Primary eosinophilic colitis (PEC) is an inflammatory disease caused by the infiltration of eosinophils into the mucosa of the colon, determined after having ruled out other possible causes. It is characterized by abdominal pain and diarrhea and its prevalence is unknown.
AimsTo know the prevalence of PEC in patients with chronic diarrhea and in those with clinical criteria for diarrhea-predominant irritable bowel syndrome (IBS-D).
Patients and methodsAll patients with chronic noninflammatory diarrhea that were consecutively seen over a decade were studied through colonoscopy, with systematic biopsy. Patients with a known or decompensated organic disease, alarm features, abnormal minimal laboratory test results, and factors associated with eosinophilia were excluded. Patients with the clinical criteria for IBS-D (Rome III) were selected from the general group. Eosinophilic colitis was defined as the presence of > 35 eosinophils/100 epithelial cells per field in any of the segments.
ResultsFrom the 683 patients seen, a total of 545 patients (361 women) were included in the study. Twenty-two cases of PEC were detected, resulting in a prevalence of 4.0%. A total of 343 patients had IBS-D criteria (223 women), with 16 cases of PEC detected, for a prevalence of 4.7%. Colonoscopy was normal, or almost normal, in all the PEC cases detected.
ConclusionsThe prevalence of PEC was 4.0% in the patients with chronic diarrhea and 4.7% in the patients with IBS-D. Peripheral eosinophilia appeared to be the main distinctive clinical characteristic, but systematic biopsy was essential for making the diagnosis.
La colitis eosinofílica primaria (CEoP) es una enfermedad inflamatoria causada por infiltración de eosinófilos en la mucosa del colon, sin otras posibles causas identificadas, que se caracteriza por dolor abdominal y diarrea. Su prevalencia se desconoce.
ObjetivosConocer la prevalencia de CEoP en pacientes con diarrea crónica y en aquellos con criterios clínicos del síndrome del intestino irritable con predominio de diarrea (SII-D).
Pacientes y métodosTodos los pacientes con diarrea crónica no inflamatoria, atendidos en forma consecutiva durante una década, fueron estudiados mediante colonoscopia con toma sistemática de biopsias. Se excluyeron aquellos con enfermedad orgánica conocida o descompensada, con datos de alarma, estudios de investigación limitada anormales y factores asociados a eosinofilia. Del grupo general, se seleccionaron aquellos con criterios clínicos de SII-D (Roma III). Colitis eosinofílica se definió como la presencia de > 35 eosinófilos/100 células epiteliales por campo en cualquiera de los segmentos.
ResultadosFueron atendidos 683 pacientes. Se incluyeron 545 enfermos (361 mujeres), entrequienes se detectaron 22 casos de CEoP, para una prevalencia del 4.0%. Trescientos cuarentay tres sujetos tenían criterios de SII-D (223 mujeres), entre los que se detectaron 16 casos deCEoP, para una prevalencia del 4.7%. La colonoscopia fue normal o casi normal en todos loscasos detectados.
ConclusionesLa prevalencia de CEoP fue de 4.0% de los enfermos con diarrea crónica y en 4.7% en aquellos con SII-D. La eosinofilia periférica parece ser la principal característica clínica distintiva, pero la toma sistemática de biopsias es indispensable para establecer el diagnóstico.
The presence of eosinophils in the different segments of the digestive tract is common and seen in diverse clinical scenarios. Eosinophilic gastrointestinal diseases (EGIDs) are a group of disorders characterized by symptoms of gastrointestinal dysfunction, caused by the inflammatory infiltration of eosinophils and include eosinophilic esophagitis, eosinophilic gastroenteritis, and eosinophilic colitis.1,2 Primary eosinophilic colitis (PEC) is defined as an inflammatory disease of unknown etiology that mainly manifests as abdominal pain and diarrhea, segmental or diffuse infiltration of eosinophils in the mucosa of the colon, for which other possible causes have been ruled out.3,4 The usual endoscopic findings tend to be nonspecific, and even though PEC is considered a rare disease, its prevalence is unknown.
Chronic diarrhea is a frequent illness in the general population and tends to be a diagnostic challenge.5 Evaluation of the colonic mucosa is very helpful in detecting inflammatory bowel disease, ischemic colitis, microscopic colitis, neoplasias, and intraepithelial infections. The endoscopic appearance of the colon is normal, or almost normal, in some of those diseases, and thus they can go by undetected. Different studies have shown that systematic endoscopic biopsies of the colon provide good diagnostic yield in patients with chronic noninflammatory diarrhea and normal colonoscopy.6–10 That conduct is recommended in specific groups of patients and in selected patients, such as those with diarrhea-predominant irritable bowel syndrome (IBS-D).11–13
The aims of the present study were to know the prevalence of PEC in patients with chronic diarrhea, for which no other causes were identified, and to determine its prevalence in patients with the clinical criteria for IBS-D.
Materials and methodsAll the patients consecutively seen in the outpatient consultation of the lead researcher (RCS), within the time frame of July 2010 and January 2020, were considered for participation in the study.
All patients above 18 years of age, that presented with chronic noninflammatory diarrhea and accepted to undergo colonoscopy with biopsy, were included. Chronic diarrhea was defined as the presence of bowel movements with decreased stool consistency and increased frequency that lasted for more than 4 weeks, as well as no abnormalities in minimal laboratory test results.7,8
Patients with a known or compensated organic disease associated with diarrhea, alarm features (e.g., involuntary weight loss > 10% of the baseline weight, within the past 6 months, anemia, clinical signs of gastrointestinal bleeding or malabsorption), cancer, a history of organ transplantation, any type of colorectal surgery, HIV-AIDS infection, patients under regular treatment with clozapine, carbamazepine, rifampin, tacrolimus, gold salts, or nonsteroidal anti-inflammatory drugs, for 6 months prior to disease onset, patients contraindicated for colonoscopy or biopsy, and patients with incomplete colonoscopy or inadequate bowel cleanliness at the time of the study (Boston Bowel Preparation Scale score equal to or less than 6) were excluded.14
All the patients had a uniform clinical history, filled out a questionnaire to determine their healthcare resource use during the year prior to their first visit, underwent a specific set of laboratory tests (complete blood count, erythrocyte sedimentation rate or C-reactive protein or both, thyroid-stimulating hormone, the search for pathogenic parasites in stools, fecal occult blood, and fecal cytology, or fecal calprotectin and lactoferrin), underwent colonoscopy with random biopsies of all the colonic segments (at least 8 biopsies per study, always including the right and left colon), even in the presence of normal-appearing mucosa, and had biopsies of the segments with minimal alterations (areas of erythema, with hemorrhagic spots, loss of vascular pattern, and a nodular appearance, among others). Colonoscopy was considered normal when the surface of the intestinal mucosa was smooth, regular, and pink, with a visible fine vascular pattern, and the presence of folds in all segments. The presence of erythematous areas, loss of vascular pattern, and subepithelial segmental hemorrhagic spots were considered minimal alterations in the colonoscopy. Biopsies were stained using hematoxylin & eosin and were independently viewed by 2 pathologists that were unaware of the clinical characteristics of the patients.
The Rome III clinical criteria were utilized to determine the prevalence of PEC in patients with IBS, selecting the patients with the IBS-D criteria.15 All the patients with Rome criteria different from those of IBS-D, or with insufficient criteria for their classification, were eliminated from that sub-analysis.
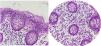
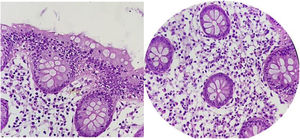
For the purpose of our study, eosinophilic colitis was histopathologically defined as the presence of more than 35 eosinophils/100 epithelial cells per field in any of the segments (Fig. 1).
Histologic slides showing the colonic mucosa. Abundant mixed inflammatory cells can be seen in the lamina propria, as well as in the epithelium of the glands and crypts, with an important quantity of eosinophils (up to 50 per high power field), consistent with eosinophilic colitis (courtesy of Dr. Claudia Peña Zepeda).
The data were captured and analyzed using the Statview (BrainPower Inc. Calabazas, CA) program. The descriptive data were expressed through percentages, means, and ranges. The chi-square test, Fisher’s exact test, and Student’s t test were employed for the statistical analysis. Odds ratio (OR) was calculated with 95% confidence intervals (CIs), using the EBM Calc application for iOs (Foundation Internet Services, LLC). All values of p > 0.05 were considered not significant (p = NS).
The present work was carried out, following the international ethics norms and the Declaration of Helsinki. Patient anonymity was maintained at all times and no identifying data appeared in the manuscript. All patients provided written statements of informed consent to participate in the study. The present research is the analysis of a case series that included the review of conventional diagnostic procedure results, in which no experimental interventions were performed, and therefore was not reviewed by a research committee.
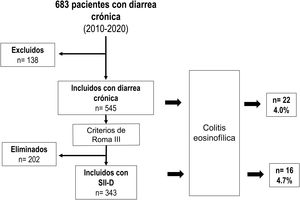
ResultsA total of 683 patients with chronic diarrhea were seen within the time frame of the present study, and 138 of them were excluded: 102 due to known or decompensated organic diseases associated with diarrhea, 14 due to incomplete colonoscopy, 13 due to alarm features, 4 due to colorectal cancer, 4 due to a history of colorectal surgery with resection of a segment, and one due to inadequate colon cleansing.
The prevalence of PEC was determined from the remaining 545 patients that were included in the study, of which 361 were women and 184 were men (66 and 34%, respectively), with a mean age of 54.8 years (range: 18 to 89 years). Twenty-two cases of PEC were detected, resulting in a prevalence of 4.0%.
The analysis of clinical characteristics showed that the patients with chronic diarrhea and PEC had abdominal pain, had received treatment for allergic conditions over the past year (e.g., asthma, rhinitis, or dermatitis), and had a greater frequency of presenting with peripheral eosinophilia. None of the cases had a cell count above 1,000 eosinophils in peripheral blood. Those characteristics are shown in Table 1. Thus, the joint presence of abdominal pain, a history of treatment for allergic conditions within the past year, and peripheral eosinophilia was associated with a greater risk for PEC in patients with chronic diarrhea (OR 3.36, 95% CI 1.18-9.58).
Clinical characteristics of the patients with chronic diarrhea, with and without eosinophilic colitis.
| Clinical characteristics | With PEC | Without PEC | p value |
|---|---|---|---|
| n (%) | n (%) | ||
| 22 (100) | 523 (100) | ||
| Women | 13 (59) | 348 (67) | NS |
| Under 50 years of age | 10 (45) | 268 (51) | NS |
| Pain | 22 (100) | 403 (77) | 0.006 |
| Bowel urgency | 17 (77) | 345 (66) | NS |
| Occasional fecal incontinence | 5 (23) | 84 (16) | NS |
| Nocturnal bowel movements | 3 (14) | 50 (10) | NS |
| Weight loss | 4 (18) | 94 (18) | NS |
| History of allergiesa | 8 (36) | 115 (22) | NS |
| Use of treatments prescribed for allergic conditions (e.g., asthma, rhinitis, or dermatitis) within the past yearb | 5 (23) | 45 (9) | 0.04 |
| Peripheral eosinophilia | 6 (27) | 42 (8) | 0.001 |
NS: not significant; PEC: primary eosinophilic colitis.
To determine the prevalence of PEC in patients with IBS-D, the Rome III criteria were applied to all the patients with chronic diarrhea (n = 545), eliminating 202 of them: 185 due to presenting with mixed IBS (IBS-M) and 17 for having unspecified IBS criteria (IBS-U). The resulting subgroup of 343 patients with IBS-D was made up of 223 women and 120 men (65 and 35%, respectively), with a mean age of 55.6 years (range: 18 to 78 years). Sixteen cases of PEC were detected, resulting in a prevalence of 4.7%.
In the clinical characteristic analysis, only peripheral eosinophilia was significantly higher in the patients with IBS-D and PEC, as shown in Table 2. Thus, the presence of peripheral eosinophilia was associated with a greater risk for presenting with PEC in patients with IBS-D (OR 3.52, 95% CI 1.29-9.61).
Clinical characteristics of the patients with the clinical criteria for diarrhea-predominant irritable bowel syndrome (IBS-D), with and without eosinophilic colitis.
| Clinical characteristics | With PEC | Without PEC | p value |
|---|---|---|---|
| n (%) | n (%) | ||
| 22 (100) | 321 (100) | ||
| Female sex | 13 (59) | 223 (69) | NS |
| Under 50 years of age | 10 (45) | 178 (55) | NS |
| Pain | 22 (100) | 321 (100) | NS |
| Bowel urgency | 17 (77) | 216 (67) | NS |
| Occasional fecal incontinence | 5 (23) | 48 (15) | NS |
| Nocturnal bowel movements | 3 (14) | 25 (8) | NS |
| Weight loss | 4 (18) | 68 (21) | NS |
| History of allergiesa | 8 (36) | 92 (29) | NS |
| Use of treatments prescribed for allergic conditions (e.g., asthma, rhinitis, or dermatitis) within the past yearb | 5 (23) | 37 (12) | NS |
| Peripheral eosinophilia | 6 (27) | 33 (10) | 0.02 |
NS: not significant; PEC: primary eosinophilic colitis.
Fig. 2 describes the distribution of all the included, excluded, and eliminated patients considered for participation in the study.
There were no significant differences in the colonoscopy findings between the study groups. Colonoscopy was normal in 387 patients with chronic diarrhea, 233 patients with IBS-D, and 14 patients with PEC (71, 68, and 64%, respectively, p = NS). Colonoscopy identified minimal segmental alterations (e.g., erythema or subepithelial hemorrhagic spots) in 158 patients with chronic diarrhea, 110 patients with IBS-D, and 8 patients with PEC (29, 32, and 36%, p = NS). No erosions, ulcers, tumor lesions, or strictures were found in any of the cases.
Sixteen of the 22 cases (73%) were treated with 9 mg/day of budesonide for at least 8 weeks, and at present, symptoms are resolved in all those patients.
Discussion and conclusionsIn the present study conducted on a selected group of patients with chronic diarrhea, the prevalence of PEC was 4.0%, and in patients with the clinical criteria for IBS-D, said prevalence was 4.7%. The endoscopic appearance of the colon was normal, or almost normal, in those patients, making systematic biopsy indispensable.
Eosinophilic gastrointestinal diseases (EGIDs) are a group of disorders characterized by symptoms of gastrointestinal dysfunction, caused by the inflammatory infiltration of eosinophils that more frequently affects the gastrointestinal mucosa, but can include muscle layers and the serosa. It is usually limited to specific organs.1,16 EGIDs include eosinophilic esophagitis, eosinophilic gastroenteritis, and eosinophilic colitis.2 PEC is defined as an inflammatory condition that mainly manifests as abdominal pain and diarrhea, characterized by a diffuse or segmental infiltration of eosinophils into the mucosa of the colon, for which other possible causes have been ruled out.3,4
Importantly, colonic eosinophilia is not the same as eosinophilic colitis. The detection of eosinophils in different gastrointestinal segments is common. It is often a sequel of acute inflammation and its presence does not necessarily indicate a specific disease.17 The presence of eosinophils in the lamina propria of the colon is a normal finding. There is a tendency toward eosinophilia in allergic disorders, but it can also be induced by drugs, parasitic infections, or autoimmune diseases. Eosinophils are part of hypereosinophilic syndrome, they are seen in inflammatory bowel disease and malignant hematologic neoplasias, and they are part of the histopathologic spectrum of gluten-induced enteropathy, as well as other entities associated with intestinal lymphoma.16 Likewise, the finding of eosinophils in routine biopsies, without considering the possible causes, or when there is no close association with clinical symptoms, has no importance. In contrast, the detection of an important quantity of eosinophilic infiltrates in the colonic mucosa, in the context of a patient with chronic diarrhea, in whom other possible causes have been reasonably ruled out, is diagnostic for PEC.3,4
However, there is no universal agreement for designating the normal number of eosinophils in biopsies of the colon. Diagnostic criteria have been established by consensus for some EGIDs, but not for PEC. At present there are no accepted criteria for distinguishing the upper limit of normal in the pathologic increase of those cells.17 The broadest accepted definition is an eosinophil count above 20-35/100 epithelial cells per field, assuming that the involvement is segmental and not uniform.2,18–25
PEC is considered a rare disease. Prevalence in the United States has been calculated at 2-3/100,000 inhabitants,26,27 but its identification has been on the rise in recent years.28 Eosinophilic colitis has been diagnosed from a small number of biopsies taken in patients undergoing colonoscopy for the study of diarrhea, but prevalence varies widely.17 In Mexico, 2 previously published studies showed a prevalence of eosinophilic colitis of 1 and 7%, in patients with chronic diarrhea and normal, or almost normal, endoscopy.7,10
Nothing is known about the prevalence of eosinophilic colitis in patients with clinical criteria for IBS-D, a group that should be carefully studied. At least 2 studies in Mexico have found an elevated prevalence of microscopic colitis in patients with clinical criteria for IBS and IBS-D that had systematic biopsy of the colon.7,29 The search for microscopic colitis should be carried out in those patients through biopsies of all the segments of the colon, even in the presence of normal mucosa and no evidence of macroscopic lesions.12 Our study showed that almost 5% of the patients with IBS-D criteria had PEC. The endoscopic appearance of the mucosa was normal in all of those patients and the diagnosis would not have been made, if it had not been intentionally sought.
The joint presence of abdominal pain, a history of allergy treatment in the past year, and peripheral eosinophilia that resulted from the analysis of clinical characteristics of the different groups included in the present study, suggests they are factors associated with the presence of eosinophilic colitis in patients with chronic diarrhea. Nevertheless, only peripheral eosinophilia persisted as a risk factor in patients with IBS-D. Unfortunately, our study design and the small number of cases detected with PEC did not enable us to draw clear conclusions. In addition, no endoscopic pattern distinctive of the disease was found. Thus, in the absence of clinical or endoscopic disease markers, systematic biopsy is indispensable for diagnosing PEC, even in the presence of a normal colon.
The present study showed the prevalence of PEC in a large case series of patients that were uniformly selected and analyzed. Possible factors associated with eosinophilia were eliminated, colonoscopies were performed by the same operator, and biopsies were taken of all segments of the colon, regardless of the appearance of the mucosa, and interpreted by 2 pathologists utilizing the same histopathologic criteria. Those characteristics increased the strength of our study, despite the methodological defects inherent in its design. There could have been selection bias, given that the patients were consecutively recruited, in accordance with their acceptance to undergo the proposed studies. Even though biopsies were taken from all the colonic segments in all the patients, no analysis differentiating the regions was carried out, despite the fact that the density of eosinophils can vary from one segment to another. There is very little information on biopsies from a truly normal colon, but the cutoff point utilized in our operational definition (35 eosinophils per every 100 epithelial cells/field) was higher than that defined by the majority of authors as normal for any of the segments of the colon.30,31
The main contribution of our study is that it highlighted the existence of PEC in a small number of patients, whose diagnosis could have been overlooked, had they not been systematically and intentionally studied. Our results reinforce the concept that patients with clinical criteria for IBS-D should be studied through colonoscopy and biopsy, even in the presence of normal mucosa. It is important for the physician to consider PEC in the differential diagnosis as an increasingly recognized entity.26–28
We believe the development of a multidisciplinary consensus that clearly establishes the diagnostic criteria for PEC, along with the proposal of a uniform diagnostic algorithm that will enable us to recognize said entity in specific groups of patients and eventually offer effective treatment regimens, are necessary. At present, the physician must have that diagnostic possibility in mind and carry out an intentional search for it, with the collaboration of a pathologist motivated to look for the disease, as well.
We conclude that PEC was found in 4.0% of the patients with chronic diarrhea and 4.7% of the patients with the clinical criteria for IBS-D. Even though peripheral eosinophilia was apparently the main distinctive clinical characteristic in those patients, systematic biopsy was indispensable for making the diagnosis, given that the endoscopic appearance of the colon was normal, or almost normal, in those cases.
Financial disclosureNo financial support was received in relation to this study/article.
No specific grants were received from public sector agencies, the business sector, or non-profit organizations in relation to this study.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Carmona-Sánchez R, Carrera-Álvarez MA, Peña-Zepeda C. Prevalencia de colitis eosinofílica primaria en pacientes con diarrea crónica y síndrome de intestino irritable con predominio de diarrea. Rev Gastroenterol México. 2022;87:135–141.