A 59-year-old man that worked as a firefighter had a remarkable past medical history of prostatic syndrome and gastroesophageal reflux, with no previous history of venous thromboembolism (VTE). The patient had stopped smoking one year before, with a smoking index of 10 packs/year. He led an active life, with no other cardiovascular risk factors. The patient had no recent history of immobilization or prolonged travel. He did not have varicose veins in his lower limbs and his BMI was 24.8kg/m2.
Six months prior to the present episode, the patient was evaluated at a gastroenterology outpatient clinic in another healthcare center for gastroesophageal reflux symptom worsening. Upper endoscopy was performed, showing a submucosal lesion in the gastric fundus. Endoscopic ultrasound revealed a hypoechogenic, 18 x 12mm tumor with regular borders. The lesion was biopsied and the anatomopathologic study of the sample revealed spindle cells arranged in fasciculi and interlacing bundles. The tumor stained positive for c-KIT (CD117) and CD34, and negative for alpha smooth muscle actin, desmin, and S-100, confirming the diagnosis of gastrointestinal stromal tumor (GIST). The Ki-67 proliferative index was less than 1%. The treating gastroenterologists decided on conservative tumor management with annual endoscopic follow-up.
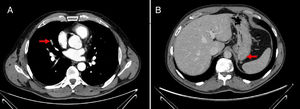
The patient arrived at the emergency room of our hospital due to sudden onset of dyspnea and chest pain. Upon physical examination, his blood pressure was 144/76, heart rate was 78 beats per minute, oxygen saturation when breathing ambient air was 97%, and respiratory rate was 15 breaths per minute. His lower extremities were normal, with no signs of deep vein thrombosis (DVT) and no varicose veins. The rest of the examination was normal. Blood analysis results were: hemoglobin 15.9mg/dl, 304,000 platelets/mm3, creatinine 0.78mg/dl, and D-dimer 1344 ng/ml. Arterial blood gases were: pO2 74mmHg, pCO2 35mmHg, and oxygen saturation 95%. A chest computed tomography (CT) scan revealed bilateral filling defects in multiple sub-segmental arteries (fig. 1). Weight-adjusted enoxaparin was then begun, and the patient was admitted to the Venous Thromboembolism Unit.
An echocardiogram showed a mildly dilated right ventricle with normal ventricular function. Estimated pulmonary artery systolic pressure (PASP) was 43mmHg. An abdominal CT scan was also performed to evaluate the extension of the gastric tumor, showing a solid 18mm lesion in the gastric fundus, with no adenopathies or metastasis. A Doppler ultrasound of the lower extremities showed no echographic findings of DVT.
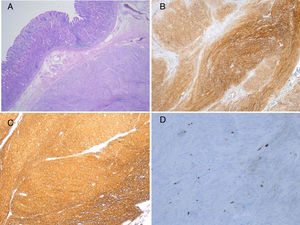
The patient's clinical situation improved, and he was discharged after 5 days. Since the tumor was possibly related to the development of VTE, the case was re-evaluated together with the General Surgery and Oncology services, and the decision was made to defer surgery until completing 3 months of anticoagulant treatment. The patient was treated with enoxaparin for 3 months, and then scheduled for partial gastrectomy. Thrombophilia testing that included antithrombin, homocysteine, protein C, protein S, antiphospholipid antibodies, factor V Leiden, and prothrombin mutation, was negative. The procedure was performed with good outcome and the sample was sent to our Pathology Department (fig. 2). Seven days after surgery, treatment with enoxaparin was discontinued and rivaroxaban 20mg/day was started.
A) Spindle cell GIST composed of fascicles of uniform cells with pale eosinophilic cytoplasm (hematoxylin-eosin, original magnification x200). B) C-KIT: strong and diffuse cytoplasmic expression in GIST (original magnification x200). C) CD34: strong and diffuse cytoplasmic expression in GIST (original magnification x200). D) KI-67: Isolated cells with nuclear expression in GIST (original magnification x400).
Patient progression was favorable, with the gradual disappearance of dyspnea on exertion. A control echocardiogram revealed a non-dilated right ventricle with normal PASP. Three months after surgery, anticoagulant treatment was discontinued. At present, after 2 years of follow up, the patient remains asymptomatic with no new episodes of VTE after anticoagulation completion.
GISTs are the most common mesenchymal tumors of the gastrointestinal tract. Their initial histologic label was leiomyosarcoma, but after the identification of unique activating mutations in the KIT gene, they were classified as a distinct entity.1 Treatment of GIST may involve surgery and/or the use of tyrosine kinase inhibitors. For tumors 2cm or smaller, with a mitotic index of 5 or less, the indication for surgery remains controversial, given that they appear to have low rates of progression and metastasis. Endoscopic surveillance is an option in those patients.2
Cancer is known to be a risk factor of the first order for the development of VTE. Approximately 20% of new cases of VTE are related to cancer. However, the direct association between GIST and VTE remains unclear. Tumors contribute to VTE through multiple mechanisms. Mucinous adenocarcinomas secrete abnormal glycosylated mucins that react with leukocytes and aid in the formation of microthrombi, creating a prothrombotic state. Pancreatic, colorectal, or hepatocellular tumors express tissue factor.3
In our case, the complete evaluation of the patient did not reveal any associated risk factors for thrombosis, other than GIST, suggesting it was the primary cause. To the best of our knowledge, there is only one case in the literature that highlights the association between those rare tumors and VTE.3 Melichar et al. reported a case of lethal venous thromboembolism in a patient with GIST, but it was caused by mechanical compression of the common iliac vein by the tumor.4
For cancer-associated thrombosis (CAT), current guidelines recommend anticoagulant therapy with low-molecular-weight heparin over oral anticoagulants. Concerning the duration of treatment for CAT, anticoagulant therapy is recommended for as long as the disease is considered active.5–7 Thus, in our patient, the development of VTE conditioned a change in the surgical strategy for GIST. According to our criteria, anticoagulant therapy should be extended in the presence of active cancer. Tumor resection made it possible to discontinue anticoagulant therapy, thereby reducing the risk of long-term bleeding complications.
We presented herein a case of association between GIST and VTE, in which the diagnosis of VTE led to a change in tumor management that reduced the duration of anticoagulant therapy. Because the association between those two entities is not clearly established, further studies evaluating the frequency of VTE in patients with GIST are needed.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Financial disclosureNo financial support was received in relation to this study/article.
Conflict of interestThe authors declare that there is no conflict of interest.
Please cite this article as: Demelo-Rodríguez P, Lavilla Olleros C, Martín Higueras E, Peligros I, del Toro-Cervera J. Tumor del estroma gastrointestinal como causa de trombosis asociada a cáncer. Revista de Gastroenterología de México. 2019;84:250–252.