Obesity is associated with nonalcoholic fatty liver disease (NAFLD) in children. Our aim was to analyze the association of dietary and sociodemographic factors with NAFLD in obese children and adolescents.
Materials and methodsThirty-three obese patients from 6-16 years of age were included in the present analytic cross-sectional study. Obesity was diagnosed with a body mass index z-score > 2 SD. NAFLD was estimated by liver ultrasound, serum amino transferases, and elastography. The sociodemographic variables were evaluated using validated questionnaires. Diet was estimated through two 24-h recall dietary surveys focused on the quantity of food energy, simple sugars, polyunsaturated fatty acids (PUFAs), and antioxidants.
ResultsHepatic steatosis was identified by abdominal ultrasound in 13 patients (39.4%) and the serum alanine aminotransferase level was above the upper reference value in 54.2%. A higher educational level in both parents, greater monthly food expenditure, and a higher socioeconomic level were associated with NAFLD. Overall, simple sugar and saturated fat consumption was above the recommended daily intake, whereas vitamin E and PUFA consumption was below those parameters.
ConclusionsNAFLD was identified in one-third of the cases. There was an association between NAFLD and sociodemographic variables. Both groups had an increased intake of simple sugars and a reduced intake of PUFAs. The selective occurrence of NAFLD could be related to a genetic predisposition that has been demonstrated in a Mexican population.
La obesidad se asocia con el hígado graso no alcohólico (HGNA) en niños. El objetivo fue explorar la asociación de factores dietéticos y sociodemográficos con el HGNA en niños y adolescentes obesos.
Material y métodosTreinta y tres pacientes obesos de 6-16 años fueron incluidos en este estudio transversal analítico. El diagnóstico de obesidad se realizó con puntuación z del índice de masa corporal > 2 DE. El HGNA se estimó con ultrasonido hepático, aminotransferasas séricas y elastografía. Las variables sociodemográficas se evaluaron con cuestionarios validados. La dieta se estimó mediante dos encuestas dietéticas por recordatorio de 24 horas enfocadas a la cantidad de energía, azúcares simples, ácidos grasos poliinsaturados (PUFAS) y antioxidantes.
ResultadosEn 13 pacientes (39.4%) se identificó esteatosis hepática por ultrasonido abdominal; 54.2% tuvieron una concentración sérica de alanina aminotransferasa por arriba de la referencia. El nivel educativo superior de ambos padres, el mayor gasto familiar mensual en alimentos y el mayor nivel socioeconómico se asociaron con HGNA. En el grupo total, el consumo de azúcares simples y grasas saturadas fue excesivo y el consumo de vitamina E y PUFAS estuvo por debajo del parámetro.
ConclusionesSe identificó HGNA en un tercio de los casos. Se demostró asociación entre HGNA y variables sociodemográficas. Ambos grupos tuvieron una ingesta aumentada de azúcares simples y disminuida de PUFAs. La ocurrencia selectiva de HGNA se podría relacionar a predisposición genética demostrada en población mexicana.
Nonalcoholic fatty liver disease (NAFLD) is currently the most frequent cause of chronic liver disease in children and adolescents.1 The prevalence of NAFLD in pediatric ages in Mexico is unknown. In 2005, Flores-Calderón et al. found a frequency of 42.6% of NAFLD in obese children at an elementary school in Mexico City,2 and in 2016, Salcedo-Flores et al. observed a frequency of 39% in obese schoolchildren seen at a pediatric hospital.3
Although obesity is the main risk factor for developing NAFLD, not all obese patients present with the disease. Its association with dietary factors, such as high ingestion of simple sugars, particularly fructose in foods with high fructose corn syrup,4–6 and poor consumption of antioxidants, such as vitamin C,7 vitamin E,8 and omega-3 polyunsaturated fatty acids (PUFAS),7,9 has been proposed. Other authors have shown the association of a higher socioeconomic level with NAFLD, providing a distinctive epidemiologic perspective on the global situation of the disease.10
Our aim was to evaluate the association of dietary and sociodemographic factors with the presence of NAFLD in Mexican school-age children and adolescents with obesity.
Materials and methodsDesign: An analytical cross-sectional study was carried out on obese children. NAFLD was the dependent variable and the nutrition and sociodemographic characteristics were the independent variables. We included patients diagnosed with obesity, according to the World Health Organization (WHO) criteria, that were seen for the first time and that agreed to participate in the study. The sample size was calculated with a formula for the comparison of means. Numerical data were taken from two manuscripts to make two sample size calculations, one that compared the intake of fructose4 and one that studied the intake of PUFAS,9 showing differences between the obese children with NAFLD and those without NAFLD. The estimated sample size values were 34 and 29 cases, respectively. Consecutive children and adolescents with obesity (zBMI > 2 SD) from 6 to 16 years of age that were seen at an obesity clinic were included in the study. Data collection was carried out from March to November 2017.
AnthropometricsStandardization: Before the data were collected, the lead author and two collaborators performed an anthropometric standardization trial on 20 pediatric patients. Consistency (intra-group individual measurements) and validity (inter-group comparison with a gold standard) were evaluated with Pearson’s bivariate correlations; when the “r” was below 0.8, the anthropometric technique was reviewed and corrected, until the desired intra and inter-group correlations were obtained.
Weight: Weight was measured with a movable weight platform-beam scale; subjects were measured wearing a minimum of clothing and no shoes. Weight was recorded to the nearest 100 g.
Height: Height was measured and recorded to the nearest 0.1 cm, using a stadiometer with a movable block. The subjects were measured while standing, without shoes, heels together, back as straight as possible, and arms hanging freely; the head was positioned utilizing the Frankfort horizontal plane and the movable block was brought down until touching the head. Body mass index (BMI) (weight in kg/height in m2) was classified according to the 2006 WHO parameter and current WHO criteria for children over 5 years of age and adolescents.11
Diagnosis of fatty liverLiver ultrasound: Liver ultrasound was performed after an 8-hour fast, employing an ACUSON S200® HELX digital system (Siemens Healthineers, Erlangen, Germany), with the patient in the dorsal decubitus position or the left oblique position. The procedure was performed by a pediatric radiologist (ELM). The criterion for steatosis was hyperechogenic liver tissue with fine, tightly packed echoes that was identified utilizing a 5 mHz transducer for posterior beam penetration. The degree of steatosis was assessed by the fall in echo amplitude with depth (rate of posterior beam attenuation), increasing the discrepancy of echo amplitude between the liver and kidney, and the loss of echoes from the walls of the portal veins. The classification used was: grade 0, no steatosis (hepatic and renal cortex with the same echogenicity); grade 1, mild steatosis: liver slightly brighter compared with the renal cortex, clear visualization of the diaphragm and interface of the hepatic veins with sharp contours; grade 2, moderate steatosis: brighter liver at the deepest parts of the liver, diaphragm and hepatic veins still visible but with blunt contours; grade 3, severe steatosis: very bright liver, severe attenuation of the ultrasound spread beam, diaphragm or hepatic veins not visible.12–14
Liver elastography: Liver ultrasound elastography was performed to support the identification of steatosis through acoustic radiation force impulse (ARFI),15 utilizing a broadband (6 MHz) transducer (3-5.5 MHz) for the transverse scan. Ten points of the liver were recorded; five located in segment V of the right lobe and five in segment VIII of the right lobe. The results were collected by means of numerical registration in m/sec and compared according to the dichotomous ultrasound classification of steatosis. The mean normality value for identifying abnormality was 1.15 ± 0.21 m/s.15,16
Serum aminotransferases: Serum concentrations of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were carried out with an immunoenzymatic assay (Beckman Coulter AU5800, Beckman Coulter Inc., Brea, CA, USA). Mean aminotransferases were compared, according to the dichotomous classification of steatosis for ultrasound. The upper limit of normal (ULN) cut-off value was 30 mg/dL for ALT and > 40 mg/dL for AST. ALT was considered elevated when its value was twice the ULN.17
Sociodemographic variablesAn ad hoc questionnaire that included data on family income, food expenditure, and parent age, occupation, and education was completed by the parents or guardians of the children and adolescents. Socioeconomic level classification was carried out utilizing the Mexican Association of Marketing Research (AMAI, Spanish acronym) questionnaire. That instrument groups and classifies Mexican households into seven levels, according to their ability to meet the needs of their family members in terms of housing, health, energy, technology, prevention, and intellectual development.18
Dietary surveysTwo 24-hour recall surveys were applied; one on a weekday and the other on a day of the weekend. Two food-frequency surveys were utilized; one designed for foods with simple sugars and the other for the search of foods with vitamins and PUFAs. The dietary analysis was performed with the Nutritionist PRO™ Diet Analysis software program (Copyright 2016-2017 Axxya Systems LLC, Redmond, WA, USA).
Statistical analysisThe statistical analysis was carried out at the bivariate comparative and associative levels with the chi-square test and the Fisher’s test. The quantitative variables with abnormal distribution were analyzed together with their nonparametric equivalents. The statistics used to assess the strength of association of the categorical variables were the Cohen's kappa and odds ratio. The nutrition variables were compared between the study groups and with reference parameters, utilizing the single sample Student’s t test.
Ethical considerationsThe recommendations of the Declaration of Helsinki, amended by the World Medical Association’s 64th Annual Assembly in 2013, were followed. The patients, or their parents or guardians, signed written statements of informed consent. The protocol was approved by the hospital’s Research and Ethics Committee (# 0117/16).
ResultsPatients: Thirty-three consecutive patients that met the inclusion criteria participated in the study. The median of the BMI z-score (zBMI) was 2.9, with an interquartile range (IQR) of 2.4-3.3. In 17 patients (51.5%), the zBMI was between +2 and +3 SD, and was above +3 SD in 16 patients (48.5%).
Hepatic steatosisUltrasound: In 13 cases (39.4%), abdominal ultrasound identified fatty liver, according to the criteria published by van Werven and Mustapic.13,14 In 10 patients (76.9%), the steatosis was considered mild (grade 1) and in the remaining 3 patients (23.1%), it was considered moderate (grade 2). The frequency of steatosis was higher in males (21.2% vs. 18.2%) with no statistical difference when compared with females (p = 0.284).
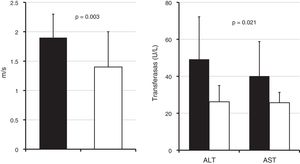
Liver elastography: The elastography results are presented in Fig. 1. The group with steatosis had statistically significant higher m/sec values. Nineteen cases (57.6%) were located above the ULN. There was a statistical association between the ultrasound diagnosis of NAFLD and elastography above the reference values (p = 0.001). Concordance of elastography results with positive or negative NAFLD by ultrasound is presented in Table 1. Fibrosis was not identified in any of the cases, with or without NAFLD.
Concordance between liver ultrasound, serum ALT concentration, and elastography performed on 33 children with obesity in relation to fatty liver screening
| Diagnostic test | NAFLD+ (ultrasound) | NAFLD– (ultrasound) | kappa | p | |||
|---|---|---|---|---|---|---|---|
| n | (%) | n | (%) | ||||
| ALT (U/L) | > 54 | 7 | (53.8) | 0 | – | 0.590 | <0.001 |
| < 54 | 6 | (46.2) | 21 | (100) | |||
| Elastography (m/s) | > 1.36 | 11 | (84.6) | 5 | (23.8) | 0.582 | 0.01 |
| < 1.36 | 2 | (15.6) | 16 | (76.2) | |||
ALT: Alanine aminotransferase; NAFLD: Nonalcoholic fatty liver disease.
Aminotransferases: There were higher serum concentrations of both AST and ALT in the group with steatosis identified by liver ultrasound, with a statistical difference in the comparison of the means (Fig. 1). The Cohen’s kappa concordance value for steatosis identified or ruled out by ultrasound and ALT, as dichotomous variables, are presented in Table 1.
Sociodemographic variablesNineteen patients (57.6%) were females. Their overall median age was 10 years (IQR = 9-13 years), with no differences between the study groups. According to the patients and their parents or guardians, there was no alcohol consumption in the year prior to the study on the part of the participants.
The comparison of the quantitative sociodemographic variables between the study groups is presented in Table 2. Family monthly income was 21% higher in the NAFLD group, when compared with the non-NAFLD group, with no statistically significant difference. Monthly food expenditure was 16% higher in the NAFLD group, with statistical difference.
Quantitative sociodemographic variables collected from 33 families of obese school-age children and adolescents. The results are presented as medians and interquartile range. Statistics: Mann-Whitney U test
| Variable | NAFLD+ (n = 13) | NAFLD– (n = 20) | p | ||
|---|---|---|---|---|---|
| Median | (IQR) | Median | (IQR) | ||
| Mother’s age | 36.5 | (31.8-41.2) | 35 | (29.5-42) | 0.740 |
| Father’s age (years) | 40 | (35.5-43.5) | 37 | (32.-42) | 0.768 |
| Number of children in the family | 2 | (2-3) | 2 | (2-3) | 0.470 |
| Father’s monthly income (MXN) | 4800 | (2300-6400) | 5000 | (3925-9125) | 0.201 |
| Family’s monthly income (MXN) | 6100 | (4400-10000) | 4800 | (4000-9000) | 0.485 |
| Monthly food expenditure (MXN) | 4800 | (4000-7500) | 4000 | (2725-4800) | 0.021 |
IQR: Interquartile range (75th-25th percentile); MXN: Mexican pesos; NAFLD: Nonalcoholic fatty liver disease.
The comparison of the categorical sociodemographic variables between the study groups is presented in Table 3. The educational level of both parents, the socioeconomic level determined through the AMAI instrument, and monthly food expenditure, treated as a categorical value, were all significantly higher in the NAFLD group.
Categorical sociodemographic variables collected from 33 families of obese school-age children and adolescents. The results are presented as frequencies and percentages. Statistics: chi-square test, odds ratio (OR), and 95% confidence interval (95% CI)
| Sociodemographic variables | NAFLD+ (n = 13) | NAFLD– (n = 20) | p | OR | 95% CI | |||
|---|---|---|---|---|---|---|---|---|
| n | (%) | n | (%) | |||||
| Educational level of both parents | Elementary and junior high school | 10 | (15.4) | 29 | (44.6) | 0.009 | 3.9 | 1.2-13.2 |
| High school and graduate school | 15 | (23.1) | 11 | (16.9) | ||||
| Socioeconomic levela | A/B, C+, C | 6 | (19.3) | 6 | (19.3) | 0.003 | 18 | 1.8-181.3 |
| C-, D+, D, E | 1 | (58.1) | 18 | (3.3) | ||||
| Monthly food expenditure | <5000 | 6 | (50) | 16 | (84.2) | 0.032b | 8.5 | 1.3-54.1 |
| < 5000 | 6 | (50) | 3 | (15.8) | ||||
| Type of family | Nuclear | 5 | (19.2) | 8 | (30.8) | 0.691 | 0.7 | 0.1-3.5 |
| Extended, reconstituted | 6 | (19.2) | 7 | (26.9) | ||||
The comparison of the food energy, macronutrient, and micronutrient intake between the study groups is presented in Table 4. The mean two-day intake of energy, proteins, and carbohydrates was higher in the NAFLD group, with no statistical differences. The amount of lipids and their fractions and other micronutrients, such as vitamins C and E, showed no statistical differences.
Mean 24-h energy and nutrient intake estimated by two 24-h recall questionnaires applied to 33 obese school-age children and adolescents with and without hepatic steatosis determined through ultrasound. Results are presented as mean and SD. Statistics: Student’s t test for independent samples
| Twenty-four hour nutrient intake | NAFLD+ (n = 13) | NAFLD– (n = 20) | p | ||
|---|---|---|---|---|---|
| X | (SD) | x | (SD) | ||
| Energy (kcal) | 1824.9 | 564.1 | 1782.9 | 685.9 | 0.856 |
| Protein (g) | 71.2 | 16.7 | 68.4 | 27 | 0.739 |
| Carbohydrates (g) | 207.8 | 76.3 | 200.2 | 78.2 | 0.785 |
| Lipids (g) | 70.4 | 32.9 | 74.9 | 34.9 | 0.715 |
| Sugar (g) | 88.2 | 54.8 | 82 | 34.9 | 0.694 |
| Sucrose (g) | 7.8 | 5.7 | 12.3 | 12.4 | 0.232 |
| Sugar (%) | 18.9 | 9.8 | 19.2 | 7.1 | 0.901 |
| SFAs (g) | 22.9 | 14.1 | 22.2 | 11.2 | 0.874 |
| MFAs (g) | 21.1 | 8.8 | 22.6 | 10.7 | 0.664 |
| PUFAs (g) | 13.7 | 11.5 | 14.6 | 8.3 | 0.794 |
| Omega-6 PUFAs (%) | 10.2 | 10.8 | 11.5 | 7.7 | 0.702 |
| Alpha-linolenic PUFA (%) | 0.9 | 0.8 | 0.7 | 0.4 | 0.259 |
| EPA (g) | 0.01 | 0.03 | 0.01 | 0.01 | 0.440 |
| DHA (g) | 0.1 | 0.1 | 0.1 | 0.2 | 0.804 |
| Omega-3 fatty acid total (%) | 1.1 | 0.8 | 0.8 | 0.5 | 0.309 |
| Vitamin C (mg) | 62.4 | 68.6 | 69.4 | 70.2 | 0.782 |
| Vitamin E (mg) | 3.1 | 9.1 | 0.8 | 1.3 | 0.272 |
DHA: docosahexaenoic fatty acid; EPA: eicosapentaenoic fatty acid; MFAs: monounsaturated fatty acids; PUFAs: polyunsaturated fatty acids; SFAs: saturated fatty acids.
The comparison of the average intake of simple sugars, fatty acid fractions, vitamin C, and vitamin E with the mean recommended intake (MRI), according to Food and Agriculture Organization (FAO) and WHO parameters, matched by age and sex in each study group, is presented in Table 5. Simple sugar intake was above the MRI in both groups. The amount of saturated fatty acids (SFAs) was above the MRI, whereas PUFA intake was below the MRI in both groups. Vitamin C intake was within the reference limits in all cases.
Comparison of two-day dietary intake of selected nutrients estimated by 24-h recall surveys with the reference parameter performed on 33 pediatric patients with obesity, 13 with NAFLD and 20 without NAFLD. Statistics: Student's t test for one sample
| Nutrients | Age (years) | MRI | Mean difference | NAFLD+ | p | NAFLD– | Mean difference | p | ||
|---|---|---|---|---|---|---|---|---|---|---|
| x | (SD) | x | (SD) | |||||||
| SFAs (%) | 8a | 2.8 | 10.8 | (3.2) | 0.008 | 11 | (2.4) | 3 | <0.001 | |
| MFAs (%) | 11a | -1.66 | 9.44 | (4.8) | 0.262 | 10.3 | (3.1) | -0.7 | 0.262 | |
| PUFAs (%) | 11a | -5.7 | 6.3 | (3.3) | <0.001 | 7.6 | (3.7) | -3.4 | 0.001 | |
| Omega-3 PUFAs (%) | 0.5a | -0.1 | 0.4 | (0.3) | <0.001 | 0.4 | (0.2) | -0.1 | <0.001 | |
| Simple sugars (%) | 10b | 8.9 | 18.9 | (9.8) | 0.007 | 19.3 | (7.1) | 9.3 | <0.001 | |
| 6-8 | 7 b | – | – | – | – | 0.62 | (0.6) | -6.38 | <0.001 | |
| 9-13 | 11b | -10.44 | 0.56 | (0.5) | <0.001 | 0.5 | (0.5) | -10.5 | <0.001 | |
| 14-16 | 15b | – | – | – | – | 1.4 | (2.4) | -13.6 | <0.001 | |
| Vitamin C (mg) | ||||||||||
| Females | 4-8 | 25c | – | – | – | – | 79.1 | (52.5) | 54.1 | 0.383 |
| 9-13 | 45c | 10.5 | 55.5 | (65) | 0.736 | 74 | (96.7) | 63.5 | 0.495 | |
| 14-18 | 57c | – | – | – | – | 69.7 | (73.1) | 11.3 | 0.717 | |
| Males | 4-8 | 25c | 54.5 | 79.5 | (86.4) | 0.536 | 35.1 | (20.1) | 10.1 | 0.618 |
| 9-13 | 45 c | 26.3 | 71.3 | (85.1) | 0.527 | 48.4 | (49.5) | 3.4 | 0.900 | |
| 14-18 | 65c | – | – | – | – | – | – | – | – | |
| Vitamin E (mg) | 6-8 | 7d | – | – | – | – | 0.62 | (0.6) | -6.38 | <0.001 |
| 9-13 | 11d | -10.44 | 0.56 | (0.5) | <0.001 | 0.5 | (0.5) | -10.5 | <0.001 | |
| 14-16 | 15d | – | – | – | – | 1.4 | (2.4) | -13.6 | <0.001 | |
MFAs: monounsaturated fatty acids; MRI: mean recommended intake; PUFAs: polyunsaturated fatty acids; SFAs: saturated fatty acids.
The available information related to the prevalence of NAFLD in Mexican children corresponds to isolated studies that were not designed as prevalence studies and only report frequencies.2,3 Regarding the number of cases of NAFLD, our study was conducted on a sample of patients from an obesity clinic that were not randomly selected, implying selection bias. However, the proportion of NAFLD was similar to that in the Flores and Salcedo studies2,3 and occurred in more than one-third of the cases studied. Those data suggest that the number of obese children in Mexico with NAFLD may be meaningful from both a clinical and epidemiologic viewpoint and could denote a public health problem.
Several studies have shown a greater frequency of NAFLD in males,10,19,20 which has been explained in adult patients by the protective effect estrogen has in women, as well as its facilitating the function of insulin, and the potentially negative effect of androgens in men. In the present study, although that association was not demonstrated, by doubling the number of cases in a theoretical exercise, we observed a significantly higher risk for developing NAFLD in boys. Another explanation for a difference between sexes could be the increased adiposity in obese boys observed in studies conducted at elementary schools in Guadalajara in recent years.21,22
More than half of the patients with steatosis had a slight elevation of transferases, which can be interpreted as mild acute hepatocellular damage. Our patients with steatosis had significantly higher ALT and AST values, which is consistent with studies in similar populations.3 However, there are studies in which that association was not found.23 Whether or not aminotransferases are elevated is probably related to the natural history of the disease, outcome time, and particularly to the genetic characteristics that determine the increased likelihood of some obese subjects having greater hepatocyte damage, such as the I148 M PNPLA3 variant that has been associated with elevated ALT levels in overweight/obese Mexican children.24,25 In 2010, Davis et al. showed that the Hispanic population frequently presents a polymorphism that can contribute to the accumulation of fat in the liver, especially due to the high consumption of sugars.26 In our study, simple sugar consumption was higher than the WHO recommendation, in patients both with and without steatosis.
Vitamin E intake in the two study groups was significantly lower than the recommended amount. Similar findings were observed in the study conducted by Vos et al.8 It has been suggested that patients with NAFLD and steatohepatitis require a greater amount of vitamin E to counteract oxidative stress. Even when those patients take an amount of vitamin E equivalent to that consumed by healthy persons, they can experience a net shortage, with reduced serum levels.27,28
The consumption of omega-3 PUFAs was significantly lower than the recommended amounts, in both groups. In contrast, SFA intake was significantly higher than the recommended amounts in the two groups. That dietary lipid imbalance, rather than total lipid consumption, could act as a trigger in the pathophysiology of NAFLD.
Obesity is the main factor associated with NAFLD in the pediatric population.19 The trend in the increasing incidence of the pathology over the past few decades strongly suggests the participation of environmental factors.10 The associations of a higher educational level, higher socioeconomic level, and higher monthly food expenditure found in our study, suggest the participation of family lifestyle in the susceptible cases. However, differences were identified when the NAFLD group was compared with the reference parameter regarding SFAs, PUFAs, simple sugars, and vitamin E, factors that have been associated with NAFLD in several studies.4–9,26–31 Other differences between the study groups may be due to an inadequate sample-related type II error. Some dietary recommendations for NAFLD can be concluded from our study. PUFA and antioxidant consumption should be increased in obese children, and sugar intake decreased, in addition to the general obesity recommendations. Those dietary suggestions have been shown to reduce liver inflammation, ALT, and changes identified in imaging studies in children with NAFLD.32
Another approach could be individual susceptibility related to the abovementioned genetic predisposition reported in a Mexican population.33 Research related to the monogenic and polygenic inheritance associated with obesity is in full development, although the current findings only explain a modest part of its heritability.33,34
Financial disclosureMLP received a Master's degree scholarship from the PNPC program of the Mexican Council of Science and Technology (CONACyT).
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: León-Plascencia M, Larrosa-Haro A, Romero-Velarde E, Bravo-Núñez EC, López-Marure E. Factores alimenticios y sociodemográficos asociados con el hígado graso no alcohólico en pacientes pediátricos obesos. Revista de Gastroenterología de México. 2021;86:236–243.