The value of leakage testing during colorectal resections to identify anastomotic leaks or bleeding has not been established. Our aim was to compare the impact of intraoperative colonoscopy (IOC) versus insufflation with a syringe, as leakage testing in lower anterior resection (LAR) for rectal cancer, with respect to the incidence of postoperative leakage (PL).
Materials and methodsA retrospective study utilizing a prospective database of 426 patients with rectal cancer that underwent elective LAR, within the time frame of January 2015 and December 2019, was conducted. The anastomotic leak test was chosen by the surgeon. The incidence of postoperative leakage was compared between patients that underwent IOC and those that had the syringe leak test, utilizing the logistic regression analysis. Propensity score matching was included.
ResultsThere were no significant differences in the clinical characteristics or morbidity and mortality rates between the two groups. Four patients were excluded, leaving a patient total of 422. Seventy patients with IOC were compared with 352 that had the syringe leak test. The incidence of postoperative leakage was 5.7% in the IOC group and 12.2% in the control group (p = 0.001). After propensity score matching (n = 221), balancing the characteristics between the groups, the incidence of postoperative leakage was 5.7% in the IOC group and 13.9% in the syringe leak test group (p = 0.001).
ConclusionIOC was shown to be a safe method for evaluating the integrity of colorectal anastomosis and was associated with a higher percentage of protective stoma use, appearing to reduce the risk for PL.
El valor de las pruebas de filtración durante resecciones colorrectales para identificar fuga o sangrado de anastomosis no está establecido. El objetivo es comparar el impacto de la colonoscopia intraoperatoria (CIO) versus insuflación con jeringa como prueba de fuga en resección anterior baja (RAB) para cáncer de recto, con respecto a la incidencia de filtración posoperatoria (FP).
Materiales y métodosEstudio retrospectivo sobre base de datos prospectiva de 426 pacientes con cáncer de recto sometidos a RAB electiva desde enero de 2015 hasta diciembre de 2019. La prueba de fuga anastomótica se realizó a criterio del cirujano. La incidencia de FP se comparó entre pacientes sometidos a CIO y prueba con jeringa, utilizando el análisis de regresión logística, incluido análisis de emparejamiento por puntaje estimado de propensión.
ResultadosLas características clínicas y morbimortalidad no mostraron diferencias significativas entre los grupos. Total 422 pacientes, excluidos 4. Se compararon 70 pacientes CIO contra 352 prueba con jeringa. La incidencia de FP fue 5.7% en CIO y 12.2% en grupo control (p = 0.001). Después del emparejamiento por puntaje de propensión (n = 221), equilibrando las características entre los grupos, la incidencia de FP fue 5.7% en CIO y 13.9% en grupo con jeringa (p = 0.001).
ConclusiónLa CIO es un método seguro para evaluar integridad de anastomosis colorrectal y se asocia con un mayor porcentaje de estoma de protección, lo que parece disminuir el riesgo de FP.
Anastomotic leakage is the most devastating complication of intestinal resection, with colorectal anastomosis. Its general incidence reaches 30%, and not only does it significantly increase morbidity and mortality, but it also leads to poor oncologic results by increasing both local recurrence and distant metastases, as well as delaying the start of adjuvant treatment1,2. Mean hospitalization duration and healthcare costs for patients with postoperative leakage (POL) are 4 to 5-times greater than for patients with no leakage3.
Anastomotic leak tests (ALTs), one of which is intraoperative colonoscopy (IOC), have been implemented to reduce the risk of complications associated with POL4,5. In addition to evaluating the colorrectal anastomosis, IOC has been utilized to locate nonpalpable lesions. When said lesions are obstructive, IOC enables synchronous lesions to be ruled out and also makes it possible to wash the colon that is proximal to the obstruction. Some doubts have been raised as to whether IOC impedes laparoscopic manipulation and dissection because of colon distension. The air directly insufflated onto the anastomosis in IOC has a constant pressure and can later be extracted. During that direct evaluation, not only can stapling failures be identified, but bleeding and the condition of the mucosa (ischemia) can also be observed6–8, enabling primary repair and/or protective stoma creation. Several studies have shown that the procedure is safe and does not increase the rate of conversion to open surgery, surgery duration, or hospital stay7,9,10. The standard test is carried out by injecting a syringe filled with 50 cc of air or a mixture of saline solution with methylene blue or an iodized solution, via the anus.
In past decades, other ALTs have been proposed, such as the use indocyanine green (ICG), to evaluate the integrity of the blood supply to the anastomosis11. Various studies have reported promising results with ICG, including reduced POL rates12,13, but there is no consensus on its routine use.
The aim of the present study was to compare the usefulness of IOC with that of the standard intraoperative syringe leak test, in relation to enabling early treatment of a leak in an anterior resection due to rectal cancer and reducing the risk for POL.
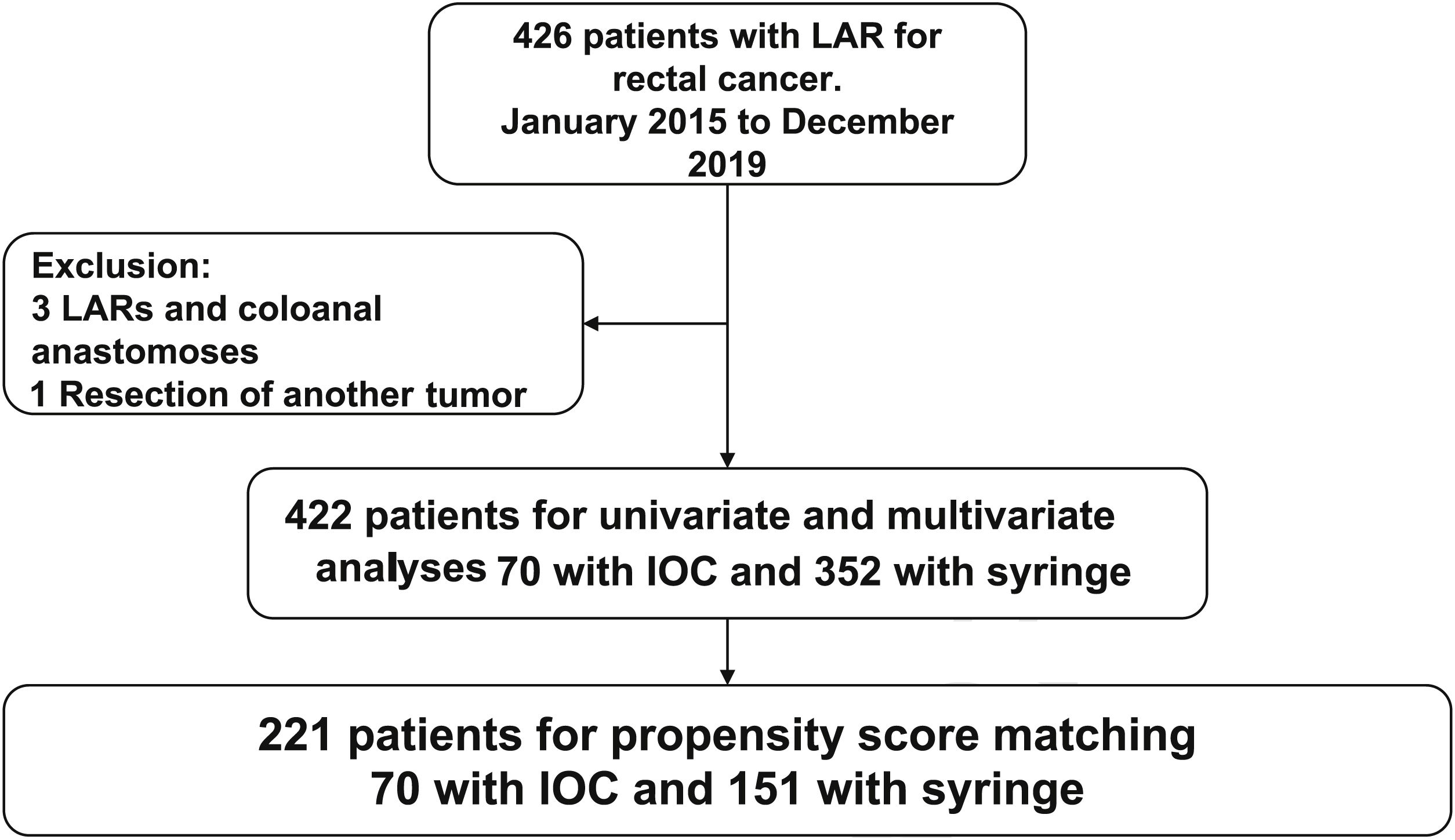
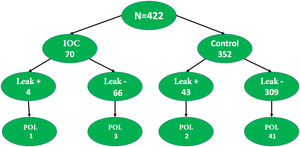
Materials and methodsPopulationA total of 426 clinical histories of patients treated for rectal cancer, within the time frame of January 2015 and December 2019, were retrieved from a prospective database from the Gastrointestinal Surgery Service of the Instituto de Cancerología Las Américas AUNA and the Clínica del Rosario in Medellín-Antioquia, Colombia, and reviewed and analyzed. Patients above 18 years of age that underwent laparoscopic lower anterior resection of the rectum and a stapled intestinal anastomosis were included. Four patients were excluded (3 that underwent coloanal anastomosis and one that had the simultaneous resection of another primary cancer), leaving a total of 422 patients enrolled in the study (Fig. 1). All the interventions were performed by a team of specialists in gastrointestinal or colorectal oncologic surgery.
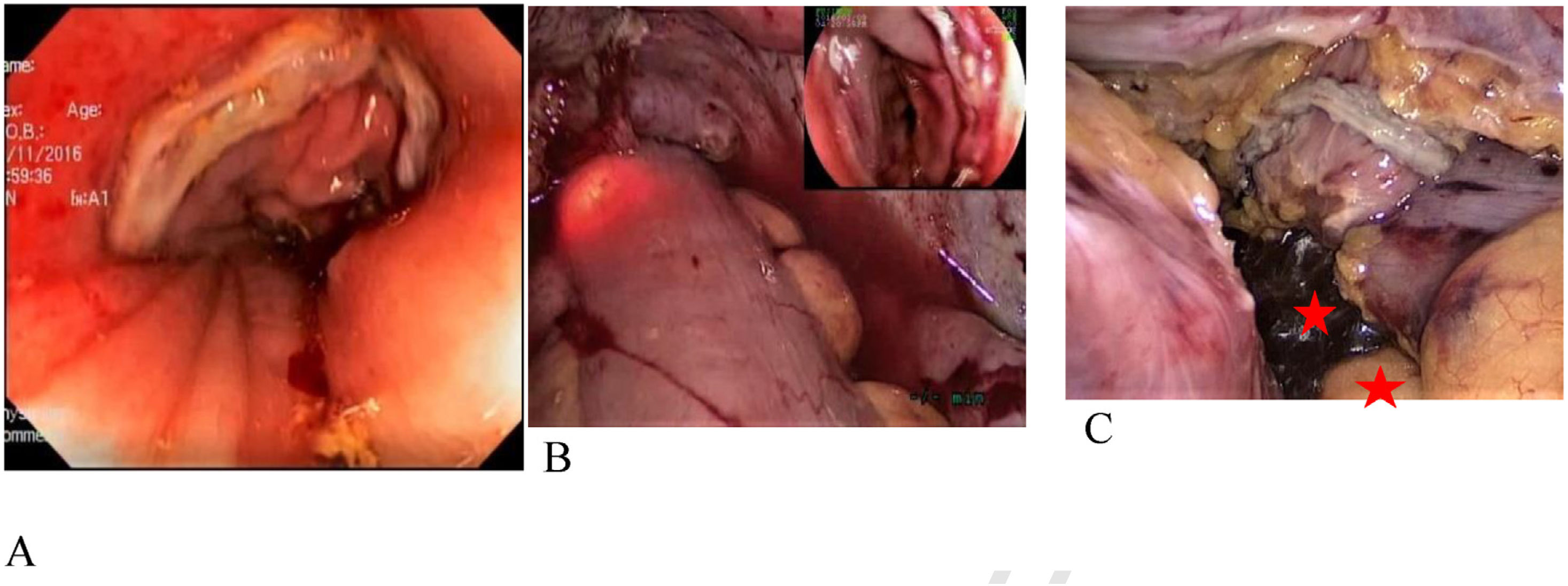
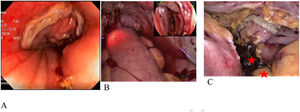
Evaluation of anastomotic integrityAfter the colorectal anastomosis, performed with a circular stapler, the proximal intestine was clamped to the anastomosis with an atraumatic clamp and the pelvic cavity was filled with warm saline solution (0.9%). At the same time, a surgeon with endoscopic training inserted the colonoscope via the anus until the anastomosis line was viewed. Sustained insufflation with the colonoscope was carried out, observing whether there was bleeding or ischemia. In the control group, 50 cc of air or a mixture of saline solution with methylene blue or an iodine solution, as determined by the surgeon, was injected with a syringe (Fig. 2). The appearance of air bubbles or the injected solution in the pelvic cavity was considered anastomotic leakage, and if present, all patients underwent protective ileostomy. Intracorporeal suturing or transnasal suturing were additionally carried out. No new anastomosis was performed in any of the cases.
Postoperative leakageThe diagnosis of POL was suspected when the patient presented with abdominal pain, fever, tachycardia, and/or the escape of fecal matter, gas, or pus from the surgical wound or through the pelvic drainage catheter, if utilized. Diagnosis was confirmed by abdominal and pelvic computed tomography with oral and/or rectal water-soluble contrast medium, rectoscopy, or surgical re-examination. Complication severity was defined according to the Clavien-Dindo extended classification14. A postoperative anastomotic leak classified as Clavien-Dindo II, or higher, within the first 30 days after the procedure, was defined as symptomatic POL.
Statistical analysisThe primary aim of the present study was to compare IOC with the standard syringe leak test for the diagnosis of intraoperative anastomotic leakage, providing early treatment that could reduce the risk for symptomatic POL, following laparoscopic colorectal surgery. Given that the incidence of POL was generally low and the assignment of the patients to either the IOC group or the syringe leak test group was not randomized, propensity score matching was utilized to adjust the possible confounding factors and ensure comparability. The propensity score was calculated using a logistic regression model. The result variable was the behavior of IOC and the plausible baseline confounding variables were included as covariates. Patient age and the distance of the anastomotic site from the anal verge were considered the continuous variables. Tumor size with logarithmic transformation was employed. The other covariates were dichotomized according to their clinically appropriate cutoff values.
The patients that underwent IOC were compared with those that underwent the syringe leak test, at a ratio of 1:2. The imbalance of the characteristics of the patients before and after the matching was examined through histograms and diagrams of the estimated propensity score (EPS) and the standardized differences. For the set of paired patients, the likelihood ratios and 95% confidence intervals (CIs) were calculated, using the univariate logistic regression (conditional) analysis. The risk ratios and their 95% CIs were calculated using unconditional models, in the same manner.
We carried out a multivariate logistic regression analysis through stepwise selection, with a 5% significance level, adjusted by the same variables as those calculated in the propensity score analyses. In addition, a simple logistic model, adjusted for the EPS, was applied. Finally, we applied stratified logistic regression models. Five post hoc strata were constructed: five equalized groups, classified according to the EPS quintiles.
All the analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC, USA) software and statistical significance was always reported as two-tailed p values.
Ethical considerationsThe protocol was carried out according to international ethics regulations and Colombian law. The present study and protocol were approved and surveilled by the Independent Ethics Committee of the Clínica Rosario Tesoro, and strictly followed the Good Clinical Practice norms.
The authors declare that this article contains no information that could identify the patients. No experiments on humans or animals were conducted, therefore the informed consent of the patients was not required.
ResultsA total of 422 patients with rectal cancer underwent elective laparoscopic lower anterior resection and intraoperative leak testing, either by IOC or the syringe leak test. The two groups of patients were comparable in terms of age, sex, body mass index, American Society of Anesthesiologists (ASA) classification, and other characteristics of the patients and tumors.
IOC was performed on 70 patients (16.4%) and the syringe leak test on 352 (83.4%) (control group). Table 1 shows the characteristics before and after the EPS adjustment. Before the EPS matching (n = 422), the IOC group had a higher frequency of smoking (p = 0.03), advanced clinical stage (p = 0.007), preoperative chemoradiotherapy (p = 0.01), and protective ostomy (p < 0.0001) than the control group. After the EPS matching (n = 221), all the adjusted covariates were well balanced, resulting in a decrease in standardized differences to less than 0.10.
Comparison of the characteristics of the total cohort (n = 422) and the paired cohort (n = 221).
| Variables | Total cohort (n = 422) | Paired cohort (n = 221) | ||||
|---|---|---|---|---|---|---|
| IOC group | Syringe leak test group (n = 352) | p | IOC group | Syringe leak test group (n = 151) | p | |
| Sex, n (%) | ||||||
| Male | 39 (54) | 185 (52.6) | 0.18 | 39 (54) | 80 (52.9) | 0.48 |
| Female | 31 (46) | 167 (47.4) | 31 (46) | 71 (47.1) | ||
| Age in years, median (IQR) | 61 (47−72) | 63 (49−74) | 0.82 | 61 (47−72) | 62 (54−66) | 0.66 |
| BMI, median (IQR) | ||||||
| > 25 | 52 (74.3) | 271 (76.9) | 0.76 | 52 (74.3) | 108 (71.5) | 0.88 |
| < 25 | 18 (25.7) | 81 (23.1) | 18 (25.7) | 43 (38.5) | ||
| ASA, n (%) | ||||||
| I | 24 (34.2) | 123 (39.4) | 0.09 | 24 (34.2) | 45 (29.8) | 0.68 |
| II–III | 46 (65.8) | 229 (60.6) | 46 (65.8) | 106 (70.2) | ||
| Stage, n (%) | ||||||
| I–II | 17 (24.3) | 82 (23.3) | 0.01 | 17 (24.3) | 31 (20.5) | 0.63 |
| III–IV | 53 (75.7) | 270 (76.7) | 53 (75.7) | 120 (79.5) | ||
| Preoperative CRT, n (%) | ||||||
| Yes | 63 (90) | 318 (90.3) | 0.35 | 63 (90) | 129 (85.4) | 0.79 |
| No | 7 (10) | 34 (9.7) | 7 (10) | 22 (14.6) | ||
| Pelvic drain use | ||||||
| Yes | 22 (31.4) | 135 (38.3) | 0.07 | 22 (31.4) | 54 (35.7) | 0.07 |
| No | 48 (68.6) | 217 (61.7) | 48 (68.6) | 97 (64.3) | ||
| Ostomy, n (%) | ||||||
| Yes | 54 (77.1) | 181 (51.4) | 0 | 54 (77.1) | 78 (51.4) | 0.52 |
| No | 16 (22.9) | 171 (48.6) | 16 (22.9) | 73 (48.6) | ||
ASA: American Society of Anesthesiologists classification; BMI: body mass index; CRT: chemoradiotherapy; IQR: interquartile range.
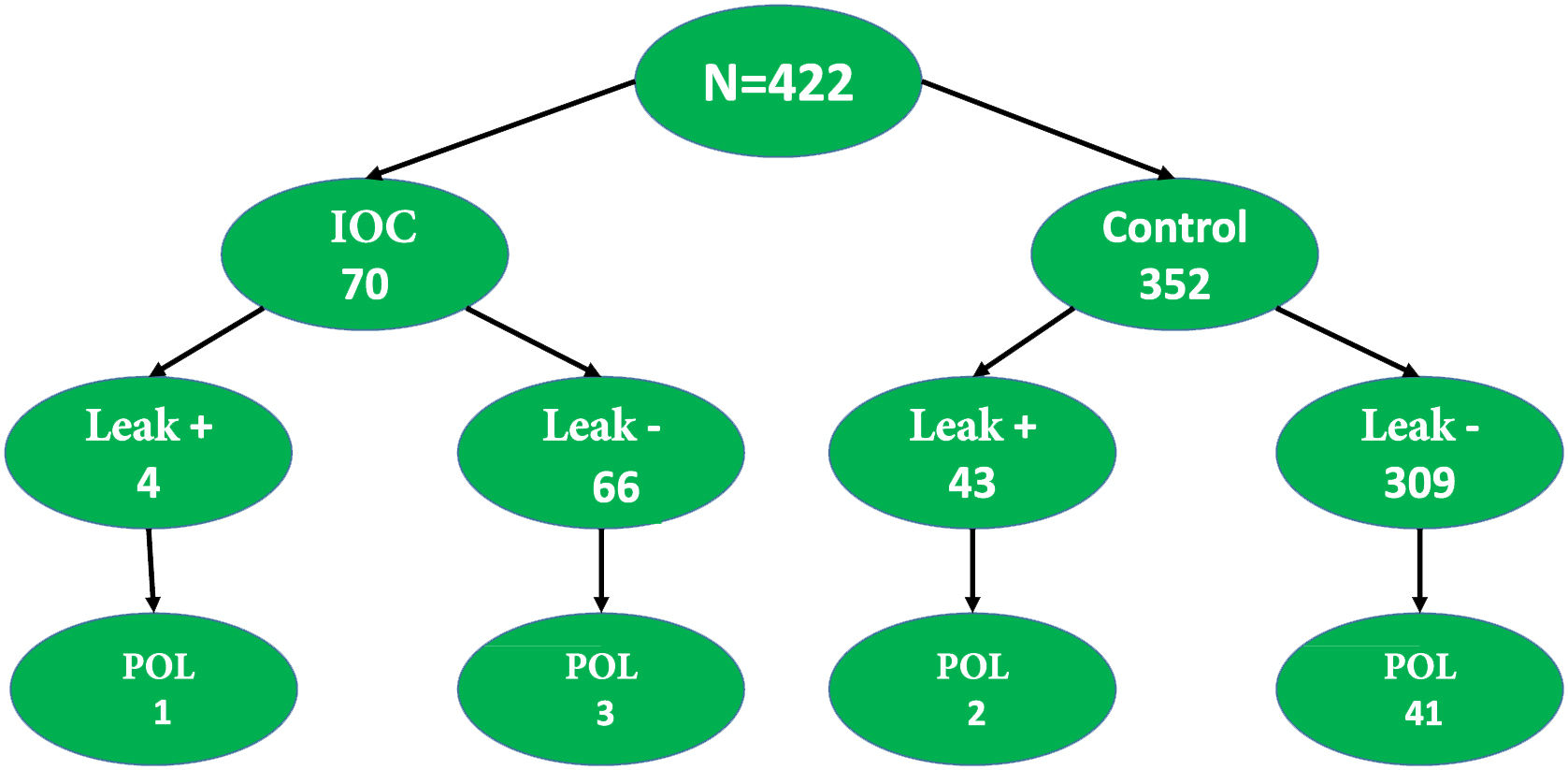
The general incidence of anastomotic leakage was 11.1% (47/422) and 11.3% (25/221) in the complete cohort and EPS-matched cohort, respectively. The incidence of POL was significantly lower in the IOC group than in the control group, in both the complete cohort (4/70, 5.7% vs. 55/352, 12.2%; p = 0.001) and the EPS-matched cohort (4/70, 5.7% vs. 21/151, 13.9%; p = 0.001). There was one death in each group.
In the IOC group, 4 patients presented with POL and the leak test had been negative during the surgery in 3 of them (3/66, 4.5%). Fig. 3 shows the POL rates in the patients that underwent IOC, compared with those that had the syringe leak test.
In the univariate logistic regression analysis, IOC significantly reduced the risk for POL (OR 0.21; 95% CI 0.075−0.57, p = 0.002). In the adjusted multivariate logistic regression analysis, without eliminating the same variables as in the EPS analysis, IOC significantly reduced the risk for POL (OR 0.17; 95% CI 0.061−0.50, p = 0.001). In the multivariate logistic regression analysis with stepwise selection, IOC significantly reduced the risk for POL (OR 0.16; 95% CI 0.057−0.45, p = 0.001). In the unconditional univariate logistic regression analysis for the cohort compatible with the EPS, IOC significantly reduced the risk for POL (OR 0.19; 95% CI 0.065−0.53, p = 0.002). In the multivariate logistic regression analysis, adjusted for EPS, IOC significantly reduced the risk for POL (OR 0.20; 95% CI 0.070−0.55, p = 0.002). Finally, in the univariate logistic regression analysis, stratified into the 5 groups constructed by the EPS, IOC significantly reduced the POL (OR 0,19; 95% CI 0.067−0.53, p = 0.002). IOC strongly and consistently reduced the risk for POL, with lower odds ratios (OR 0.16−0.23), in all the regression analyses using EPS.
Table 2 describes the clinical implications after one month of evolution, in the IOC group and the syringe leak test group, standardized with the EPS.
Postoperative progression of the patients with IOC and those with the syringe leak test, and standardized through the EPS (n = 221).
| Progression | IOC (n = 70) | Syringe leak test (n = 151) | p |
|---|---|---|---|
| Total number of leaks | 4 | 21 | 0.001 |
| Ileus | 11 | 16 | 0.234 |
| Abdominal or pelvic sepsis | 1 | 6 | 0.21 |
| New surgery | 1 | 5 | 0.43 |
| Hospital stay, days | 17 | 21 | 0.34 |
| Mortality | 1 | 1 | 0.91 |
EPS: estimated propensity score; IOC: intraoperative colonoscopy.
POL is influenced by many factors that are related to the patient, the tumor, and the surgeon. Table 3 shows the factors associated with POL.
Factors associated with postoperative leakage.
| Preoperative | Intraoperative | ||
|---|---|---|---|
| Unmodifiable | Modifiable | High-risk | Protective |
| Sex | Smoking | Contamination | Intravenous antibiotics |
| Distal anastomosis | Obesity | Surgery longer than 4 h | Heart monitorization |
| Tumor > 3 cm | Alcoholism | Inotropes | Surgical technique |
| Stage > 2 | Steroids | Blood loss | Stapling |
| Metastasis | Bevacizumab | Transfusion | Leak test |
| Radiotherapy | Infliximab | Tumor height | Ostomy |
| ASA > 2 | Tacrolimus | ||
| Diabetes | Everolimus | ||
| COPD/ex-smoker | Azathioprine | ||
| Vascular disease | Mycophenolate | ||
| Emergency surgery | Malnutrition | ||
| Kidney disease | |||
ASA: American Society of Anesthesiologists classification; COPD: chronic obstructive pulmonary disease.
Source: modified from McDermott et al.15.
Anastomotic leak testing is thought to be a factor that reduces the risk for POL because it enables the early performance of an intervention to correct a leak and/or protect against one.
Gilbert and Trapnell16 described the intrarectal instillation of saline solution as a leak test, after an intestinal anastomosis, using a distension pressure of 25 cm of water. Leaks found in 24% of the patients were oversewn and only one patient developed POL. Those authors concluded that it was a good method, given that, compared with air insufflation, the leakage site could be seen, and the leak treated. The test is considered simple and effective17, and some studies even suggest that a protective stoma can be safely omitted, in patients with a negative leak test18. In leak tests with water or gas, the instillation pressure must be controlled because excess pressure can cause anastomotic disruption.
The combination of saline solution and methylene blue has also been used to evaluate anastomotic integrity, by identifying the leakage site through the passage of the dye. In a case series of 229 patients that underwent the ALT with methylene blue, it was positive in 7% of the cases and all leaks were corrected through oversewing or redoing the anastomosis. Those authors feel that methylene blue spillage can be easier to detect, compared with leak tests with air/water19. In a recent study, Yang et al.20 compared the performance of IOC with the syringe leak test in 430 patients that underwent surgery for rectal cancer. They found that the POL rate was 11.7% in the group with negative syringe leak tests, compared with 4.3% in the group with negative IOC, suggesting that IOC performs better than the other anastomotic leak tests. The creation of a protective stoma after a positive leak test was significantly higher in the IOC group than in the group without IOC (4.65 vs. 0.93%). Other authors question the effectiveness of leak tests for reducing the risk for POL. Wu et al.21 reported that the performance of an intraoperative leak test did not reduce the incidence of POL.
In the present case series, an ALT was routinely performed in all patients that underwent lower anterior resection of the rectum, but the choice of one type of leak test over another depended on the preference or training of each surgeon (oncologist, gastrointestinal surgeon, or coloproctologist). The IOC group and the syringe leak test group were compared in terms of the impact on the incidence of POL, the incidence of immediate and late bleeding, and the need for a protective stoma. An attempt was made to clarify whether the intraoperative ALT through IOC was a more reliable method for detecting technical defects in the anastomosis, compared with the conventional syringe method, and consequently, for reducing the risk of POL. There were interesting results regarding the POL rate between the two methods employed. The POL rate was lower in the IOC group, but the incidence of protective stoma creation was higher. On the premise that there was no difference in the surgical technique of the anastomoses between the two groups, the higher positive intraoperative leak test in the IOC group could indicate that leak assessment through IOC is a more efficacious method for detecting small defects in the anastomosis.
One patient with a negative leak assessment through IOC presented with POL. POL can always be an outcome, given that surgical technique is an important factor for leakage, and other factors, such as comorbidities, nutritional status, and vascular alterations, among other characteristics of the patient, are involved in POL.
There is concern as to the safety and potential complications related to IOC. It is speculated that IOC can damage the anastomosis due to elevated intraluminal pressure, but the procedure is considered safe, given that studies have shown that the maximum pressure in the neorectum, after resection and the colorectal anastomosis, can reach up to 200 mmHg. The maximum air pressure produced by the power source of the colonoscope varies from 300 to 375 mmHg, and the air pressure at the tip of the colonoscope ranges from 72 to 166 mmHg. The latter value is lower than the former due to the leakage of air through the channel and the connection with the light source22,23. Kozarek and Sanowski22 demonstrated that the mean sustained intraluminal air pressure during diagnostic colonoscopy was 22 mmHg (range 9–57 mmHg). IOC should be performed by experienced trained surgical endoscopists.
Previous studies have been conducted on the efficacy of fluorescence imaging with ICG for evaluating perfusion in the anastomosis13. In our patients, ischemia may have been the main cause of postoperative anastomotic leak because, of the 66 patients with negative leak tests through IOC, 3 presented with POL. Intraoperative evaluation of the mechanical integrity of IOC, together with ICG, could greatly contribute to a reduced incidence of anastomotic leakage.
Limitations of the present study were its retrospective, nonrandomized design, the small number of patients in the IOC group, and the fact that the decision to perform a leak test using IOC depended on the preference of the surgeon. Nevertheless, the study provides useful information and paves the way for larger, randomized, controlled studies to be conducted that produce stronger evidence of the usefulness of IOC, enabling its recommendation. The learning curve involved in performing the procedure and the higher cost of the surgery should also be kept in mind.
We believe that the ALT, using IOC, is a valid option in cases in which intraoperative colonoscopy is considered necessary for an additional reason or at centers in which said technique does not produce an increase in costs.
In conclusion, the evaluation of anastomoses through IOC is a safe technique that is associated with a significant reduction in POL in patients undergoing surgery for rectal cancer. It could also be secondary to the creation of more protective stomas and the eventual use of pelvic cavity drains. In addition to providing useful information, the direct visualization of the anastomosis through colonoscopy can aid in the performance of intraoperative treatment that is necessary for preventing POL. The results of the present article suggest the need for more methodologically rigorous studies on the identification and intraoperative treatment of anastomotic leakage and its clinical impact, leading to better patient outcome.
Conflict of interestThe authors declare that there is no conflict of interest.
Financial disclosureThe present study was carried out with the support of the Vicerrectoría de Investigación sustainability project of the Universidad de Antioquia, Medellín, Colombia.
The authors wish to thank the biostatistician and epidemiologist, Víctor Calvo, for his collaboration.
Please cite this article as: Castaño R, Molina-Meneses SP, Puerta JD, Palacios LJ, Jaramillo R, Piñeres A, et al. Impacto de la colonoscopia intraoperatoria en la detección y prevención de la fuga de la anastomosis colorrectal por cáncer de recto. Rev Gastroenterol Méx. 2022;87:312–319.