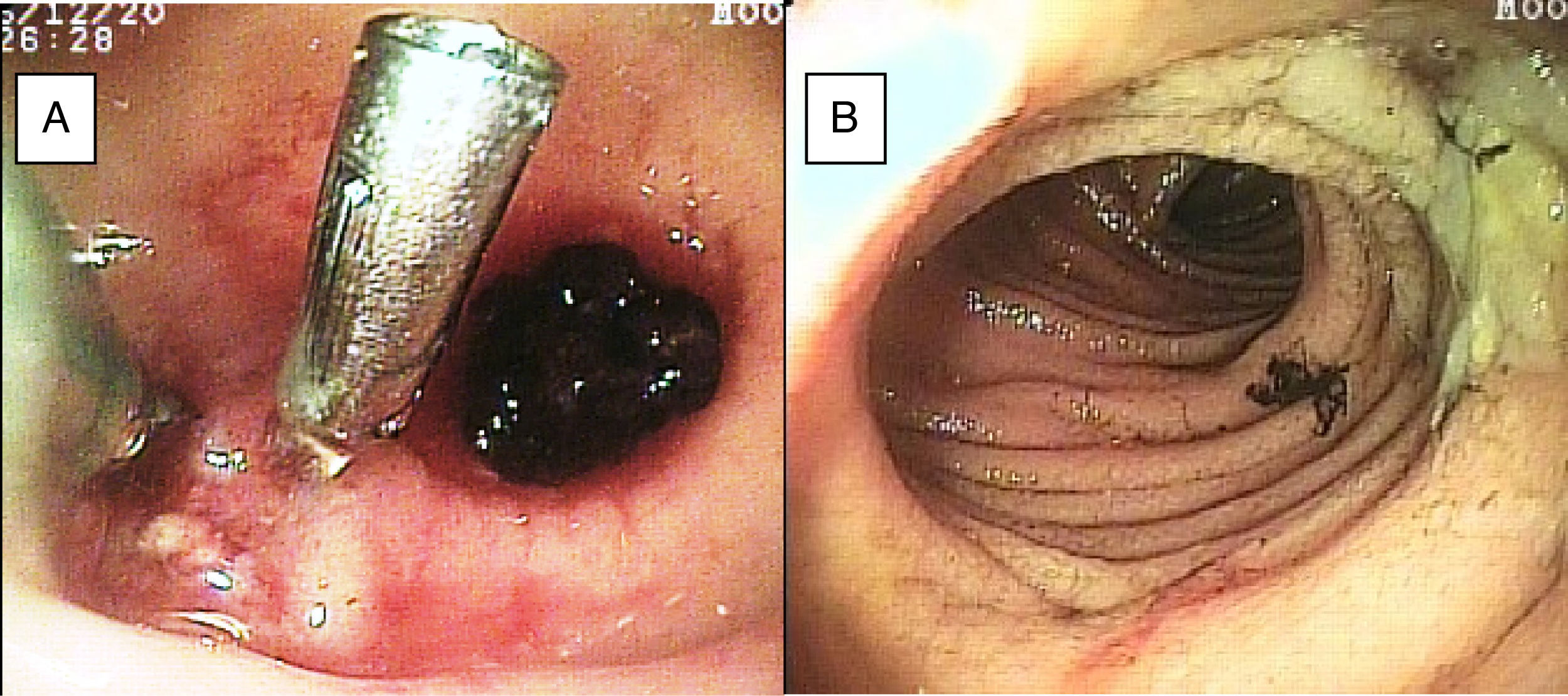
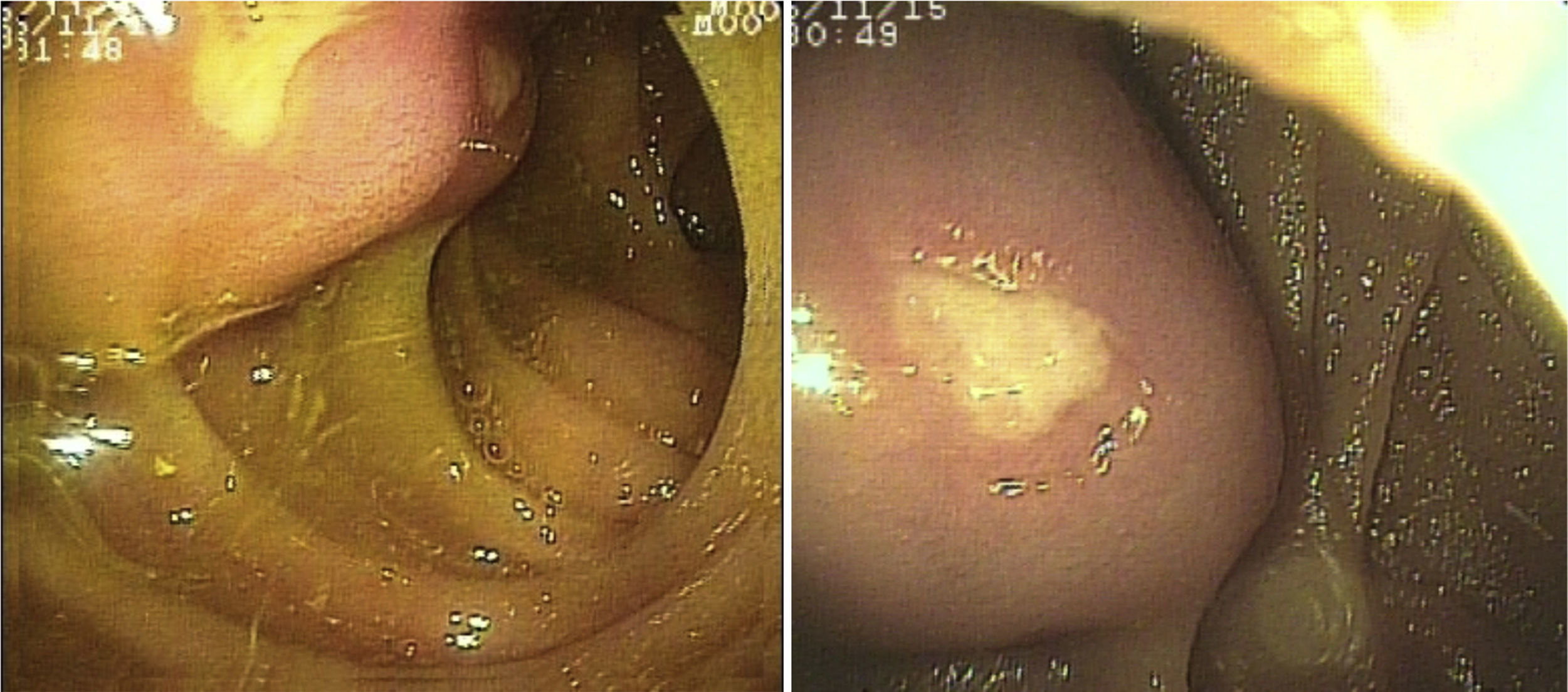
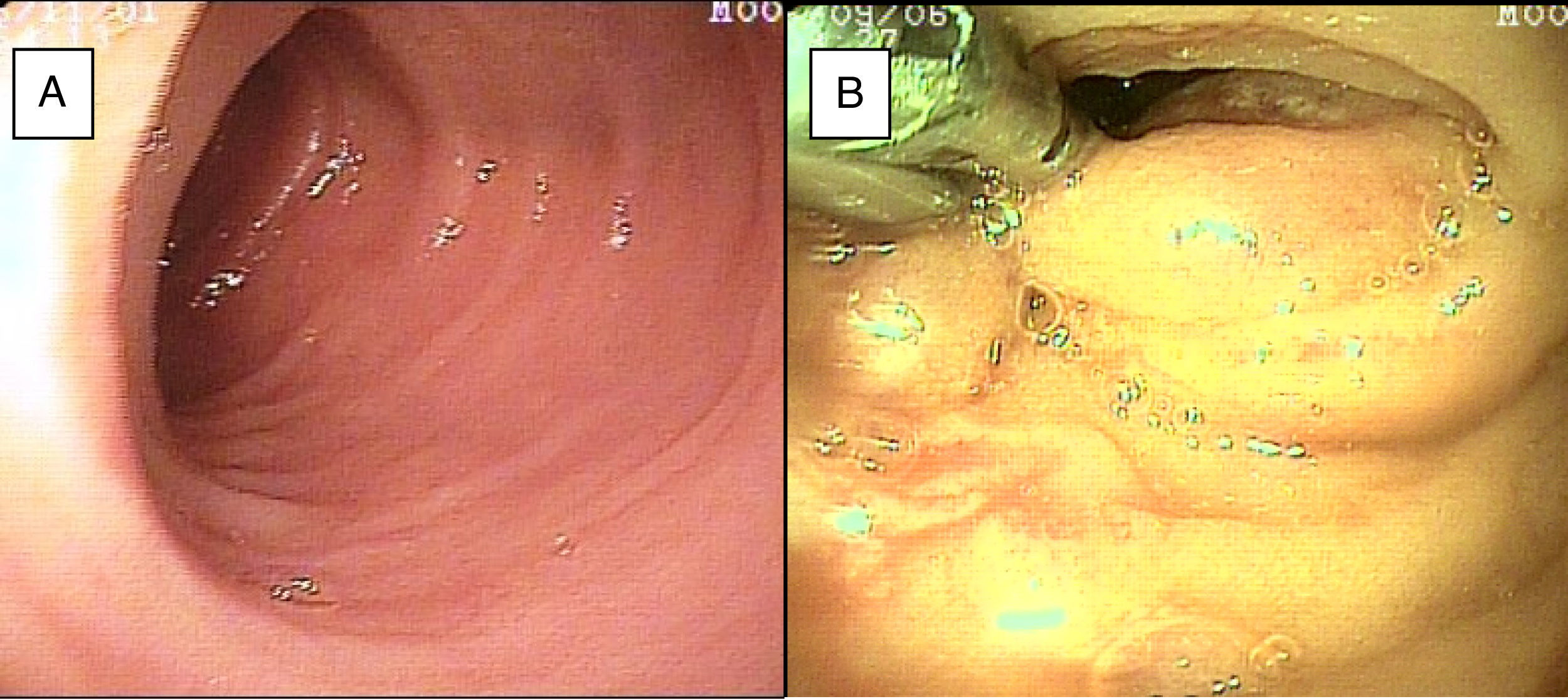
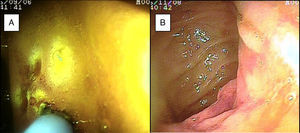
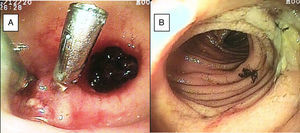
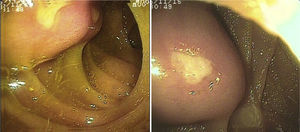
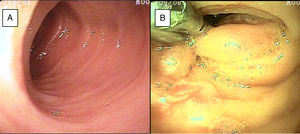
SBB was the main indication for the examination in 82.6% of the cases (19 patients). Sixteen of those patients had a referral diagnosis of overt SBB, and 5 of them underwent both insertion routes because of active bleeding (it was impossible to identify the cause of the bleeding in the first enteroscopy). One of those patients presented with duodenal angiodysplasia (Fig. 2A) and endoscopic changes of ischemia in the ileum, and another patient presented with jejunal angiodysplasia (Fig. 2B). A diverticulum with a coagulate and a visible vessel was found in a third case and was satisfactorily treated through hemoclip placement (Fig. 3A). The fourth patient (Fig. 3B) presented with an ulcerated tumor-like lesion in the ileum (histologic study reported a benign deep ulcer). The last case with the 2 approaches was a patient with chronic kidney disease, on hemodialysis. In the examination with the initial anal insertion route, there was abundant fresh blood in the ileum due to active bleeding, complicated with severe low blood pressure, and the procedure was suspended. Two days later, a new anterograde enteroscopy was performed due to the suspicion of a more proximal lesion, but the site of the bleeding was not found. This last patient had no new bleeding episodes in the following weeks. The rest of the cases with overt SBB diagnosis (11 patients) underwent only one type of approach, 10 oral and one anal. Of the 11 patients, 4 had angiodysplasias (2 in the jejunum and the other 2 in the ileum). One patient presented with acute jejunitis, two patients had an ulcerated GIST in the jejunum (Fig. 4) and ileum (corroborated through endoscopic biopsy and surgical specimen, given that they were referred to elective surgery with bowel resection). Another patient presented with a polypoid lesion with a histopathologic report of lymphangiectasia, and 3 patients had normal enteroscopies. One of the patients with angiodysplasia in the jejunum initially underwent oral insertion route, but due to the heavy bleeding, a lesion in the terminal ileum was suspected and the anal approach was decided upon, but no alterations were found in that segment. The 3 patients with the referral diagnosis of occult SBB had the oral approach. Two of those patients had normal enteroscopies and the third had angiodysplasias in the duodenum and jejunum (Fig. 5A before treatment and 5B after the application of argon plasma).
The oral insertion route was used in the patient with the referral diagnosis of intermittent partial small bowel obstruction, and changes due to a possible intussusception in the jejunum were found and resolved through DBE. The patient with chronic diarrhea underwent the oral approach, with normal endoscopic findings. The mucosa was biopsied and no alterations were reported. In the 2 patients referred with diagnoses of chronic abdominal pain, one underwent the anterograde procedure, with normal findings, and the other had the retrograde procedure, finding endoscopic and histologic changes of nonspecific ileitis (Fig. 6A).
Of the 28 enteroscopies performed, biopsies were taken with forceps in 8 procedures, argon therapy was applied in 7, and hemoclips were placed in 2 (one due to the finding of recent bleeding with a visible vessel in a diverticulum, and the other because of deep mucosal laceration during argon plasma application). In one patient, a possible intussusception found in the jejunum was resolved through the DBE and there was no need for intervention in 11 of the procedures (normal findings). Therapeutic intervention was efficacious in all the cases.
In relation to major complications from the procedure, one patient presented with low blood pressure due to active bleeding, causing study suspension. It was newly performed 2 days later with no new complications. Another patient presented with deep laceration of the mucosa during argon plasma application that was detected during the same procedure and treated with the placement of 2 hemoclips. The laceration was only documented through endoscopic appearance and no other confirmation methods were employed (fig. 6B). Two patients were referred to elective surgery and bowel resection due to GIST diagnosis.
Because ours was a cross-sectional study, there was no follow-up on patient progression. However, we stayed in contact with the treating physician for the possible appearance of complications in the cases that had endoscopic biopsy or in those that were referred for surgery (only 2 patients). For the same reason, no long-term follow-up was conducted, rebleeding rates were not evaluated, nor were other symptoms analyzed after the application of any type of endoscopic therapy.
Discussion and conclusionsDBE has been performed at our gastrointestinal endoscopy unit for approximately 15 months. During this period, we have carried out 28 DBEs on 23 patients. A total of 69.5% of those patients (16 out of 23) had procedures with the oral insertion route, 8.6% (2 out of 23) with the anal insertion route, and 21.7% (5 out of 23 patients; 10 procedures) had both approaches (oral and anal). The oral approach was the most widely used insertion route in our patients, which concurs with other studies.11–14 The oral route in DBE is the recommended initial approach when there are no clinical manifestations or results from other study methods (such as capsule endoscopy or radiologic imaging) that indicate that the lesions are in the terminal part of the small bowel.5,15 Small bowel bleeding was the main indication for DBE in our study group (82.6%; 19 of 23 patients), and included overt SBB and occult SBB. It has also been the main indication in other studies,11–14 but its frequency was higher in our study than that reported by other authors. Mönkemüller et al. stated that 55% of their patients underwent DBE due to SBB,12 whereas in a study by di Caro et al. conducted at 4 European centers, frequency was 53%,11 and Kita et al. reported a frequency of 49.2%.12 In a more recent study by Akarsu et al. at a single center in Turkey, small bowel bleeding was the main indication for DBE in 26%.14 Some of those studies separated patients with anemia, overt SBB, and occult SBB into separate categories,11,14 possibly contributing to the differences in frequencies found, given that we included them in the same group. Our small sample could also be another factor. Other DBE indications in our study were: chronic abdominal pain in 8.69%, chronic diarrhea in 4.34%, and intermittent partial small bowel obstruction in 4.34%.
The general diagnostic yield reported in the literature for DBE varies widely from 40-80% and it has a 15-55% therapeutic intervention rate.11–17 In our study, the general diagnostic yield for DBE was 65.21%, the therapeutic intervention rate was 39.1%, and the biopsy rate was 34.7%. Likewise, the success rate of complete enteroscopy reported by other authors ranges from 8%13 to 77.4%.12 We were not able to completely examine the small bowel in any of the patients in whom it was indicated (5 cases). In 4 of the 5 patients with both insertion routes, lesions were identified that justified the referral of the patient (and thus the aim of the procedure), and the examination in the fifth patient had to be suspended due to low blood pressure caused by bleeding. Of the 5 cases in which complete examination of the bowel could not be performed, 2 patients had identifiable lesions, and active bleeding stopped spontaneously in one other patient. Given the favorable progression of those 3 patients, the procedure, although incomplete, was considered sufficient as initial treatment, leaving capsule endoscopy to be indicated to rule out residual lesions in the unexplored areas. Unfortunately, the present study did not include long-term patient follow-up in relation to the indication of this procedure. The abovementioned reasons, together with the small size of the group of patients that had the two approaches, contributed to the null rate of complete enteroscopy in our study.
Gastrointestinal bleeding with no apparent lesion following upper endoscopy and colonoscopy has previously been called occult gastrointestinal bleeding. It is the cause of 5% of the cases of gastrointestinal bleeding. It originates in the small bowel (between the ampulla of Vater and the ileocecal valve) in 60-80% of the cases, and the remaining cases arise from lesions in the esophagus, stomach, proximal duodenum, and colon that go unnoticed in the initial endoscopic examination.6,7,15,18–21 A change in the nomenclature of this entity to small bowel bleeding in patients with normal upper and lower endoscopies before capsule endoscopy has recently been proposed. Overt SBB is that which manifests with melena or hematochezia, whereas occult SBB presents with anemia, with or without a positive fecal occult blood test. According to the current terminology, gastrointestinal bleeding of obscure origin should be those cases of gastrointestinal bleeding with no alterations in upper or lower endoscopy, capsule endoscopy, and/or enteroscopy and radiologic studies.22 We employed these new terms in our study. Today, capsule endoscopy and DBE are the main methods for studying small bowel bleeding, and they have a similar diagnostic detection rate (61 vs 55.5%, respectively).1 However, the consensus is that capsule endoscopy is the initial study of choice.1,5–7,15–24 DBE can have a primary role in this group of patients when capsule endoscopy is not available or when it is contraindicated. On the other hand, there is evidence that emergency DBE (defined as that which is performed within the first 24-72h) can also have an initial role, given that it has a high diagnostic yield of 70-90% and allows treatment during the same procedure.1 Some authors have reported determining factors in the diagnostic yield of capsule endoscopy, such as: active bleeding at the time of the study (92.3%), occult SBB (44.2%), and remitted small bowel bleeding (12.9%), as well as the time of its performance (15 days before or after the last episode of bleeding, 92 vs 34%, respectively).1,18 Diagnostic detection rates are similar for DBE in relation to the abovementioned determining factors, especially when it is performed a short time after the bleeding (up to one month after the last episode of overt bleeding).1 In our group of patients with small bowel bleeding, we had a general diagnostic yield of 68.42%. Likewise, diagnostic yield was higher (75%) in the subgroup of patients with overt SBB, than in the occult SBB subgroup (33.33%). There is improved diagnostic sensitivity of DBE when the procedure is performed with the orientation of prior video capsule endoscopy (73-93%).7,18,23 Only 4 (21%) of our study patients had a prior capsule endoscopy study that could guide the DBE (due to its high cost and low availability), and in all the cases of overt SBB, the endoscopic procedures were performed on the days following the last episode of bleeding, contributing to improving our detection rate. The most frequent lesions found were angiodysplasias in 36.8%. This concurs with previous reports that have found a higher frequency of said lesions in those patients in western countries and ulcer and/or erosions in Asian countries.5,6,11–13,18,21 GISTs represented 10.5% of our patients with small bowel bleeding and they were the second most frequently found lesion, one in the jejunum and another in the proximal ileum. In their study on DBE in mesenchymal tumors of the small bowel, He et al. reported that they were more frequent in men, and the mean patient age was 47.7 years. The majority of cases presented with manifestations of small bowel bleeding (81.8%) and abdominal pain (10.4%). The lesions generally appeared as an unsmooth superficial mass protruding into the intestinal lumen, with the presence of an ulcer or superficial erosion, and were more frequently located in the jejunum and ileum. In their study covering a 9-year period, those authors reported a lower diagnostic confirmation rate through endoscopic biopsy (12.2%) and the most common tumor was a GIST.25 Mitsui et al. described a higher confirmation rate through endoscopic biopsy (40.9%).26 The general characteristics found in our patients were similar to those in the studies described, but histologic confirmation before surgical management was possible in our 2 patients. Other less frequent lesions were: lymphangiectasia, diverticulum in the ileum with bleeding, benign ulcer of the ileum, and nonspecific ileitis (5.2% each one).
The diagnostic yield in the rest of the subgroups (except for the intermittent partial small bowel obstruction subgroup) was much lower than that of the group of patients with small bowel bleeding, with the following percentages: 50% for chronic abdominal pain, 100% for intermittent partial small bowel obstruction, and 0% for chronic diarrhea. Low yields in similar patient subgroups have also been described in other studies.5 The main complications reported in DBE are: abdominal pain (up to 20%) and acute pancreatitis (0.3%) and they increase if therapeutic interventions such as polypectomy are carried out during the enteroscopy, presenting with higher rates of bleeding and perforation (10%).7 There were 2 major complications (8.6% 2-23) in our study: a deep laceration of the mucosa after argon plasma application that was successfully treated with the placement of 2 hemoclips, and another that was inherent in the patient (low blood pressure caused by severe, active bleeding), which is within the range of foreseen complications. Age did not seem to be a risk factor for the appearance of those complications. Choi et al. found a lower DBE complication rate in patients above 65 years of age, compared with those below that age (1.7 vs 2.6%), even when the older group had a greater number of therapeutic interventions.27
A larger number of patients is required, especially in the chronic diarrhea and chronic abdominal pain subgroups, to corroborate the previously described data.
In conclusion, for the study and management of diseases arising in the small bowel, DBE is a safe and efficacious method with a high diagnostic yield and success rate. This is especially true for patients with overt SBB, in whom it could have a major role, compared with other examination methods.
Ethical responsibilitiesProtection of persons and animalsThe authors declare that no experiments were performed on humans or animals for this study.
Data confidentialityThe authors declare that they have followed the protocols of their work center in relation to the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: García-Correa JJE, Ramírez-García JJ, García-Contreras LF, Fuentes-Orozco C, Irusteta-Jiménez L, Michel-Espinoza LR, et al. Enteroscopia de doble balón: indicaciones, abordajes, eficacia diagnóstica y terapéutica y seguridad. Experiencia temprana de un solo centro. Revista de Gastroenterología de México. 2018;83:31–40.