Percutaneous liver biopsy with histopathologic analysis is a valuable tool for the diagnosis, prognosis, and treatment evaluation of liver diseases. Its ultrasound-guided performance is useful, making the procedure safer and reducing the risk for complications and hospital stay. Our aim was to describe the indications, histopathologic study, and complications associated with the performance of ultrasound-guided percutaneous liver biopsy in pediatric patients.
Material and methodsThe study included 102 ultrasound-guided percutaneous liver biopsies performed on patients <16 years of age, within the time frame of January 2014 and December 2019. The information was obtained from electronic files and histopathologic studies and the data were analyzed through descriptive statistics.
ResultsA total of 102 procedures were carried out on 99 patients. Mean patient age was 72 months and 58.8% of the patients were female. Over 65% of the indications for liver biopsy included autoimmune hepatitis (23.5%), elevated liver enzymes (21.5%), and chronic liver disease (20.5%). Four patients presented with immediate complications (3.9%), three of which were major (2.9%), concurring with that reported in the international literature.
ConclusionsOur study corroborates the importance of ultrasound-guided liver biopsy in the diagnosis and follow-up of pediatric patients. The procedure also had a low complication rate of only 3.9%.
La biopsia hepática percutánea con análisis histopatológico es una herramienta valiosa para el diagnóstico, pronóstico y evaluación del tratamiento en las enfermedades hepáticas. Su realización guiada por ecografía es útil, pues permite realizar el procedimiento de manera más segura, disminuyendo el riesgo de complicaciones y estancia hospitalaria. El objetivo es describir las indicaciones, el reporte histopatológico y complicaciones asociadas a la realización de biopsia hepática percutánea guiada por ultrasonido en pacientes pediátricos.
Material y métodosSe incluyeron 102 procedimientos en pacientes <16 años a los cuales se les realizó biopsia hepática percutánea guiada por ultrasonido de enero 2014 a diciembre 2019. La información se obtuvo del archivo electrónico y reporte histopatológico. Los datos se analizaron mediante estadística descriptiva.
ResultadosSe realizaron 102 procedimientos en 99 pacientes, el 58.8% correspondiente al sexo femenino, con una mediana de edad de 72 meses. La hepatitis autoinmune (23.5%), transaminasemia (21.5%) y hepatopatía crónica (20.5%), representaron más del 65% de las indicaciones para la toma de biopsia hepática. Cuatro pacientes presentaron complicaciones inmediatas (3.9%), tres de ellas fueron mayores (2.9%), lo que concuerda con lo reportado en la literatura internacional.
ConclusionesNuestro estudio corrobora la importancia de la biopsia hepática percutánea guiada por ecografía en el diagnóstico y seguimiento en la población pediátrica, además de demostrar que es un procedimiento con baja tasa de complicaciones, observándose únicamente en el 3.9% de los pacientes.
Ultrasound-guided percutaneous liver biopsy (PLB) in pediatrics has gained great relevance over time as a safe method of specimen collection for histopathologic analysis in patients with acute and chronic liver diseases, providing diagnostic and prognostic data and treatment evaluation information.1–3
In recent years, the American Association for the Study of Liver Diseases (AASLD)4 and the European Society for Paediatric Gastroenterology Hepatology and Nutrition (ESPGHAN)5 have published recommendations for taking liver biopsies in the adult and pediatric populations that include indications, pre-procedure care, suggested technique, and specimen care for proper analysis. Ultrasound support enables the procedure to be performed more safely,3,5 reducing the risk of complications, and can be carried out in real time or through preoperative site marking.5
The ESPGHAN divides complications related to liver biopsy into 2 groups. Minor complications are grouped as those that do not require transfusion, prolonged hospitalization, surgical management, or hospitalization in the intensive care unit and major complications are those described above and/or hemothorax, pneumothorax, and death.5
At present there are few studies that evaluate the experience of PLB in pediatric patients in Mexico and Latin America. The aim of the present study was to describe the main indications, histopathologic report, and complications associated with the performance of ultrasound-guided PLB in pediatric patients at our hospital center.
Materials and methodsA retrospective, descriptive, cross-sectional study was conducted. The electronic medical records of pediatric patients under 16 years of age, seen at the Pediatric Gastroenterology and Nutrition Service of the Centro Médico Nacional de Occidente (CMNO), that underwent ultrasound-guided PLB under sedation, within the time frame of January 2014 and December 2019, were reviewed.
The following protocol is carried out at our hospital for taking PLBs: statements of informed consent are signed by the parent or guardian, preoperative laboratory exams that include complete blood count, partial thromboplastin time (PTT), prothrombin time (PT), international normalized ratio (INR), aspartate aminotransferase (AST), alanine aminotransferase (ALT), albumin, and total bilirubin, are carried out 24 h prior to the procedure. If the INR is ≥ 1.5 or the platelet count is ≤ 60 × 109/l, frozen fresh plasma (10 ml/kg) or platelet concentrate (one concentrate/10 kg of weight) is administered prior to the puncture, to prevent bleeding. After the patient has fasted 6 to 8 h, depending on age, the PLB is performed in the radiology service by an interventional radiologist, aided by an anesthesiologist. An initial ultrasound scan identifies the puncture site, asepsis is carried out, and local anesthesia with lidocaine 1% is administered. The biopsy is taken through the intercostal or subcostal approach, with a cutting needle and real-time ultrasound scan. The puncture site is compressed for 5 min, after which another ultrasound scan is performed, in an intentional search for bleeding.
All patients were routinely given nonsteroidal anti-inflammatory drugs as analgesia. A new ultrasound scan was carried out 24 h after the PLB. The biopsy specimens were analyzed by pathologists experienced in gastrointestinal and liver diseases.
Non-probability convenience sampling of consecutive cases was carried out and all the patients that met the inclusion criteria were included in the study. The inclusion criteria were case records of pediatric patients who underwent PLB within the study period. Exclusion criteria were case records with incomplete information.
Demographic data (age, sex, associated comorbidities), indications for biopsy, laboratory exam results prior to the procedure (hemoglobin, hematocrit, platelet count, PT, PTT, INR), major and minor complications (pain at the puncture site or the right shoulder, infection, bleeding, pneumothorax, hemothorax, and death), and the histopathologic report were collected and entered as variables into a structured database.
Statistical analysisA descriptive analysis of the population was carried out. The Kolmogorov-Smirnov test was initially used to analyze the distribution curve of the data. The qualitative variables were then reported through frequencies and proportions and the quantitative variables through measures of central tendency. Parametric tests (mean and standard deviation) were applied, as were nonparametric measures (median and range), when distribution was not normal. The statistical analysis was performed using NCSS® version 10 software.
Ethical considerationsSigned statements of informed consent were provided by a parent or guardian of the patients before the procedures. The present study is a review of medical records and the authors declare that patient confidentiality was absolutely preserved, and no data appeared that could identify them. Based on guideline 10 of the International Ethical Guidelines for Health-related Research Involving Humans prepared by the Council for International Organizations of Medical Sciences (CIOMS), in collaboration with the World Health Organization (WHO), informed consent was not required.
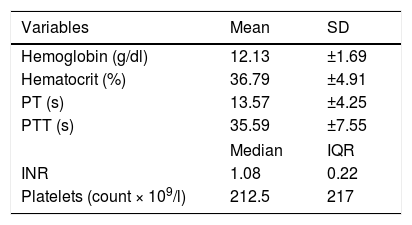
ResultsA total of 104 PLBs were performed. Two were excluded due to incomplete data in the corresponding medical records that prevented their analysis, resulting in 102 ultrasound-guided PLBs in 99 patients. The procedures were carried out, following the general recommendations of the ESPGHAN, as described in Table 1. Female sex was predominant, at 58.8%. Age ranged from 3 months to 15 years, with a median age of 72 months (±51 months). The PLBs was performed in native liver in 97 procedures (95.1%) and in a transplanted graft in 5 (4.9%). A second PLB was carried out in 3 cases during the study period. Table 2 shows the biochemical data.
General recommendations for performing PLB.
| 1. Identification of risk factors |
| Special population: patients with CKD, Alagille syndrome, breastfeeding infants < 3 months of age and low birth weight babies, patients with hematologic-oncologic diseases. PLB must be avoided in cases of ascites |
| 2. Preoperative period |
| Carry out complete blood count, complete liver function tests (that include TP, PTT, INR, fibrinogen). Determine PT, PTT, INR, and platelets within 24 h prior to the procedure. |
| Suspend antiplatelet medications for 7-10 days and resume 48-72 h after the PLB |
| Suspend warfarin 5 days prior to the PLB and resume 24 h after the procedure. |
| Obtain signed statement of informed consent. |
| 3. During the procedure |
| Patient has fasted a minimum of 4 h |
| Asepsis with alcoholic solution at the puncture site |
| Preferably use cutting needles (the standard 16 G is recommended) |
| Perform a maximum of 2 passes per PLB |
| Use general anesthesia or sedation + long-acting local anesthesia at the puncture site |
| Fix the specimen with formalin and evaluate its immediate division, in case additional special studies are required |
| 4. Post-procedure monitoring |
| Immediate compression on the biopsy site to achieve correct hemostasis |
| A minimum 2-h fast after the procedure |
| Strict monitoring of vital signs and signs and symptoms for at least 6 h |
| Hb and Ht quantification, if clinically justified |
| If bleeding is suspected, perform a new blood count and USG. |
| Avoid contact sports for at least one week |
CKD: chronic kidney disease; Hb: hemoglobin; Ht: hematocrit; INR: international normalized ratio; PLB: percutaneous liver biopsy; PT: prothrombin time; PTT: partial thromboplastin time; USG: ultrasonography.
24-h pre-procedure laboratory test data of the patients that underwent percutaneous liver biopsy.
| Variables | Mean | SD |
|---|---|---|
| Hemoglobin (g/dl) | 12.13 | ±1.69 |
| Hematocrit (%) | 36.79 | ±4.91 |
| PT (s) | 13.57 | ±4.25 |
| PTT (s) | 35.59 | ±7.55 |
| Median | IQR | |
| INR | 1.08 | 0.22 |
| Platelets (count × 109/l) | 212.5 | 217 |
INR: international normalized ratio; PT: prothrombin time; PTT: partial thromboplastin time; IQR: interquartile range; SD: standard deviation.
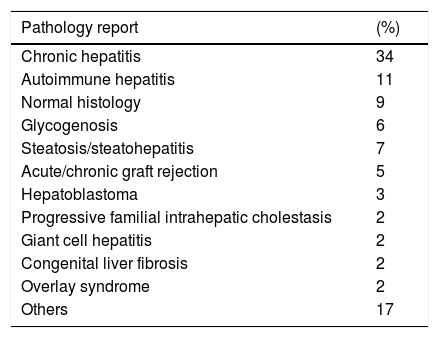
The most frequent indications for PLB were its use as a diagnostic tool in chronic liver disease of undetermined etiology, for elevated liver enzymes, and for autoimmune hepatitis in more than 65% of the cases. The remaining indications are shown in Table 3. The most common histologic diagnoses reported were chronic hepatitis (34%), autoimmune hepatitis (11%), normal histology (9%), steatosis/steatohepatitis (7%), and glycogenosis (6%). Table 4 shows the histopathologic results.
Indications for percutaneous liver biopsy.
| Indication for the procedure | No. of procedures | % |
|---|---|---|
| Autoimmune hepatitis (diagnosis and follow-up) | 24 | 23.5 |
| Transaminasemia | 22 | 21.5 |
| Chronic liver disease | 21 | 20.5 |
| Hepatosplenomegaly | 13 | 12.7 |
| Tumors and focal lesions | 8 | 7.8 |
| Chronic viral hepatitis | 6 | 5.8 |
| Graft rejection | 5 | 4.9 |
| Cholestasis | 3 | 2.9 |
Histopathologic report of the 102 liver biopsies.
| Pathology report | (%) |
|---|---|
| Chronic hepatitis | 34 |
| Autoimmune hepatitis | 11 |
| Normal histology | 9 |
| Glycogenosis | 6 |
| Steatosis/steatohepatitis | 7 |
| Acute/chronic graft rejection | 5 |
| Hepatoblastoma | 3 |
| Progressive familial intrahepatic cholestasis | 2 |
| Giant cell hepatitis | 2 |
| Congenital liver fibrosis | 2 |
| Overlay syndrome | 2 |
| Others | 17 |
Among the main risk factors identified, 4 patients presented with hematologic-oncologic diseases and one patient with chronic kidney disease. Ten patients had a platelet count below 60 × 109/l, requiring platelet transfusion before the procedure. The rest of the coagulation tests (PT, PTT, and INR) had no alterations. None of the patients that underwent PLB presented with ascites or were taking anticoagulants.
The associated comorbidities in the patients were overweight or obesity (5.8%); hematologic diseases, such as acute leukemia, bone marrow aplasia, and Hodgkin’s lymphoma (3.9%); ulcerative colitis (3.9%); systemic lupus erythematosus (3.9%); hypothyroidism (2.9%); Turner syndrome (1.9%); chronic kidney disease (1%); and polyglandular syndrome (1%).
There were immediate complications in 4 (3.9%) of the 102 biopsies performed. Two of those patients presented with bleeding at the puncture site, with a drop in hemoglobin above 2 g/dl (one required blood transfusion and the other prolonged hospital stay), one patient had a subcapsular hematoma, followed by the formation of an abscess that required percutaneous drainage, and one patient presented with a semiliquid liver collection that was managed conservatively.
The description above shows there were major complications in 2.9% of the PLBs performed. Of the 4 procedures with complications, the risk factor of a platelet count below 60 × 109/l was identified in one of the patients, whereas there was no evidence of any risk factors in the other 3 patients.
Discussion and conclusionsPLB is a safe, reliable, and minimally invasive procedure for evaluating pediatric patients with liver disease.2,3,5 It is carried out routinely at our institution through ultrasound guidance, which helps reduce the rate of associated complications and improves specimen collection.
In our study, the most frequent indication for PLB was autoimmune hepatitis, for both diagnosis and follow-up, unlike published results from other hospital centers, in which neonatal cholestasis is one of the main indications for the procedure.6–10 At our hospital, the diagnostic approach to neonatal cholestasis is open liver biopsy, which includes examination of the biliary tree, thereby ruling out bile duct atresia, the most frequent neonatal anatomic pathology.
Some studies state that sufficient tissue for histologic evaluation is obtained in 80-100% of PLBs,5,11–15 with few cases requiring a new procedure. In their study, Short et al. reported insufficient specimen collection for histopathologic analysis in 19.2% of their biopsies, resulting in the need for a second PLB in 37% of those patients and an open liver biopsy in 8%.16 In our population, a second PLB was performed in 3 patients due to insufficient sample.
No pain at the puncture site was reported in our patients, given that they routinely received a dose of a nonsteroidal anti-inflammatory drug, within the first 24 h after PLB. Almeida et al. stated that pain was present in 59% of their patients and it subsided with acetaminophen in the majority of cases.6
Bleeding is the most dreaded complication in PLB because it is the primary cause of death.17 According to a 2019 review conducted by Midia et al. on children and adults, bleeding occurred in up to 10.9% of the imaging-guided PLBs, with major bleeding events in 0.1-4.6%.18 Those results were comparable with ours, in which a bleeding event presented in 3.9% of the ultrasound-guided PLBs, with major bleeding in 2.9% (drop of 2 g/dl in hemoglobin, need for transfusion, increase in hospital stay, and/or need for surgical management).
In our patients that presented with complications, none had the risk factors described in the ESPGHAN guidelines. For example, no patient was < 2 years of age or had an INR > 1.5, which are considered risk factors for bleeding in those guidelines.5 However, one of the patients had a platelet count < 60,000 prior to the procedure, which was corrected through platelet transfusion.
Complications generally present within the first 2-4 h after the procedure,4,5,18 as was similarly detected in our population, given that they were identified on the ultrasound examination carried out immediately after the PLB. We confirmed the results reported by Almeida et al.,6 who stated that PLB can be safely performed as an outpatient procedure in patients with no comorbidities or risk factors, as is done at some hospital centers.19,20
The main limitation of our study was its retrospective design, providing limited information about the procedures performed.
In conclusion, liver biopsy continues to be an essential resource for the study of liver diseases in the pediatric population. The present study described the protocol of our hospital center, regarding ultrasound-guided liver biopsy, in an effort to confirm that PLB is a useful and reproducible procedure, with a low complication rate.
Future studies should explore the possibility of performing PLB as an outpatient procedure in Mexico, in selected pediatric patients, with 2-4 h of monitoring post-PLB, which would reduce hospital stay and its corresponding costs.
Ethical disclosuresProtection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this study/article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Hernández-Chávez E, Alfaro-Hurtado M, Sánchez-López CE, Badallo-Rivas GA, Gómez-Navarro G, Castillo-de León YA. Biopsia hepática percutánea guiada por ultrasonido en pediatría. Experiencia de 5 años en un centro de tercer nivel. Rev Gastroenterol Méx. 2022;87:170–175.